Advanced Solid-Liquid Combined Synthesis Strategy For Commercial Scale-Up Of Complex Peptide APIs
Advanced Solid-Liquid Combined Synthesis Strategy For Commercial Scale-Up Of Complex Peptide APIs
The pharmaceutical industry continuously seeks robust manufacturing pathways for complex peptide therapeutics, and the preparation method detailed in patent CN112521482A represents a significant technological leap for producing Nesiritide. This specific intellectual property outlines a sophisticated solid-liquid combination strategy that addresses the longstanding challenges associated with the total solid-phase synthesis of long-chain peptides containing difficult sequences. By strategically dividing the peptide chain into two manageable fragments and utilizing a novel disulfide bond exchange mechanism, this methodology offers a compelling solution for enhancing both preparation efficiency and final product purity. For global procurement teams and R&D directors, understanding the nuances of this patented approach is critical for evaluating potential partners capable of delivering high-purity nesiritide. The technical breakthroughs described herein not only mitigate the risks of racemization but also streamline the production timeline, making it a viable candidate for cost reduction in pharmaceutical intermediates manufacturing. As a reliable peptide API supplier, analyzing such proprietary methods allows us to align our internal process development with the highest industry standards for cardiovascular therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional manufacturing routes for Nesiritide, such as those relying entirely on recombinant DNA technology or stepwise solid-phase peptide synthesis (SPPS), present substantial hurdles for large-scale commercial production. Genetic engineering methods, while effective for certain biologics, often struggle with the specific folding and post-translational modifications required for active natriuretic peptides, leading to complex downstream purification burdens. Furthermore, conventional SPPS approaches face severe difficulties when coupling the Ser19-Ser-Ser-Ser22 fragment, where the high propensity for aspartimide formation and racemization drastically reduces the yield of the final product. Previous patents, such as CN 104447979B, highlight issues with iodine oxidation processes causing the fall-off of fully protected peptides and the generation of hard-to-remove impurities. These technical bottlenecks result in increased material consumption, extended processing times, and inconsistent batch-to-batch quality, which are unacceptable for a reliable agrochemical intermediate supplier or pharma partner aiming for strict regulatory compliance. The accumulation of deletion sequences and epimerization byproducts necessitates extensive chromatographic purification, driving up the cost of goods sold and limiting supply chain reliability.
The Novel Approach
The innovative strategy disclosed in the reference patent overcomes these historical limitations by employing a hybrid solid-liquid phase synthesis that optimizes the assembly of the 32-amino acid sequence. Instead of attempting a linear synthesis of the entire chain, the method divides the target molecule into two distinct fragments: Intermediate 1 (residues 21-32) and Intermediate 4 (residues 1-20 with activated C-terminus). This fragmentation significantly reduces the difficulty of coupling the problematic serine-rich region by handling it within a shorter, more controllable liquid-phase segment. The activation of the C-terminal carboxyl group using salicylaldehyde glycol acetal derivatives ensures a highly reactive ester species that facilitates efficient amide bond formation without excessive racemization. Moreover, the use of a disulfide bond exchange reaction between a Cys(Npys) moiety and a free thiol allows for precise control over the critical Cys10-Cys26 bridge formation. This approach effectively shortens the production time by nearly half compared to traditional methods and drastically simplifies the impurity profile, offering substantial cost savings in electronic chemical manufacturing or similar high-precision sectors. The result is a streamlined process that enhances supply chain continuity by reducing the dependency on multiple purification cycles.
