Optimizing 4-CNAB Production for Scalable Oral Peptide Delivery Systems
Optimizing 4-CNAB Production for Scalable Oral Peptide Delivery Systems
The rapid advancement of oral peptide therapeutics, particularly in the realm of GLP-1 receptor agonists, has placed unprecedented demand on high-performance absorption enhancers. Patent CN111925298B, published in May 2023, introduces a groundbreaking preparation method for 4-CNAB (Sodium 4-(2-hydroxy-4-chlorobenzoylamino) butyrate), a critical analogue to the widely recognized SNAC excipient. This technology addresses long-standing bottlenecks in the synthesis of this pharmaceutical intermediate, offering a robust pathway for manufacturers seeking to support the next generation of oral biologics. By fundamentally re-engineering the reaction conditions and purification protocols, this patent provides a viable solution for producing high-purity materials essential for gastrointestinal drug delivery systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
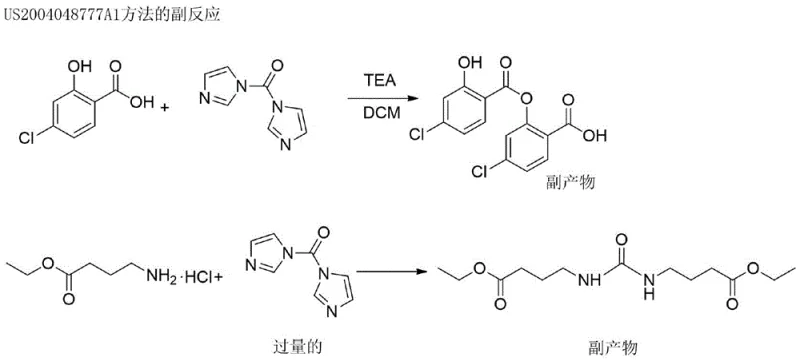
Prior art, specifically referenced in patent US2004048777A1, established the foundational synthetic route for 4-CNAB but suffered from severe inefficiencies that hindered commercial viability. The conventional method relied on a standard amide coupling followed by ester hydrolysis and salification, yet the overall yield for the first two critical steps was abysmally low, hovering around merely 37%. A primary culprit for this inefficiency was the difficulty in removing unreacted 4-chlorosalicylic acid, which persisted as a stubborn impurity at levels reaching approximately 5% in the unpurified free acid stage. Furthermore, the final salification step using acetone resulted in the formation of a jelly-like substance that was practically impossible to filter using standard industrial suction filtration equipment, creating a significant processing bottleneck.
The Novel Approach
The innovative methodology disclosed in CN111925298B systematically dismantles these barriers through precise chemical engineering and solvent optimization. By introducing sodium bentonite during the activation phase, the new process effectively suppresses the intermolecular condensation of 4-chlorosalicylic acid, a side reaction that previously wasted valuable starting materials and complicated purification. This strategic modification, combined with a controlled pH workup between 4.5 and 5, allows for the direct precipitation of high-purity intermediates, bypassing the need for multiple recrystallizations. Consequently, the combined yield for the initial synthesis and hydrolysis steps surges to a range of 64.2% to 73.7%, representing a near-doubling of efficiency compared to legacy methods while ensuring product purity exceeds 99.7%.
Mechanistic Insights into Sodium Bentonite-Assisted Amidation
The core chemical breakthrough lies in the modulation of the activation environment during the formation of the amide bond. In traditional carbonyldiimidazole (CDI) mediated couplings, the carboxylic acid can often react with itself or form symmetric anhydrides, especially when stoichiometry is not perfectly managed. The addition of sodium bentonite acts as a heterogeneous promoter that likely stabilizes the reactive acyl imidazole intermediate, preventing the self-condensation pathway that leads to ester byproducts. This ensures that the CDI is utilized almost exclusively for the activation of the 4-chlorosalicylic acid intended for coupling with the ethyl 4-aminobutyrate hydrochloride. The reaction is conducted in dichloromethane at mild temperatures between 20°C and 30°C, preserving the integrity of the sensitive functional groups while driving the reaction to completion over a 22 to 30-hour period.
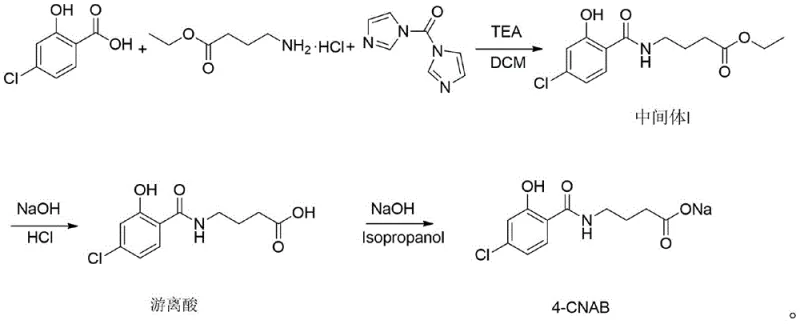
Impurity control is further enhanced through a sophisticated aqueous workup strategy that leverages pH-dependent solubility differences. After the initial coupling, the reaction mixture is treated with dilute hydrochloric acid to precipitate specific byproducts, which are removed via filtration before the organic phase is extracted. The subsequent wash with dilute ammonia water is critical; it selectively removes unconverted 4-chlorosalicylic acid, which is the primary impurity concern identified in prior art. This targeted removal at the intermediate stage prevents the carryover of impurities into the final hydrolysis step, thereby eliminating the need for the cumbersome methanol-water recrystallization previously required. The result is a free acid intermediate that is not only chemically pure but also physically conducive to downstream processing.
