Advanced Solid-Liquid Phase Synthesis of AMG416 for Commercial Scale-Up and Purity Optimization
Advanced Solid-Liquid Phase Synthesis of AMG416 for Commercial Scale-Up and Purity Optimization
The pharmaceutical industry continuously seeks robust manufacturing pathways for complex polypeptides like AMG416 (Velcalcetide), particularly for treating secondary hyperparathyroidism in dialysis patients. Patent CN108218957B introduces a transformative solid-liquid phase combination method that addresses critical bottlenecks in traditional peptide synthesis. Unlike conventional approaches that rely solely on solid-phase oxidation or cumbersome liquid-phase fragment condensation, this innovation integrates resin cleavage, side-chain deprotection, and disulfide bond formation into a unified reaction system. This strategic integration not only drastically shortens reaction timelines from over 24 hours to approximately 2 hours but also significantly enhances the purity profile of the target peptide by minimizing byproduct generation associated with prolonged exposure to oxidative conditions. For R&D directors and procurement specialists, this represents a pivotal shift towards more efficient, scalable, and environmentally compliant manufacturing protocols for high-value therapeutic intermediates.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of AMG416 has been plagued by inefficiencies inherent in purely solid-phase or purely liquid-phase strategies. Traditional solid-phase synthesis often employs Rink amide resin followed by air oxidation to form the critical disulfide bond. This process is notoriously slow, frequently requiring reaction times exceeding 24 hours to achieve completion, which inevitably leads to the accumulation of oxidation byproducts and sequence mismatches. Furthermore, the post-treatment involves complex purification steps to separate these impurities, driving up costs and reducing overall yield. On the other hand, conventional liquid-phase methods, while effective for certain sequences, suffer from excessive solvent consumption during the oxidation step. The large volumes of organic solvents required not only inflate production costs but also pose significant environmental disposal challenges. Additionally, liquid-phase disulfide formation is prone to intermolecular mismatching, necessitating multiple purification cycles that further erode process efficiency and final product recovery rates.
The Novel Approach
The patented solid-liquid phase combination method offers a sophisticated solution by leveraging the strengths of both synthesis modalities while mitigating their respective weaknesses. By utilizing acid-sensitive amino resins, such as Sieber resin, the process allows for the controlled release of the peptide backbone under mild acidic conditions. Crucially, the addition of a dilute acid lysate containing a specialized cysteine derivative, Y-Cys(X)-OH (specifically Boc-Cys(Npys)-OH), enables the simultaneous occurrence of three distinct chemical events: resin cleavage, removal of acid-labile protecting groups, and the formation of the disulfide bond. This concurrency eliminates the need for isolated oxidation steps and extensive intermediate purifications. The result is a streamlined workflow that reduces solvent usage, minimizes the risk of disulfide scrambling, and delivers a fully protected target peptide with superior purity characteristics, ready for final global deprotection and polishing.
Mechanistic Insights into Solid-Liquid Hybrid Disulfide Formation
The core mechanistic advantage of this technology lies in the precise orchestration of protecting group chemistry and reaction kinetics. The process begins with the solid-phase assembly of the linear peptide backbone, Ac-D-Cys1(Mmt)-D-Ala-D-Arg(pbf)-D-Ala-D-Arg(pbf)-Sieber resin, using standard Fmoc chemistry. The Mmt (4-methoxytrityl) group on the N-terminal cysteine is selected for its high sensitivity to weak acids. In the pivotal second step, a dilute acid lysate composed of TFA/TIS/DCM (typically in a 10/10/80 volume ratio) is introduced. This specific cocktail is strong enough to cleave the peptide from the Sieber resin and remove the Mmt group, exposing the free sulfhydryl (-SH) of the D-Cys1 residue, yet mild enough to preserve other acid-stable protecting groups initially. Simultaneously, the added Boc-Cys(Npys)-OH reacts with this newly exposed thiol. The Npys (3-nitro-2-pyridylthio) group serves as an activating moiety that is highly reactive towards free thiols but stable to the weak acid conditions, ensuring rapid and selective disulfide bond formation without the need for external oxidants like air or iodine.
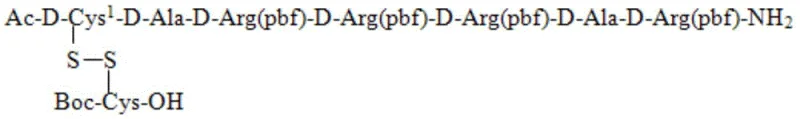
This mechanistic design effectively prevents the formation of intermolecular disulfide dimers or oligomers, a common impurity in liquid-phase oxidations. By forming the disulfide bond immediately upon the release of the thiol from the resin, the intramolecular or specific intermolecular pairing is kinetically favored over random oxidation. The use of Boc-Cys(Npys)-OH ensures that the second cysteine is introduced in a pre-activated state, facilitating a nucleophilic attack by the resin-bound cysteine thiol. Following this hybrid step, the fully protected peptide is isolated via precipitation with MTBE, removing resin debris and excess reagents in a single filtration. The final cleavage step uses a standard TFA cocktail to remove all remaining side-chain protecting groups (such as Pbf on arginine), yielding the crude AMG416 peptide. This sequential yet integrated approach ensures that the most chemically sensitive step—disulfide formation—occurs in a controlled environment, drastically reducing the impurity load before the final purification stage.
