Advanced Synthesis of R-(+)-2-(4-hydroxyphenoxy)propionic Acid for Herbicide Manufacturing
Introduction to Patent CN110105201B Technology
The pharmaceutical and agrochemical industries constantly seek robust synthetic routes that balance high purity with economic feasibility. Patent CN110105201B introduces a groundbreaking preparation method for R-(+)-2-(4-hydroxyphenoxy)propionic acid, a critical chiral intermediate used in the synthesis of prominent aryloxy propionic acid herbicides such as fluazifop-butyl and clodinafop-propargyl. This technology addresses long-standing challenges in the field, specifically the issues of over-alkylation and phenolic oxidation that have plagued conventional hydroquinone-based routes. By shifting the substrate strategy to p-halophenols and utilizing aniline derivatives as a recyclable solvent-catalyst system, this method achieves conversion rates exceeding 99% and product purities greater than 99%. For global procurement teams and R&D directors, this represents a significant opportunity to secure a reliable agrochemical intermediate supplier capable of delivering high-specification materials with reduced environmental impact and optimized production costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
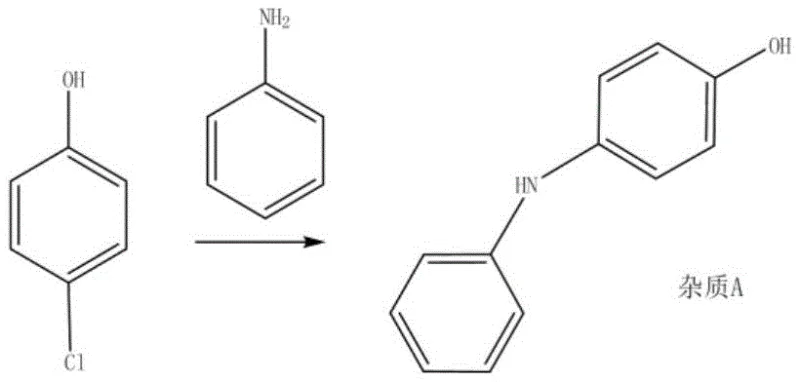
Historically, the synthesis of R-(+)-2-(4-hydroxyphenoxy)propionic acid has relied heavily on the reaction of hydroquinone with S-2-halopropanoic acid, a method documented in earlier patents like EP352168. This traditional approach suffers from two critical structural defects that complicate downstream processing and inflate manufacturing costs. Firstly, hydroquinone possesses two reactive hydroxyl groups, leading to unavoidable over-alkylation where both positions react, generating difficult-to-remove poly-alkylated impurities. Secondly, the phenolic structure is highly susceptible to oxidation under reaction conditions, necessitating the addition of reducing agents like sodium bisulfite and complex purification schemes to maintain product quality. Alternative biological methods, such as those described in CN10619305 involving yeast fermentation, introduce their own set of logistical burdens, including difficult culture maintenance, excessive wastewater generation, and prolonged reaction times that hinder scalable commercial production.
The Novel Approach
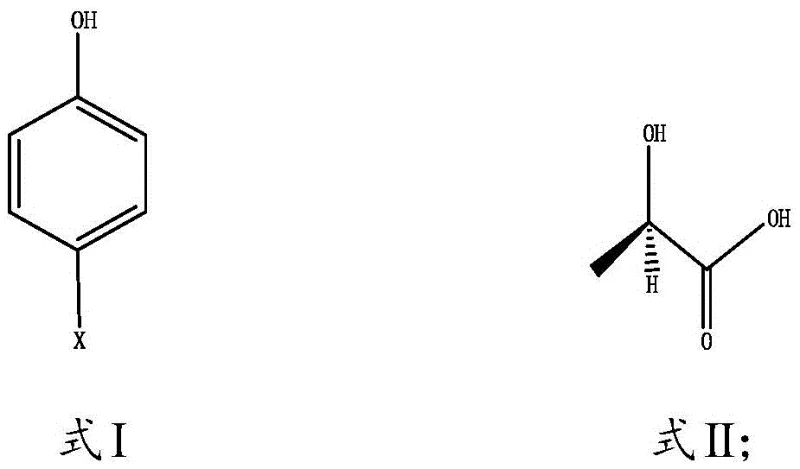
The innovative methodology disclosed in CN110105201B fundamentally restructures the synthetic pathway to bypass these inherent chemical limitations. Instead of using hydroquinone, the process employs p-halophenols (where X is F, Cl, Br, or I), which contain only a single reactive hydroxyl group, thereby sterically and chemically preventing the formation of over-alkylated byproducts. Furthermore, the reaction is conducted under an inert gas environment, typically nitrogen, which effectively suppresses the oxidation of the phenolic moiety without the need for stoichiometric reducing agents. The use of aniline or its derivatives serves a dual purpose, acting simultaneously as the reaction medium and a basic catalyst to absorb the hydrogen chloride byproduct. This streamlined approach not only simplifies the reaction profile but also enables a closed-loop solvent recovery system, marking a substantial advancement in cost reduction in agrochemical intermediate manufacturing.