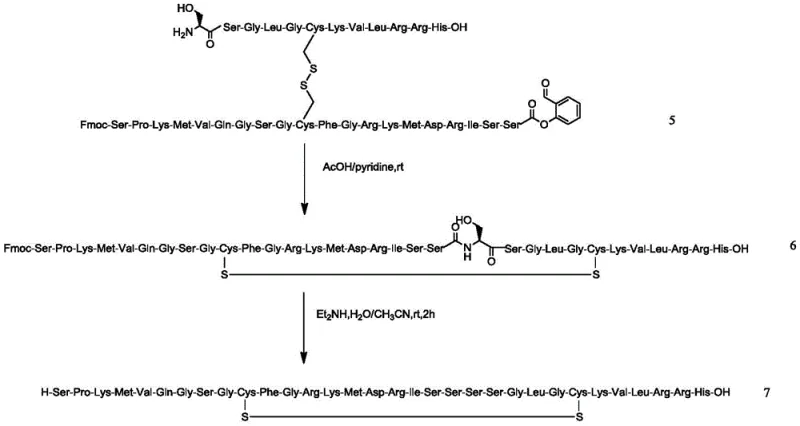
Mechanistic Insights into Solid-Liquid Fragment Condensation
The core chemical ingenuity of this process lies in the meticulous design of the fragment coupling and disulfide bridge formation mechanisms. Intermediate 1 is synthesized on a 2-CTC resin using Fmoc chemistry, with the critical Cysteine residue at position 26 protected by a 3-nitro-2-pyridinesulfenyl (Npys) group. This specific protecting group is pivotal because it renders the sulfur atom electrophilic, priming it for a nucleophilic attack by the free thiol of Cysteine at position 10 on Intermediate 4. When these two fragments are mixed in an aqueous solution, a spontaneous disulfide bond exchange occurs, forming the intermolecular disulfide linkage with high regioselectivity. This mechanism avoids the random oxidation pitfalls associated with iodine or air oxidation methods, which often lead to scrambled disulfide isomers and polymeric byproducts. The subsequent cyclization step utilizes a pyridine/acetic acid buffer system to promote the intramolecular amidation between the N-terminal amine and the activated C-terminal ester. This mild acidic environment preserves the stereochemical integrity of the sensitive amino acid residues while driving the macrocyclization to completion. Understanding these mechanistic details is essential for any technical procurement team evaluating the feasibility of commercial scale-up of complex polymer additives or peptide APIs.
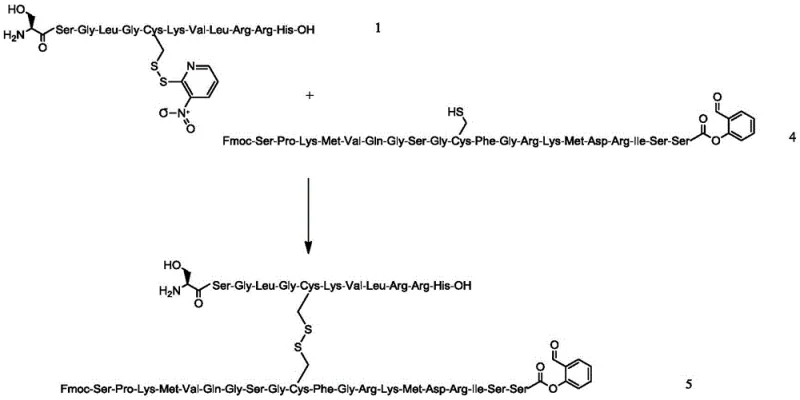
Impurity control is rigorously managed through the selection of orthogonal protecting groups and specific cleavage conditions throughout the synthesis. For instance, the side chain protecting groups on Intermediate 2 are removed using a mixture of TFA, TIS, and water in a 90:5:5 ratio, which effectively scavenges carbocations generated during deprotection without damaging the peptide backbone. The activation of the C-terminus using salicylaldehyde dimethyl acetal ensures that the reactive ester is formed in situ with minimal exposure to harsh conditions that could induce epimerization. Furthermore, the final purification protocol employs a three-step gradient HPLC method using C18 stationary phases with varying mobile phase compositions, including TFA, sodium dihydrogen phosphate, and acetic acid buffers. This multi-dimensional purification strategy is designed to separate the target nesiritide from closely related impurities such as deletion sequences, oxidized methionine variants, and diastereomers. By controlling the pH and organic modifier gradients precisely, the process achieves a final purity specification that exceeds 99.5%, meeting the stringent requirements for high-purity OLED material or pharmaceutical grade substances. This level of analytical control ensures that the final API is safe for clinical use and consistent across large manufacturing batches.
How to Synthesize Nesiritide Efficiently
Implementing this synthesis route requires precise adherence to the reaction conditions and reagent stoichiometries outlined in the patent examples to ensure reproducibility and high yield. The process begins with the solid-phase assembly of the two fragments using standard Fmoc protocols, followed by selective cleavage and activation steps that must be monitored closely via HPLC. The critical disulfide exchange and cyclization steps are performed in solution phase, requiring careful control of pH and concentration to favor the desired intramolecular reactions over intermolecular polymerization. Detailed standardized synthesis steps see the guide below for specific reagent grades and reaction times.
