Advanced Synthesis of Potassium Quinoline Trifluoroborates for Commercial Pharmaceutical Manufacturing
Advanced Synthesis of Potassium Quinoline Trifluoroborates for Commercial Pharmaceutical Manufacturing
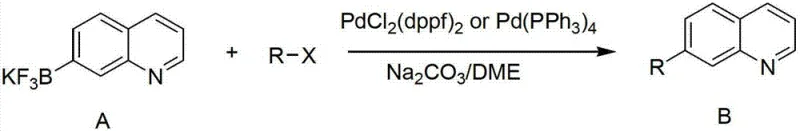
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more stable and versatile building blocks for cross-coupling reactions. A pivotal advancement in this domain is detailed in patent CN102786542A, which discloses a highly efficient preparation method for potassium quinoline trifluoroborate and potassium isoquinoline trifluoroborate derivatives. These compounds serve as critical precursors in Suzuki-Miyaura coupling reactions, enabling the construction of complex biaryl scaffolds found in numerous active pharmaceutical ingredients (APIs). Unlike traditional boronic acids or pinacol esters which can suffer from stability issues such as protodeboronation or difficult purification, the trifluoroborate salts described in this patent exhibit exceptional thermal and hydrolytic stability. This technical breakthrough addresses a significant gap in the market for reliable heterocyclic boron reagents, offering a pathway to higher purity intermediates essential for modern drug discovery and development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinoline-based boron reagents has relied heavily on boronic acids or their pinacol esters. While widely used, these conventional forms present distinct challenges in an industrial setting. Boronic acids are prone to self-condensation to form boroxines and are susceptible to protodeboronation, especially under acidic or basic conditions often encountered during downstream processing. Furthermore, pinacol esters, while more stable than free acids, can be expensive to produce due to the cost of pinacol and may require chromatographic purification which is not feasible on a multi-ton scale. The instability of these traditional reagents often leads to inconsistent yields in subsequent coupling reactions, creating bottlenecks in the supply chain for key drug intermediates. Additionally, the purification of boronic acids often involves complex recrystallization processes that result in significant material loss, thereby inflating the overall cost of goods sold (COGS) for the final API.
The Novel Approach
The methodology outlined in patent CN102786542A introduces a robust two-step sequence that circumvents these traditional pitfalls. By converting the intermediate boronic acid directly into a potassium trifluoroborate salt, the process leverages the unique stability profile of the BF3K moiety. This novel approach utilizes readily available and inexpensive reagents such as potassium bifluoride (KHF2) and triisopropyl borate, significantly reducing raw material costs. The process is designed for operational simplicity, employing standard unit operations like extraction, slurry, and filtration rather than complex chromatography. This shift not only enhances the chemical stability of the product, ensuring a longer shelf life and reduced degradation during transport, but also streamlines the manufacturing workflow. The ability to produce high-purity quinoline trifluoroborates through such a direct route represents a substantial technological leap, facilitating the reliable supply of high-quality intermediates for global pharmaceutical manufacturers.
Mechanistic Insights into Organolithium Borylation and Fluorination
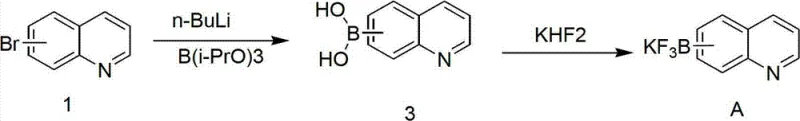
The core of this synthesis lies in a carefully controlled organolithium transformation followed by a salt metathesis. The process initiates with the halogen-lithium exchange or directed ortho-lithiation of bromo(iso)quinoline using n-butyllithium. This step is critically sensitive to temperature; the patent specifies cooling the reaction mixture to -60°C using a dry ice/acetone bath and maintaining the temperature below -50°C during the addition of the base. This rigorous thermal control is paramount to prevent side reactions such as nucleophilic attack on the quinoline ring or decomposition of the highly reactive aryl lithium species. Once the lithiated intermediate is formed, it is quenched with a borate ester, preferably triisopropyl borate, which acts as the electrophile to install the boron functionality. The resulting boronate ester complex is then hydrolyzed under acidic conditions to yield the free boronic acid.
Following the isolation of the boronic acid, the second mechanistic phase involves the formation of the trifluoroborate salt. The boronic acid is treated with potassium bifluoride (KHF2) in a mixed solvent system of methanol and water at ambient temperature. This transformation proceeds through the coordination of fluoride ions to the boron center, displacing hydroxyl groups and forming the stable tetrahedral trifluoroborate anion. The potassium cation serves to precipitate the salt, driving the equilibrium towards the product. This step is remarkably efficient, with the patent reporting yields ranging from 90% to 98% for the salt formation. The mechanism ensures that the boron atom is fully saturated and protected by three fluorine atoms, rendering the molecule inert to many conditions that would degrade a standard boronic acid. This mechanistic robustness is what makes the final product so valuable for downstream applications, particularly in palladium-catalyzed cross-couplings where reagent stability is crucial for high turnover numbers.
