Advanced One-Pot Synthesis of Raatinib Intermediates for Commercial Scale-Up
Advanced One-Pot Synthesis of Raatinib Intermediates for Commercial Scale-Up
The pharmaceutical industry is constantly seeking more efficient pathways to synthesize complex oncology drug intermediates, and the technology disclosed in patent CN111302997A represents a significant leap forward in this domain. This patent details a novel one-pot method for preparing the key intermediate of the broad-spectrum targeting tumor drug Raatinib, specifically (R)-2-(2,5-difluorophenyl)pyrrolidine (R)-2-hydroxysuccinate. Unlike traditional multi-step syntheses that require tedious isolation and purification after every transformation, this innovative approach consolidates Grignard formation, cyclization, and reduction into a seamless sequence. For R&D directors and process chemists, this translates to a dramatically simplified workflow that minimizes material loss and maximizes throughput. The core breakthrough lies in the ability to carry the reaction mixture directly from one stage to the next without changing the solvent system, thereby addressing critical pain points regarding waste generation and operational complexity in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of this critical heterocyclic scaffold was plagued by inefficiencies and high costs associated with fragmented processing. As illustrated in the earlier patent CN108218754A, conventional routes often involved four distinct steps with an overall yield of only about 72 percent, necessitating different solvents for each stage which complicated recovery efforts. 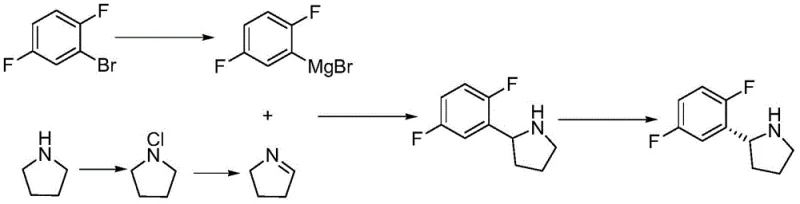 Furthermore, alternative methods such as those described in CN102224153A relied heavily on expensive transition metal catalysts like palladium acetate and required harsh cryogenic conditions ranging from -78 to -65 degrees Celsius.
Furthermore, alternative methods such as those described in CN102224153A relied heavily on expensive transition metal catalysts like palladium acetate and required harsh cryogenic conditions ranging from -78 to -65 degrees Celsius. 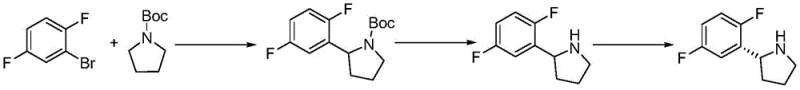 These low-temperature requirements impose severe constraints on industrial equipment, demanding specialized refrigeration infrastructure that significantly drives up energy consumption and capital expenditure. Additionally, the generation of large volumes of wastewater during the post-treatment of each isolated step creates a substantial environmental burden, making these legacy processes increasingly unsustainable for modern green chemistry standards.
These low-temperature requirements impose severe constraints on industrial equipment, demanding specialized refrigeration infrastructure that significantly drives up energy consumption and capital expenditure. Additionally, the generation of large volumes of wastewater during the post-treatment of each isolated step creates a substantial environmental burden, making these legacy processes increasingly unsustainable for modern green chemistry standards.
The Novel Approach
In stark contrast, the methodology presented in CN111302997A introduces a unified one-pot strategy that fundamentally restructures the synthetic logic. By utilizing 2,5-difluorobromobenzene as the starting material, the process generates a Grignard reagent in situ which immediately reacts with N-Boc-pyrrolidone to form the initial adduct. Instead of isolating this intermediate, the reaction mixture is treated directly with acid to remove the Boc protecting group, followed by the addition of a base to induce cyclization. This telescoping of reactions means that compounds I, II, and III never need to be isolated or dried; the solution simply flows from one chemical transformation to the next within the same reactor vessel. This continuity not only simplifies the operation process by eliminating complex post-treatments like washing, filtering, and drying between steps but also facilitates the recovery of solvents since the same medium is used throughout. The result is a process that is not only chemically elegant but also industrially robust, avoiding the use of various organic solvents and the generation of excessive waste water that characterizes the older technologies.
