Scalable Synthesis of Propafenone Intermediates via Non-Hydrogenation Reduction Technology
The pharmaceutical industry continuously seeks robust manufacturing pathways that balance safety, cost, and scalability, particularly for high-volume antiarrhythmic agents like propafenone hydrochloride. Patent CN111217690B introduces a transformative approach to synthesizing the critical intermediate, 2'-hydroxydihydrochalcone, by replacing traditional catalytic hydrogenation with a novel chemical reduction system. This technology utilizes 2-phenyl-benzothiazoline as a specialized reducing agent in the presence of aluminum oxide, effectively circumventing the logistical and safety hazards associated with high-pressure hydrogen gas and precious metal catalysts. For R&D directors and supply chain managers, this represents a pivotal shift towards safer, more controllable process chemistry that maintains high purity standards while simplifying operational requirements. The method ensures efficient selective reduction of the substrate's carbon-carbon double bond, preserving the integrity of other functional groups essential for the final API structure.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 2'-hydroxydihydrochalcone has relied heavily on catalytic hydrogenation using palladium on carbon (Pd/C) under hydrogen pressure. This conventional route presents significant bottlenecks for large-scale manufacturing, primarily due to the exorbitant cost of noble metal catalysts and the inherent dangers of handling explosive hydrogen gas in bulk quantities. Facilities must invest in specialized high-pressure reactors and rigorous safety protocols to mitigate explosion risks, which drastically inflates capital expenditure and operational overhead. Furthermore, the removal of trace palladium residues to meet stringent pharmaceutical purity specifications often requires additional purification steps, complicating the workflow and potentially reducing overall yield. These factors collectively hinder the ability to achieve rapid, cost-effective scale-up, making the traditional hydrogenation route less attractive for modern, agile supply chains seeking to minimize risk exposure.
The Novel Approach
In stark contrast, the methodology disclosed in the patent employs a transfer hydrogenation-like strategy using 2-phenyl-benzothiazoline as a benign, organic reducing agent. This system operates under atmospheric pressure in standard glass-lined or stainless steel reactors, removing the need for complex high-pressure infrastructure. The reaction proceeds efficiently in common aromatic solvents like toluene or xylene at moderate temperatures ranging from 80°C to 170°C, offering a wide operational window that enhances process robustness. By utilizing aluminum oxide as a reusable additive, the process not only promotes the reduction but also simplifies workup through simple filtration. This approach fundamentally decouples the synthesis from the volatility of hydrogen markets and the supply constraints of precious metals, offering a stable, predictable manufacturing baseline.
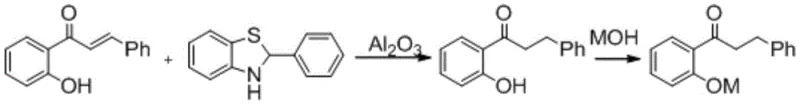
Mechanistic Insights into 2-Phenyl-Benzothiazoline Mediated Reduction
The core of this innovation lies in the unique reactivity of 2-phenyl-benzothiazoline, which serves as a hydrogen donor capable of selectively reducing the olefinic double bond of the chalcone derivative. Mechanistically, the aluminum oxide likely acts as a Lewis acid support or promoter, facilitating the activation of the reducing agent and ensuring precise chemoselectivity. This selectivity is paramount, as it prevents the unwanted reduction of the carbonyl group, thereby preserving the ketone functionality required for the subsequent etherification and amination steps in the propafenone synthesis pathway. The interaction between the substrate, the reducing agent, and the alumina surface creates a localized environment that favors the formation of the saturated dihydrochalcone skeleton with high fidelity. Understanding this mechanism allows process chemists to fine-tune parameters such as temperature and stoichiometry to maximize conversion while minimizing byproduct formation.
Furthermore, the impurity profile generated by this chemical reduction is distinctively cleaner compared to metal-catalyzed methods, which often suffer from over-reduction or metal-induced side reactions. The absence of transition metals eliminates the risk of metal-catalyzed degradation during storage or downstream processing, leading to a more stable intermediate salt. The patent data indicates that the alumina can be recovered, washed with methanol, and reused multiple times without significant loss of activity, suggesting a heterogeneous interaction that does not consume the additive. This recyclability not only reduces solid waste generation but also contributes to a greener process footprint, aligning with modern environmental compliance standards. The ability to isolate the product as a stable salt via base precipitation further streamlines purification, avoiding the need for chromatographic separation or extensive aqueous workups.