How to Synthesize Potassium Quinoline Trifluoroborate Efficiently
Implementing this synthesis requires precise adherence to the reaction parameters defined in the patent to ensure optimal yield and purity. The process is divided into two distinct stages: the generation of the boronic acid precursor and its subsequent conversion to the trifluoroborate salt. Operators must pay close attention to the stoichiometry of the reagents, specifically using 1.2 to 2.0 equivalents of borate ester and 1.1 to 1.5 equivalents of n-butyllithium relative to the bromo-substrate. The workup procedure involving slurry with petroleum ether and methyl tert-butyl ether is a key purification step that removes inorganic impurities without the need for column chromatography. For a comprehensive understanding of the specific operational details, including exact mixing times and filtration protocols, please refer to the standardized guide below.
- Dissolve bromo(iso)quinoline in anhydrous THF, cool to -60°C, and treat with n-butyllithium followed by triisopropyl borate to form the boronic acid intermediate.
- Quench the reaction with hydrochloric acid, extract the organic layer, and purify the crude boronic acid via slurry with petroleum ether and MTBE.
- React the purified boronic acid with potassium bifluoride (KHF2) in methanol/water at room temperature, then isolate the final trifluoroborate salt via filtration and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers compelling strategic advantages that extend beyond mere chemical efficiency. The primary benefit lies in the drastic simplification of the supply chain for critical coupling partners. By utilizing a reagent that is inherently stable and easy to handle, companies can reduce the risks associated with material degradation during storage and logistics. This stability translates directly into reduced waste and lower inventory write-offs, contributing to a leaner and more cost-effective operation. Furthermore, the reliance on commodity chemicals like potassium bifluoride and common solvents ensures that the production of these intermediates is not bottlenecked by the availability of exotic or expensive catalysts.
- Cost Reduction in Manufacturing: The economic impact of this process is significant due to the elimination of costly purification steps. Traditional methods often require silica gel chromatography to achieve pharmaceutical-grade purity, a process that is both solvent-intensive and laborious. In contrast, this novel method achieves high purity through simple crystallization and slurry techniques, which drastically reduces solvent consumption and waste disposal costs. Additionally, the high yields reported in the patent, often exceeding 90% for the final salt formation, mean that less starting material is required to produce the same amount of product, effectively lowering the raw material cost per kilogram. The use of triisopropyl borate, which is generally more cost-effective and easier to handle than some specialized boron reagents, further contributes to the overall cost optimization of the manufacturing process.
- Enhanced Supply Chain Reliability: Supply continuity is a top priority for any pharmaceutical manufacturer, and the robustness of this synthesis method plays a vital role in ensuring consistent delivery. The reagents involved are widely available in the global chemical market, reducing the risk of supply disruptions caused by single-source dependencies. Moreover, the stability of the potassium trifluoroborate product allows for larger batch sizes to be produced and stored for longer periods without quality degradation. This capability enables manufacturers to build strategic stockpiles, buffering against potential market fluctuations or unexpected surges in demand. The simplified workflow also means that production cycles are shorter and more predictable, allowing for better planning and faster response times to customer orders.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process aligns well with green chemistry principles. The avoidance of chromatographic purification significantly reduces the volume of organic waste generated, simplifying wastewater treatment and lowering the environmental footprint of the facility. The reaction conditions, while requiring low temperatures for the lithiation step, utilize standard industrial cooling systems and do not require extreme pressures or hazardous high-energy inputs. This makes the process highly scalable from pilot plant to commercial production volumes without significant engineering hurdles. The ability to scale up efficiently ensures that the technology can meet the growing demand for complex heterocyclic intermediates in the pharmaceutical and agrochemical sectors, supporting sustainable growth and compliance with increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of potassium quinoline trifluoroborates. These insights are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these nuances is essential for integrating this technology into existing manufacturing frameworks and maximizing its potential benefits.
Q: Why are potassium trifluoroborate salts preferred over pinacol boronic esters for quinoline derivatives?
A: Potassium trifluoroborate salts offer superior stability against protodeboronation and oxidation compared to pinacol esters, making them ideal for long-term storage and harsh reaction conditions in pharmaceutical manufacturing.
Q: What are the critical temperature controls required for the lithiation step?
A: The lithiation step requires strict temperature control below -50°C, ideally around -60°C, to ensure regioselectivity and prevent decomposition of the organolithium intermediate before borylation.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the method utilizes cheap raw materials like potassium bifluoride and standard solvents, with simple purification steps like slurry and filtration, making it highly suitable for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Potassium Quinoline Trifluoroborate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of specific isomers or large-scale supply of standard catalog items, our infrastructure is designed to support your needs from early-stage research through to commercial launch.
We invite you to collaborate with us to explore how this advanced synthesis technology can optimize your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can drive efficiency and reliability in your manufacturing operations. Let us be your partner in transforming innovative chemistry into commercial reality.