Mechanistic Insights into Aniline-Mediated Alkylation
The core of this technological breakthrough lies in the unique mechanistic role played by the aniline solvent system. In this nucleophilic substitution reaction, the aniline molecules do not merely dissolve the reactants; they actively participate in the catalytic cycle by neutralizing the acid generated during the etherification process. As the S-2-hydroxypropionic acid reacts with the p-halophenol, hydrogen chloride is released, which is immediately scavenged by the basic aniline to form an aniline hydrochloride salt. This in-situ acid trapping drives the equilibrium forward, ensuring high conversion rates of the chiral starting material without the need for external inorganic bases that could induce racemization. The reaction proceeds smoothly at mild temperatures ranging from 30°C to 100°C, with optimal results observed between 50°C and 70°C, preserving the optical integrity of the S-configured starting material to yield the desired R-(+) product with high enantiomeric excess.
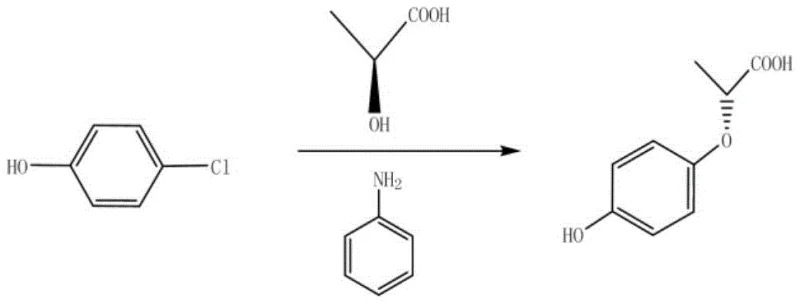
Controlling the impurity profile is paramount for any reliable agrochemical intermediate supplier, and this patent offers a sophisticated mechanism for managing side reactions. While the primary pathway yields the target ether, minor side reactions can generate Impurity A, as illustrated in the patent documentation. The brilliance of the workup procedure lies in its pH-dependent separation strategy. Upon completion of the alkylation, the reaction mixture is treated with an alkaline solution to adjust the pH to ≥8. Under these basic conditions, Impurity A, which likely possesses different acidity or solubility characteristics compared to the target product, partitions preferentially into the aqueous layer. This allows for a clean physical separation before the final acidification step, ensuring that the subsequent crystallization yields a product with purity specifications often exceeding 99.5%, thereby minimizing the burden on downstream purification units.
How to Synthesize R-(+)-2-(4-hydroxyphenoxy)propionic Acid Efficiently
The operational protocol for this synthesis is designed for industrial scalability, focusing on simplicity and material efficiency. The process begins with the precise mixing of p-chlorophenol, aniline, and S-2-hydroxypropionic acid in a reactor purged with nitrogen. The mixture is heated to maintain a temperature between 50°C and 70°C for a duration of 4 to 6 hours, monitored by HPLC to ensure the conversion of the chiral acid exceeds 99.5%. Following the reaction, the mixture undergoes a critical phase separation where alkaline washing removes acidic impurities and the aniline salt is regenerated. The detailed standardized operating procedures, including specific flow rates, column temperatures for QC analysis, and exact recrystallization parameters, are essential for maintaining batch-to-batch consistency in a commercial setting.
