Advanced 3-Step Synthesis of 7-Nitroindole: A Breakthrough for Scalable Pharmaceutical Manufacturing
Advanced 3-Step Synthesis of 7-Nitroindole: A Breakthrough for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry constantly seeks robust, scalable pathways for critical heterocyclic intermediates, and the recent disclosure in patent CN110845388A represents a significant leap forward in the synthesis of 7-nitroindole. This vital building block serves as a precursor for a wide array of bioactive molecules, including influenza inhibitors, JAK1 selective inhibitors, and novel antitumor agents targeting tubulin. Historically, the production of 7-nitroindole has been plagued by hazardous conditions, expensive reagents, and poor scalability, creating bottlenecks for drug developers. The new methodology outlined in this patent introduces a streamlined three-step sequence starting from the readily available commodity chemical o-nitroaniline. By leveraging a unique sodium borohydride-zirconium chloride reduction system, this process achieves high chemoselectivity and yield under mild conditions, effectively solving the long-standing challenges of nitro-group preservation during reduction. For R&D directors and supply chain managers alike, this innovation offers a compelling alternative to legacy routes, promising enhanced safety profiles and substantial economic advantages for commercial manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of 7-nitroindole was dominated by methodologies that were either prohibitively dangerous or economically unviable for large-scale operations. One of the most cited historical approaches is the Bartoli indole synthesis, which typically relies on the reaction of vinyl Grignard reagents with nitroarenes. As illustrated in the reaction scheme below, this pathway necessitates the use of 1,2-dinitrobenzene, a raw material known for its high toxicity and explosive potential, making it extremely difficult to source in bulk quantities safely.

Furthermore, the Bartoli reaction demands cryogenic conditions, often requiring temperatures below -60 °C to control exothermicity and prevent decomposition. Such extreme thermal requirements impose massive energy costs and require specialized refrigeration infrastructure that many standard chemical plants lack. Additionally, alternative routes such as the Fischer indole synthesis involve lengthy five-step sequences with dangerous diazotization steps and high-temperature decarboxylation near 200 °C. Other methods involving palladium-catalyzed coupling of 7-chloroindole suffer from the high cost of noble metal catalysts and phosphine ligands, alongside the scarcity of the chlorinated starting material. These cumulative factors—safety risks, high energy consumption, and expensive inputs—have historically rendered 7-nitroindole a high-cost, low-availability intermediate.
The Novel Approach
In stark contrast to these legacy challenges, the method disclosed in patent CN110845388A utilizes a concise three-step strategy that prioritizes safety and cost-efficiency without compromising on yield. The core of this innovation lies in the strategic selection of o-nitroaniline as the starting material, a commodity chemical that is abundant, stable, and inexpensive compared to dinitrobenzenes or halogenated indoles. The reaction pathway proceeds through the formation of an oximino intermediate, followed by acid-catalyzed cyclization to 7-nitroisatin, and finally, a highly selective reduction to the target indole. This approach completely eliminates the need for cryogenic cooling, high-pressure hydrogenation, or toxic heavy metals.
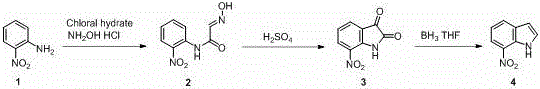
The operational simplicity of this new route is a game-changer for process chemistry teams. By avoiding the use of Grignard reagents and palladium catalysts, the process significantly reduces the complexity of waste treatment and raw material procurement. The reaction conditions are remarkably mild, with the initial condensation occurring at 40-45 °C and the final reduction taking place between 0-5 °C, temperatures that are easily manageable in standard glass-lined reactors. This shift from extreme conditions to ambient or near-ambient processing not only lowers the barrier to entry for manufacturing but also drastically reduces the risk of thermal runaways, making it an ideal candidate for reliable pharmaceutical intermediates supplier networks aiming for consistent quality and supply continuity.
Mechanistic Insights into Chemoselective Reduction and Cyclization
From a mechanistic perspective, the success of this synthesis hinges on the precise control of reactivity, particularly during the final reduction step. The transformation of 7-nitroisatin to 7-nitroindole presents a classic chemoselectivity challenge: reducing the carbonyl functionality of the isatin ring without affecting the sensitive nitro group on the aromatic ring. Traditional reducing agents like borane (BH3) or sodium borohydride activated by boron trifluoride (BF3) often lack this specificity, frequently leading to the over-reduction of the nitro group to an amine, which generates difficult-to-separate impurities and lowers the overall yield. The patent discloses a novel reduction system utilizing sodium borohydride (NaBH4) in combination with zirconium tetrachloride (ZrCl4). The interaction between NaBH4 and ZrCl4 likely generates a zirconium-hydride species in situ that is sufficiently potent to reduce the ketone and lactam carbonyls of the isatin scaffold but mild enough to leave the electron-deficient nitro group untouched.
This mechanistic nuance is critical for maintaining the integrity of the molecule and ensuring high purity specifications required for API synthesis. The preceding cyclization step, where the oximino intermediate converts to 7-nitroisatin, is driven by strong acid catalysis (e.g., sulfuric acid) which facilitates the nucleophilic attack of the aromatic ring onto the C=N double bond. This intramolecular electrophilic aromatic substitution is highly efficient under the specified conditions of 75-80 °C. By optimizing the molar ratios of reagents—such as using a 1:2.5 ratio of o-nitroaniline to chloral hydrate—the process minimizes side reactions and maximizes the conversion to the desired isatin intermediate. The result is a robust process where impurity profiles are tightly controlled, directly addressing the concerns of R&D directors regarding the purity and reproducibility of complex heterocyclic intermediates.
