Advanced Bromopentafluorobenzene Manufacturing: Leveraging BrCl Technology for Commercial Scale-Up
Advanced Bromopentafluorobenzene Manufacturing: Leveraging BrCl Technology for Commercial Scale-Up
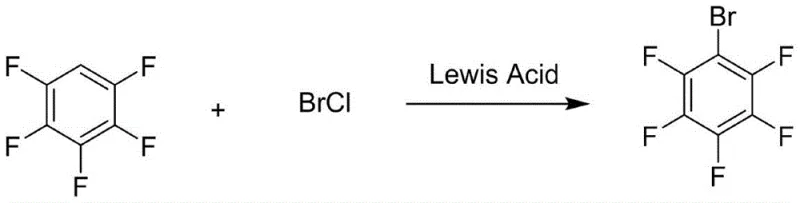
The global demand for electron-deficient aromatic compounds continues to surge, driven by their critical roles in pharmaceutical intermediates, agrochemical formulations, and advanced liquid crystal materials. Within this landscape, the synthesis of bromopentafluorobenzene stands out as a pivotal process, yet traditional methodologies have long been plagued by inefficiencies and prohibitive costs. Patent CN109438168B introduces a transformative approach that fundamentally alters the economic and technical feasibility of producing this high-value intermediate. By shifting from elemental bromine to an in-situ generated bromine chloride (BrCl) system, this technology addresses the chronic issue of low atom economy that has hindered the industry for decades. The innovation lies not merely in a reagent swap, but in a complete re-engineering of the electrophilic substitution mechanism to maximize resource utilization. For R&D directors and procurement strategists alike, this patent represents a significant opportunity to optimize supply chains and reduce the cost basis of complex fluorinated building blocks. The following analysis dissects the technical nuances and commercial implications of this novel synthetic route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of bromopentafluorobenzene has relied on several distinct pathways, each carrying substantial drawbacks that impact both operational expenditure and environmental compliance. Early methods, such as those described in CN102531832, utilized potassium bromate as the brominating source; however, this approach is economically unviable due to the high cost of the oxidant and the generation of significant inorganic salt waste. Alternative routes involving phosphorus tribromide, as seen in CN104529698, face severe supply chain bottlenecks because the precursor, pentafluorophenol, is difficult to source in bulk quantities and commands a premium price. Perhaps the most common industrial attempt involves direct bromination with elemental bromine (Br2), referenced in JP06-065120, but this method suffers from a fundamental stoichiometric inefficiency. In a standard electrophilic aromatic substitution with Br2, only one bromine atom is incorporated into the organic framework, while the other is lost as hydrogen bromide gas, resulting in a theoretical bromine utilization rate of merely 50%. This inherent waste translates directly into higher unit consumption of raw materials and increased burdens on waste treatment facilities, making these conventional processes increasingly unsustainable in a cost-sensitive market.
The Novel Approach
The methodology disclosed in patent CN109438168B circumvents these historical inefficiencies by employing bromine chloride (BrCl) as the active brominating species. This interhalogen compound is generated in situ by reacting chlorine gas with bromine in a halogenated solvent, creating a highly reactive electrophile where the bromine atom exists in a +1 oxidation state. Unlike elemental bromine, where the bond cleavage leads to one useful bromine cation and one wasted bromide anion, the BrCl mechanism allows for the complete utilization of the bromine molecule. The chlorine atom acts as the leaving group, forming HCl which is easier to manage and absorb than HBr, while the bromine is quantitatively transferred to the pentafluorobenzene ring. This shift effectively doubles the theoretical efficiency of bromine usage, drastically reducing the unit consumption of this expensive halogen. Furthermore, the reaction conditions are mild and adaptable, utilizing common organic solvents like chloroform or dichloroethane, which facilitates seamless integration into existing fine chemical manufacturing infrastructure without requiring exotic equipment or extreme operating parameters.