- Mix p-halophenol, aniline solvent, and S-2-hydroxypropionic acid under inert gas and heat to 50-70°C.
- Adjust pH to ≥8 with alkaline solution post-reaction to separate layers and remove impurities.
- Acidify the aqueous layer to pH <1, crystallize, and recover the aniline solvent for reuse.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates directly into enhanced operational efficiency and risk mitigation. The most significant economic driver is the recyclability of the aniline solvent. Unlike traditional processes where solvents are consumed or require energy-intensive distillation to remove complex byproducts, this system allows the aniline to be separated, washed, and returned to the reactor with purity levels sufficient for immediate reuse. This closed-loop capability drastically reduces the consumption of raw materials and minimizes the volume of hazardous waste requiring disposal. Consequently, this leads to substantial cost savings in agrochemical intermediate manufacturing by lowering both the bill of materials and the environmental compliance costs associated with waste treatment.
- Cost Reduction in Manufacturing: The elimination of expensive reducing agents and the minimization of over-alkylation byproducts significantly streamline the production cost structure. By avoiding the formation of difficult-to-separate poly-alkylated impurities, the process reduces the need for complex chromatographic purification or multiple recrystallization steps, which are often the most costly phases of fine chemical production. Furthermore, the high conversion rate ensures that the valuable chiral starting material, S-2-hydroxypropionic acid, is utilized with maximum efficiency, preventing financial loss through unreacted feedstock.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as p-chlorophenol and aniline ensures a stable and resilient supply chain, free from the bottlenecks associated with specialized biocatalysts or rare reagents. The robustness of the reaction conditions, which tolerate mild temperatures and standard inert gas protections, means that production can be scaled across multiple facilities without requiring exotic equipment. This flexibility allows for rapid capacity expansion to meet market demand surges for herbicide intermediates, ensuring consistent delivery schedules for downstream formulators.
- Scalability and Environmental Compliance: The process is inherently greener due to the solvent recycling mechanism and the absence of heavy metal catalysts or stoichiometric oxidants. The ability to recover and reuse aniline not only lowers the carbon footprint of the manufacturing process but also simplifies regulatory compliance regarding volatile organic compound (VOC) emissions. The simplified workup procedure generates less aqueous waste compared to fermentation-based alternatives, making it easier to manage effluent treatment and adhere to increasingly stringent environmental regulations in major chemical manufacturing hubs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering clarity on yield optimization, impurity management, and solvent recovery protocols. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this technology into existing production lines or for procurement specialists assessing the long-term viability of the supply source.
Q: How does this method prevent over-alkylation compared to hydroquinone routes?
A: By utilizing p-halophenol instead of hydroquinone, the process inherently possesses only one reactive phenolic hydroxyl group, effectively eliminating the risk of double alkylation and simplifying purification.
Q: What is the role of aniline in this synthesis?
A: Aniline serves a dual function as both the reaction solvent and a catalyst. It absorbs generated hydrogen chloride and can be recovered with high purity (>98%) for repeated cycles, drastically reducing material costs.
Q: How is Impurity A managed in this process?
A: Impurity A, formed via side reactions, is effectively removed during the alkaline workup phase where it partitions into the aqueous layer, ensuring the final crystallized product maintains purity above 99%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable R-(+)-2-(4-hydroxyphenoxy)propionic Acid Supplier
As the global demand for high-efficiency herbicides continues to grow, securing a partner with deep technical expertise in chiral intermediate synthesis is essential. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced methodologies like the one described in CN110105201B to deliver superior quality materials. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We operate with stringent purity specifications and utilize rigorous QC labs equipped with state-of-the-art HPLC and chiral analysis capabilities to guarantee that every batch meets the exacting standards required for agrochemical registration.
We invite you to collaborate with us to optimize your supply chain for R-(+)-2-(4-hydroxyphenoxy)propionic acid. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating how our optimized synthetic routes can reduce your overall landed costs. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples for your validation process, and let us demonstrate why we are the preferred partner for complex fine chemical intermediates.