How to Synthesize 2'-Hydroxydihydrochalcone Efficiently
The synthesis protocol outlined in the patent provides a clear, reproducible pathway for generating high-purity intermediates suitable for GMP manufacturing. The process begins with the charging of 2'-hydroxychalcone, the reducing agent, and the alumina additive into a reactor containing an aromatic solvent, followed by heating to initiate the reduction. Upon completion, the mixture is cooled, and the solid alumina is removed via filtration, leaving the product in solution for direct salt formation.
- Combine 2'-hydroxychalcone, 2-phenyl-benzothiazoline, and aluminum oxide in a suitable aromatic solvent such as toluene.
- Heat the reaction mixture to between 80°C and 170°C for 0.5 to 12 hours to effect selective reduction of the carbon-carbon double bond.
- Filter off the alumina, treat the filtrate with a base like sodium hydroxide to precipitate the product salt, and isolate for downstream etherification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement strategists and supply chain leaders, the adoption of this non-hydrogenation technology offers profound advantages in terms of cost stability and operational continuity. By eliminating the dependency on palladium catalysts, manufacturers can insulate their production costs from the extreme price volatility characteristic of the precious metals market. Additionally, the removal of high-pressure hydrogenation steps significantly lowers the barrier to entry for contract manufacturing organizations (CMOs) that may lack specialized high-pressure facilities, thereby expanding the pool of qualified suppliers and enhancing supply chain resilience. The simplified equipment requirements translate to lower maintenance costs and reduced downtime, ensuring more reliable delivery schedules for critical API intermediates.
- Cost Reduction in Manufacturing: The substitution of expensive noble metal catalysts with readily available organic reducing agents and reusable alumina results in substantial raw material cost savings. The process avoids the capital-intensive infrastructure needed for hydrogen handling, leading to lower depreciation and utility costs per kilogram of product. Furthermore, the ability to recycle the alumina additive multiple times minimizes waste disposal fees and reduces the overall consumption of auxiliary materials. These cumulative efficiencies drive down the cost of goods sold (COGS), allowing for more competitive pricing strategies in the global pharmaceutical market without compromising on quality standards.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like 2-phenyl-benzothiazoline and alumina rather than specialized catalysts or compressed gases mitigates the risk of supply disruptions. The raw materials are widely sourced and easy to transport, removing the logistical complexities associated with hazardous gas delivery. This stability ensures consistent production throughput, enabling manufacturers to meet tight deadlines and maintain adequate inventory levels. The robustness of the reaction conditions also means that production is less susceptible to minor fluctuations in utility supplies or environmental conditions, guaranteeing a steady flow of intermediates to downstream formulation units.
- Scalability and Environmental Compliance: The ambient pressure nature of this reaction makes it inherently safer and easier to scale from pilot plant to multi-ton commercial production without the need for exponential increases in safety engineering controls. The reduction in hazardous waste, particularly the absence of spent metal catalysts requiring special treatment, simplifies environmental permitting and regulatory compliance. The process aligns with green chemistry principles by reducing energy consumption and waste generation, which is increasingly becoming a prerequisite for partnerships with major multinational pharmaceutical companies focused on sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel reduction technology in industrial settings. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a factual basis for decision-making.
Q: Why is the 2-phenyl-benzothiazoline reduction method superior to traditional hydrogenation?
A: This method eliminates the need for expensive palladium on carbon catalysts and high-pressure hydrogen gas, significantly reducing equipment costs and safety risks while maintaining high selectivity for the C=C bond.
Q: Can the aluminum oxide additive be recycled in this process?
A: Yes, the patent explicitly demonstrates that the filtered aluminum oxide can be washed with methanol, dried, and reused multiple times without significant loss of catalytic efficiency, enhancing process sustainability.
Q: Is the intermediate produced compatible with downstream propafenone synthesis?
A: Absolutely. The process yields a stable salt form of the intermediate that can be directly utilized in the subsequent etherification step with epichlorohydrin without requiring acid neutralization, streamlining the overall workflow.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Propafenone Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting advanced synthetic routes like the one described in CN111217690B to enhance the reliability of our supply chain. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are seamlessly translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of propafenone intermediate meets the highest global regulatory standards, providing our partners with absolute confidence in product quality and consistency.
We invite forward-thinking pharmaceutical companies to collaborate with us on optimizing their propafenone supply chains through this cost-effective and safe technology. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can drive efficiency and value for your organization.