How to Synthesize 7-Nitroindole Efficiently
Implementing this synthesis in a pilot or production plant requires strict adherence to the optimized parameters defined in the patent to ensure maximum yield and safety. The process is divided into three distinct operational stages: the formation of the oximino intermediate, the cyclization to isatin, and the final reductive cyclization. Each step has been fine-tuned to balance reaction kinetics with thermal safety, allowing for a smooth transition from laboratory scale to commercial production. Operators should pay particular attention to the temperature control during the exothermic addition of reagents in the first step and the careful quenching procedures in the final reduction to prevent gas evolution issues. The detailed standardized synthesis steps, including specific reagent grades, addition rates, and workup protocols, are outlined in the guide below to facilitate immediate technology transfer.
- Condense o-nitroaniline with chloral hydrate and hydroxylamine hydrochloride in water with hydrochloric acid at 40-45°C to form the oximino intermediate.
- Cyclize the oximino intermediate using concentrated sulfuric acid at 75-80°C to generate 7-nitroisatin.
- Reduce 7-nitroisatin using a sodium borohydride and zirconium tetrachloride system in tetrahydrofuran at 0-5°C to obtain 7-nitroindole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates into tangible strategic benefits that go beyond mere technical feasibility. The primary advantage lies in the radical simplification of the supply chain; by utilizing o-nitroaniline, chloral hydrate, and hydroxylamine hydrochloride, manufacturers can source all key inputs from the global commodity chemical market. These materials are produced in massive volumes for other industries, ensuring a stable supply base that is immune to the shortages often seen with specialized fine chemicals or custom-synthesized precursors. This abundance directly contributes to cost reduction in pharmaceutical intermediates manufacturing, as the volatility associated with niche raw material pricing is eliminated. Furthermore, the absence of noble metals like palladium removes the need for expensive catalyst recovery systems or stringent heavy metal clearance testing, streamlining the quality control workflow.
- Cost Reduction in Manufacturing: The elimination of cryogenic cooling and high-pressure equipment significantly lowers capital expenditure (CAPEX) and operational expenditure (OPEX). Traditional routes requiring temperatures below -60 °C demand specialized refrigeration units that consume vast amounts of energy and require frequent maintenance. By operating at mild temperatures between 0 °C and 80 °C, this new method allows production to proceed in standard reactor trains, drastically reducing utility costs. Additionally, the avoidance of expensive palladium catalysts and phosphine ligands removes a major cost driver, while the high yields reported in the patent examples (up to 88% in the first step and 86% in the final step) minimize raw material waste, further enhancing the overall economic efficiency of the process.
- Enhanced Supply Chain Reliability: Reliance on commodity chemicals ensures that production schedules are not held hostage by the lead times of custom synthesis. Materials like o-nitroaniline and sodium borohydride are stocked by numerous global suppliers, providing multiple sourcing options that mitigate the risk of single-supplier dependency. This diversification is crucial for maintaining supply chain reliability in the face of geopolitical disruptions or logistics bottlenecks. Moreover, the simplified reaction sequence reduces the total production cycle time compared to five-step alternatives, allowing manufacturers to respond more agilely to fluctuating market demand and shorten the lead time for delivering high-purity 7-nitroindole to downstream customers.
- Scalability and Environmental Compliance: The mild reaction conditions and aqueous workups inherent in this process make it inherently safer and easier to scale from kilogram to metric-ton quantities. The absence of explosive intermediates like 1,2-dinitrobenzene and the avoidance of high-temperature decarboxylation steps reduce the process safety index, facilitating smoother regulatory approvals for new manufacturing sites. From an environmental standpoint, the process generates less hazardous waste compared to routes involving heavy metals or chlorinated solvents in excess. The ability to perform the reaction in water or common solvents like tetrahydrofuran, combined with the high atom economy of the cyclization steps, supports sustainable manufacturing practices and helps companies meet increasingly stringent environmental, social, and governance (ESG) targets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in patent CN110845388A, offering clarity on why this route is superior for industrial applications. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: Why is the NaBH4/ZrCl4 reduction system superior for 7-nitroisatin?
A: Traditional borane or NaBH4/BF3 systems often cause transitional reduction of the sensitive nitro group, leading to low yields and impurities. The NaBH4/ZrCl4 complex specifically targets the carbonyl group while leaving the nitro group intact, ensuring high chemoselectivity and purity.
Q: How does this method improve safety compared to the Bartoli synthesis?
A: The Bartoli synthesis requires cryogenic conditions below -60°C and uses highly toxic, explosive 1,2-dinitrobenzene. This new method operates at mild temperatures (0-80°C) using stable, commercially available o-nitroaniline, eliminating severe safety hazards.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process avoids expensive noble metal catalysts, high-pressure hydrogenation, and extreme temperatures. The use of commodity chemicals like o-nitroaniline and simple workup procedures makes it highly scalable and cost-effective for metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Nitroindole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust infrastructure. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel synthesis are fully realized in practice. Our state-of-the-art facilities are equipped to handle the specific thermal and safety requirements of this process, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of 7-nitroindole meets the exacting standards of the global pharmaceutical industry. We are committed to delivering not just a chemical product, but a secure, high-quality supply solution that empowers your drug development pipeline.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Whether you require custom synthesis services or bulk supply of intermediates, our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive quotations. Let us help you optimize your supply chain and accelerate your time to market with our reliable, cost-effective manufacturing solutions.