How to Synthesize 4-CNAB Efficiently
The synthesis of 4-CNAB via this optimized route involves three distinct operational phases that transition smoothly from coupling to salification. The process begins with the activation of 4-chlorosalicylic acid in dichloromethane, followed by the controlled addition of the amine component to generate Intermediate I. The second phase involves the hydrolysis of the ethyl ester under basic conditions, followed by precise acidification to isolate the free acid. The final phase converts this free acid into the stable sodium salt using isopropanol as the crystallization solvent. For detailed standard operating procedures and specific parameter settings, please refer to the standardized synthesis guide below.
- Activate 4-chlorosalicylic acid with CDI and sodium bentonite in DCM, then react with ethyl 4-aminobutyrate hydrochloride to form Intermediate I.
- Hydrolyze Intermediate I with sodium hydroxide, adjust pH to 4.5-5 to precipitate high-purity free acid.
- Convert free acid to sodium salt in isopropanol to ensure easy filtration and drying of the final 4-CNAB product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel manufacturing process offers substantial strategic advantages beyond mere technical metrics. The elimination of complex recrystallization steps and the replacement of problematic solvents directly translate to a streamlined production workflow that reduces operational overhead. By avoiding the formation of jelly-like products that clog filtration systems, manufacturers can significantly reduce batch cycle times and minimize equipment downtime associated with cleaning and maintenance. This operational fluidity ensures a more predictable production schedule, which is critical for maintaining the continuity of supply required by large-scale pharmaceutical clients developing oral peptide formulations.
- Cost Reduction in Manufacturing: The dramatic improvement in yield from approximately 37% to over 64% fundamentally alters the cost structure of the raw material. Higher yields mean less starting material is required to produce the same amount of final product, directly lowering the variable cost per kilogram. Additionally, the removal of the methanol-water recrystallization step reduces solvent consumption and waste disposal costs, contributing to significant overall cost savings. The ability to filter the final product easily also reduces labor costs associated with manual intervention or specialized drying processes required for gelatinous materials.
- Enhanced Supply Chain Reliability: The robustness of this synthesis route enhances supply chain resilience by reducing the risk of batch failures due to purification issues. The use of common, readily available solvents like dichloromethane and isopropanol mitigates the risk of supply disruptions associated with specialty reagents. Furthermore, the simplified workup procedure allows for faster turnaround times between batches, enabling suppliers to respond more agilely to fluctuations in market demand. This reliability is paramount for pharmaceutical companies that require consistent quality and timely delivery to meet their own regulatory and commercial obligations.
- Scalability and Environmental Compliance: From a scale-up perspective, the switch to isopropanol for the final salification step is a game-changer for industrial production. Isopropanol is easier to recover and recycle compared to mixed solvent systems, aligning better with green chemistry principles and environmental regulations. The process generates less hazardous waste due to higher efficiency and fewer purification cycles, simplifying compliance with increasingly stringent environmental standards. This scalability ensures that the technology can be seamlessly transferred from pilot plant to multi-ton commercial production without encountering the physical limitations that plagued previous methods.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 4-CNAB. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their formulation pipelines.
Q: How does the new method improve yield compared to US2004048777A1?
A: The new method utilizes sodium bentonite to prevent self-condensation of 4-chlorosalicylic acid, increasing the combined yield of the first two steps from 37% to over 64%.
Q: Why is isopropanol preferred over acetone for the salification step?
A: Acetone produces a jelly-like product that is difficult to filter, whereas isopropanol allows for the formation of a crystalline solid that is easily filtered and dried on a large scale.
Q: What is the purity level achievable with this optimized process?
A: The optimized process consistently achieves a product purity of greater than 99.7%, significantly reducing the need for complex recrystallization steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-CNAB Supplier
As the global demand for oral peptide delivery systems continues to surge, securing a supply of high-quality absorption enhancers like 4-CNAB is critical for pharmaceutical innovation. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of 4-CNAB meets the exacting standards required for clinical and commercial applications. We understand the complexities of scaling amide coupling reactions and crystallization processes, positioning us as an ideal partner for your long-term supply needs.
We invite you to collaborate with our technical procurement team to explore how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this high-efficiency manufacturing method. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your volume and quality expectations. Let us help you accelerate your development timeline with a reliable and cost-effective supply of this vital pharmaceutical intermediate.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