How to Synthesize AMG416 Efficiently
The implementation of this solid-liquid phase combination strategy requires precise control over reagent stoichiometry and reaction conditions to maximize yield and purity. The process initiates with the loading of the first amino acid onto Sieber resin, followed by iterative coupling cycles using activators like HBTU or DIC/HoBt to build the linear sequence. Critical attention must be paid to the coupling of the first D-Cys residue to prevent racemization, often utilizing DIC/Cl-HoBt at low temperatures. Once the backbone is assembled and N-terminally acetylated, the resin is subjected to the hybrid reaction conditions described previously. The detailed operational parameters, including specific solvent ratios and reaction times, are optimized to ensure complete conversion while maintaining the integrity of the peptide sequence. For laboratory and pilot-scale operations, adhering to the standardized protocol ensures reproducibility and facilitates the transition to larger manufacturing batches.
- Synthesize the nitrogen-terminal acetylated fully-protected main chain peptide resin using acid-sensitive Sieber resin and Fmoc protection strategy.
- Treat the resin with a dilute acid lysate containing Boc-Cys(Npys)-OH to simultaneously cleave the resin, remove side-chain protecting groups, and form the critical disulfide bond.
- Perform final global deprotection cleavage using TFA-based cocktails followed by HPLC purification to obtain high-purity AMG416.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis method translates into tangible operational efficiencies and cost optimizations. The primary economic driver is the significant reduction in processing time; compressing the disulfide bond formation from over 24 hours to just 2 hours dramatically increases reactor throughput and asset utilization. This acceleration allows manufacturers to respond more agilely to market demand fluctuations without the need for excessive capital investment in additional reactor capacity. Furthermore, the elimination of multiple purification steps between the linear peptide synthesis and the final product reduces the consumption of expensive chromatography media and solvents. The simplified workflow also lowers labor costs and minimizes the potential for human error during complex multi-step transfers, thereby enhancing overall process reliability and batch-to-batch consistency.
- Cost Reduction in Manufacturing: The integrated nature of the solid-liquid phase method eliminates the need for separate oxidation reactors and the extensive solvent exchanges typically required in pure liquid-phase synthesis. By performing resin cleavage and disulfide formation in a single pot, the process significantly reduces the total volume of organic solvents consumed, directly lowering raw material costs and waste disposal fees. Additionally, the high selectivity of the Npys-mediated disulfide formation reduces the formation of hard-to-remove impurities, which in turn improves the yield of the final purification step. This cumulative effect results in a lower cost of goods sold (COGS) per gram of active pharmaceutical ingredient, providing a competitive pricing advantage in the global market for polypeptide therapeutics.
- Enhanced Supply Chain Reliability: The robustness of this synthesis route contributes to a more stable and predictable supply chain. Traditional methods prone to long reaction times and variable oxidation rates often suffer from batch failures or inconsistent quality, leading to supply disruptions. In contrast, the controlled kinetics of the hybrid method ensure high reproducibility, reducing the risk of production delays. The use of commercially available, stable reagents like Boc-Cys(Npys)-OH and standard acid-sensitive resins ensures that raw material sourcing is straightforward and less susceptible to geopolitical or logistical bottlenecks. This reliability is crucial for maintaining continuous production schedules and meeting the stringent delivery commitments required by pharmaceutical partners.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental sustainability intensify, the reduced solvent footprint of this method offers a distinct compliance advantage. The minimization of solvent waste aligns with green chemistry principles, simplifying the permitting process for manufacturing facilities and reducing the burden on wastewater treatment systems. Moreover, the process is inherently scalable; the transition from gram-scale laboratory synthesis to kilogram or ton-scale commercial production does not require fundamental changes to the reaction chemistry. The simplicity of the workup procedure, involving straightforward filtration and precipitation, is easily adapted to large-scale industrial equipment, facilitating rapid commercial scale-up of complex polypeptide intermediates without the engineering challenges associated with heterogeneous solid-phase oxidation on a massive scale.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of AMG416 using this advanced solid-liquid phase methodology. These insights are derived directly from the experimental data and technical specifications outlined in the patent literature, providing clarity on process capabilities and quality standards. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this technology into their existing manufacturing portfolios or sourcing strategies.
Q: How does the solid-liquid phase combination method improve AMG416 purity compared to traditional solid-phase synthesis?
A: Traditional solid-phase oxidation often requires over 24 hours, leading to byproduct accumulation. The hybrid method completes disulfide bond formation in approximately 2 hours using a specific dilute acid lysate, significantly reducing side reactions and simplifying downstream purification.
Q: What specific reagents are used to prevent disulfide mismatching during the synthesis?
A: The process utilizes Boc-Cys(Npys)-OH, where the Npys group acts as a weak acid-insensitive protecting group that is highly sensitive to free sulfhydryls. This ensures selective reaction with the exposed cysteine thiol on the main chain, preventing intermolecular mismatching.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the method is designed for scalability. By integrating resin cleavage and disulfide formation into a single step, it reduces solvent consumption and operational complexity, making it more environmentally friendly and cost-effective for manufacturing compared to multi-step liquid phase oxidations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable AMG416 Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthesis technologies to meet the evolving demands of the pharmaceutical sector. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering high-purity AMG416 and related polypeptide intermediates that adhere to the most stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our facility is designed to handle complex peptide chemistries with the utmost precision, guaranteeing product consistency and regulatory compliance for our global clientele.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced solid-liquid phase synthesis technology for your projects. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and comprehensive quotations. Let us help you optimize your supply chain and accelerate the development of life-saving therapies through our expertise in fine chemical manufacturing and peptide synthesis.