- Synthesize Intermediate 1 (C-terminal fragment) using Fmoc solid-phase synthesis on 2-CTC resin, ensuring Cys side chain protection with Npys group.
- Synthesize Intermediate 2 (N-terminal fragment) via solid-phase methods, then activate the C-terminal carboxyl group using salicylaldehyde acetal to form Intermediate 4.
- Perform disulfide bond exchange between Intermediate 1 and Intermediate 4 in aqueous solution, followed by cyclization with pyridine/acetic acid and final HPLC purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this solid-liquid combined synthesis method offers transformative benefits for procurement managers and supply chain heads focused on efficiency and risk mitigation. The primary advantage lies in the significant reduction of raw material waste and processing time, which directly translates to improved cost structures without compromising quality. By avoiding the low-yield steps associated with full-length solid-phase synthesis, manufacturers can achieve higher throughput per batch, thereby enhancing supply chain reliability and reducing the risk of stockouts for critical cardiovascular medications. The simplified purification profile also means less solvent consumption and lower environmental impact, aligning with modern sustainability goals and regulatory expectations for green chemistry practices. For partners seeking a reliable nesiritide supplier, this technology provides a robust foundation for long-term contracts and stable pricing models.
- Cost Reduction in Manufacturing: The elimination of multiple low-yield coupling cycles and the reduction in purification complexity lead to a drastic simplification of the overall production workflow. By utilizing fragment condensation, the consumption of expensive protected amino acids and coupling reagents is optimized, resulting in substantial cost savings compared to linear synthesis routes. The avoidance of transition metal catalysts for oxidation further removes the need for costly heavy metal removal steps, streamlining the downstream processing and reducing the burden on quality control laboratories. This economic efficiency allows for more competitive pricing strategies while maintaining healthy margins for continuous investment in process improvement.
- Enhanced Supply Chain Reliability: The robustness of the fragment-based approach ensures consistent batch-to-batch performance, which is critical for maintaining uninterrupted supply to global markets. The use of stable intermediates and well-defined reaction conditions minimizes the risk of batch failures due to unpredictable side reactions or impurity buildup. This predictability enables better production planning and inventory management, reducing lead time for high-purity peptide APIs and ensuring that customer demands are met promptly. Additionally, the scalability of the liquid-phase coupling steps facilitates easy transition from pilot scale to multi-ton commercial production without significant re-engineering of the process equipment.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing common reagents and standard reactor configurations that are readily available in GMP facilities. The reduction in solvent usage and the elimination of hazardous oxidants contribute to a lower environmental footprint, making it easier to comply with increasingly strict environmental regulations. The efficient waste profile simplifies effluent treatment processes, reducing operational costs associated with waste disposal. This alignment with environmental, social, and governance (ESG) criteria enhances the corporate reputation of manufacturers and appeals to socially conscious investors and partners in the fine chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Nesiritide using this advanced methodology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and quality assurances associated with this synthesis route. Understanding these details helps stakeholders make informed decisions regarding supplier qualification and process validation.
Q: Why is the solid-liquid combined method superior to full solid-phase synthesis for Nesiritide?
A: The solid-liquid combined approach significantly reduces the formation of defective Ser impurities and racemization often seen in long sequential solid-phase couplings, particularly around the Ser19-Ser-Ser-Ser22 fragment, thereby improving overall yield and purity.
Q: How does the new method control disulfide bond formation accuracy?
A: By utilizing a specific disulfide bond exchange reaction between a Cys(Npys) protected fragment and a free Cys thiol fragment, the process ensures correct intermolecular pairing while minimizing polymerization and incorrect intramolecular bridging.
Q: What purification standards are achieved with this synthesis route?
A: The process employs a rigorous three-step preparative HPLC purification strategy using C18 columns with specific mobile phase gradients, consistently achieving final product purity exceeding 99.5% suitable for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nesiritide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthesis technologies to meet the evolving demands of the global pharmaceutical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex peptide routes like the one described in CN112521482A can be successfully translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of nesiritide meets the highest international standards. Our commitment to technical excellence allows us to offer customized solutions that address specific client needs while maintaining the utmost integrity and compliance with regulatory guidelines.
We invite you to collaborate with us to explore how this innovative synthesis method can benefit your supply chain and product portfolio. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability as your trusted partner in the production of high-value cardiovascular therapeutics. Let us work together to bring safer and more affordable medicines to patients worldwide through scientific innovation and operational excellence.