Mechanistic Insights into Lewis Acid-Catalyzed Electrophilic Substitution
The core of this technological advancement lies in the precise manipulation of electrophilic aromatic substitution on a highly deactivated ring system. Pentafluorobenzene is an electron-deficient substrate due to the strong inductive withdrawal of the five fluorine atoms, making it significantly less reactive towards electrophiles compared to benzene. To overcome this kinetic barrier, the process employs a Lewis acid catalyst to activate the bromine chloride complex. The Lewis acid coordinates with the chlorine end of the BrCl molecule, polarizing the Br-Cl bond and facilitating the generation of a potent bromonium ion equivalent (Br+). This activated species is sufficiently electrophilic to attack the pi-system of the pentafluorobenzene, forming a sigma complex (arenium ion). The subsequent deprotonation restores aromaticity, yielding the desired bromopentafluorobenzene. The patent highlights that without a catalyst, the reaction yield is negligible (less than 10%), underscoring the critical role of the Lewis acid in lowering the activation energy of the rate-determining step. This mechanistic understanding allows for the fine-tuning of catalyst loading to balance reaction rate against cost.
Impurity control is another critical aspect of the mechanism, particularly regarding the suppression of competitive chlorination. Since chlorine is present in the reaction mixture, there is a thermodynamic possibility of forming pentafluorochlorobenzene as a byproduct. However, the choice of catalyst plays a decisive role in selectivity. While traditional metal halides like aluminum trichloride or ferric trichloride provide good activity, they can sometimes promote side reactions or require rigorous quenching steps that generate metal-laden wastewater. The patent reveals a surprising finding regarding organoboron catalysts, specifically tris(pentafluorophenyl)boron (B(C6F5)3). This specialized Lewis acid not only accelerates the reaction rate dramatically—completing conversion in just 2 hours compared to 10-12 hours for metal salts—but also enhances regioselectivity and chemoselectivity. The unique electronic properties of the perfluorinated boron center appear to favor bromine transfer over chlorine transfer, resulting in product purity exceeding 99.7% with chlorinated impurities reduced to trace levels (0.01%). This level of purity is essential for downstream applications in pharmaceuticals and liquid crystals, where trace halogens can poison subsequent catalytic steps or degrade material performance.
How to Synthesize Bromopentafluorobenzene Efficiently
Implementing this synthesis requires careful attention to the sequential addition of reagents and temperature control to ensure safety and maximize yield. The process begins with the generation of the brominating agent, followed by the catalytic coupling with the aromatic substrate. Detailed operational parameters, including specific molar ratios and thermal profiles, are critical for reproducing the high yields reported in the patent examples. Operators must ensure that the exothermic formation of BrCl is managed under cooling to prevent volatilization losses, and that the subsequent bromination is maintained within the optimal window of 30°C to 50°C to balance kinetics and selectivity. For a comprehensive breakdown of the standardized operating procedures and safety protocols required for this transformation, please refer to the technical guide below.
- Preparation of Bromine Chloride: React chlorine gas with bromine in a halogenated solvent at low temperatures (-10°C to 10°C) to generate the active brominating agent.
- Catalytic Bromination: Introduce the bromine chloride solution to pentafluorobenzene in the presence of a Lewis acid catalyst (such as AlCl3 or specialized boron compounds) at 30-50°C.
- Isolation and Purification: Perform post-treatment to remove catalyst residues, followed by reduced pressure distillation to isolate high-purity bromopentafluorobenzene.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this BrCl-based methodology offers tangible strategic benefits that extend beyond simple yield improvements. The primary value driver is the drastic reduction in raw material intensity, specifically regarding bromine consumption. By theoretically doubling the utilization efficiency of bromine, manufacturers can significantly decouple production volumes from the volatile pricing of elemental bromine, leading to a more stable and predictable cost structure. This efficiency gain is compounded by the use of commodity-grade solvents and catalysts, which eliminates the dependency on niche, high-cost reagents like potassium bromate or pentafluorophenol. Consequently, the overall cost of goods sold (COGS) for bromopentafluorobenzene can be substantially lowered, providing a competitive edge in bidding for long-term supply contracts with major pharmaceutical and agrochemical companies. The robustness of the process also reduces the risk of batch failures, ensuring consistent availability of this critical intermediate.
- Cost Reduction in Manufacturing: The economic argument for this process is anchored in the superior atom economy of the bromine chloride reagent. In traditional bromination, half of the expensive bromine payload is discarded as waste acid, representing a direct financial loss. By contrast, the BrCl route ensures that every mole of bromine purchased contributes to the final product mass, effectively halving the stoichiometric requirement of bromine relative to the product output. This reduction in unit consumption translates directly into lower variable costs per kilogram of product. Additionally, the ability to use cheaper Lewis acid catalysts or highly efficient organoboron catalysts at low loadings further optimizes the reagent bill. The elimination of costly purification steps required to remove metal residues when using specific boron catalysts also reduces downstream processing expenses, contributing to a leaner and more profitable manufacturing operation.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on widely available bulk chemicals rather than specialized intermediates. Elemental bromine and chlorine gas are produced on a massive industrial scale globally, ensuring a secure and continuous supply even during market fluctuations. In contrast, alternative routes relying on pentafluorophenol or potassium bromate expose the supply chain to single-source risks and capacity constraints. Furthermore, the simplified reaction workflow reduces the complexity of logistics, as fewer distinct reagents need to be sourced, stored, and handled. The robustness of the reaction conditions, which tolerate slight variations in temperature and stoichiometry without catastrophic yield loss, adds another layer of reliability, minimizing the likelihood of production delays caused by sensitive process upsets.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this process aligns well with modern green chemistry principles and regulatory requirements. The reduction in bromine waste directly correlates to a lower environmental footprint, easing the burden on effluent treatment plants and reducing the costs associated with hazardous waste disposal. The patent explicitly demonstrates successful scale-up from gram-scale laboratory experiments to 500-liter reactor batches, proving the technology's readiness for commercial deployment. The use of standard halogenated solvents allows for established recovery and recycling protocols, further enhancing sustainability. Moreover, the option to utilize organoboron catalysts mitigates the generation of heavy metal wastewater, a significant concern for facilities operating under strict environmental discharge limits, thereby future-proofing the manufacturing site against tightening regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this bromination technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation. Understanding these nuances is vital for technical teams evaluating the feasibility of adopting this route for their specific production needs. The answers provided reflect the proven capabilities of the method as demonstrated in the cited examples.
Q: Why is the BrCl method superior to traditional Br2 bromination for pentafluorobenzene?
A: Traditional bromination with Br2 suffers from low atom economy, utilizing only 50% of the bromine molecule while generating HBr waste. The BrCl method converts both bromine atoms into the active +1 valence state, theoretically achieving 100% utilization and significantly reducing raw material costs.
Q: What are the critical impurities in this synthesis and how are they controlled?
A: The primary impurity is pentafluorochlorobenzene, formed via competitive chlorination. Using optimized Lewis acid catalysts like tris(pentafluorophenyl)boron minimizes this side reaction, achieving purity levels exceeding 99.7% with impurity content below 0.04%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates scalability from laboratory (100mL) to pilot/industrial scales (500L reactors). The use of common halogenated solvents and the elimination of expensive reagents like potassium bromate make it highly viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bromopentafluorobenzene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies promised by patent CN109438168B are fully realized in practice. Our state-of-the-art facilities are equipped to handle the specific safety and corrosion challenges associated with halogen chemistry, while our rigorous QC labs enforce stringent purity specifications to meet the exacting standards of the global pharmaceutical and electronics industries. We are committed to delivering high-purity bromopentafluorobenzene that empowers your downstream synthesis with reliability and consistency.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain. Our technical team is prepared to conduct a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this BrCl route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable, cost-effective supply of this critical fluorinated intermediate.