Mechanistic Insights into Grignard-Mediated Cyclization and Reduction
The chemical elegance of this one-pot synthesis relies on the precise control of reactivity and compatibility between the sequential reagents. The process initiates with the formation of a Grignard reagent from 2,5-difluorobromobenzene using isopropyl magnesium chloride at controlled temperatures between -10 and 15 degrees Celsius. This organometallic species then attacks the carbonyl group of N-Boc-pyrrolidone, establishing the carbon-carbon bond necessary for the pyrrolidine ring structure. Following this addition, the introduction of an acid, such as hydrochloric acid or trifluoroacetic acid, serves a dual purpose: it quenches any remaining Grignard reagent and simultaneously cleaves the tert-butoxycarbonyl (Boc) protecting group to reveal the free amine. Subsequent adjustment of the pH with a base like sodium hydroxide or potassium carbonate triggers an intramolecular cyclization, closing the ring to form the dihydropyrrole intermediate. Finally, the reduction step, achievable with sodium borohydride or catalytic hydrogenation using Pd/C or Pt/C, saturates the double bond to yield the target pyrrolidine scaffold. 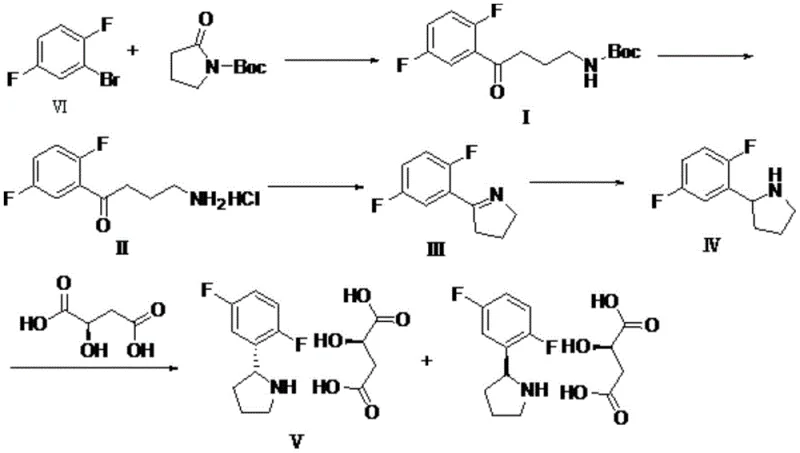 This mechanistic flow ensures that reactive intermediates are consumed immediately, minimizing side reactions and degradation.
This mechanistic flow ensures that reactive intermediates are consumed immediately, minimizing side reactions and degradation.
Impurity control is inherently built into this continuous flow design, as the avoidance of isolation prevents the accumulation of impurities that often occur during drying and storage of unstable intermediates. The final purification is achieved through a classical resolution strategy where the crude amine is salified with D-malic acid. This step is critical for achieving the high stereochemical purity required for pharmaceutical applications, leveraging the differential solubility of diastereomeric salts in ethanol. The recrystallization process effectively filters out the unwanted enantiomer and residual organic impurities, yielding the final product with an enantiomeric excess (ee) value exceeding 99 percent and chemical purity approaching 99.96 percent. This high level of purity is attained without the need for preparative chromatography, which is often a bottleneck in scaling up chiral syntheses, thus demonstrating the practical superiority of this resolution-based approach for large-scale manufacturing.
How to Synthesize (R)-2-(2,5-difluorophenyl)pyrrolidine Efficiently
Implementing this synthesis requires careful attention to temperature control and stoichiometry to ensure the cascade of reactions proceeds smoothly without stalling. The protocol begins with the generation of the Grignard reagent under inert atmosphere, followed by the sequential addition of reagents for deprotection, cyclization, and reduction without breaking the reaction containment. Detailed standard operating procedures regarding specific addition rates, stirring speeds, and quenching protocols are essential for reproducibility. Adhering to these standardized steps ensures that the benefits of the one-pot design are fully realized in a production environment.
- Generate Grignard reagent from 2,5-difluorobromobenzene and react with N-Boc-pyrrolidone to form Compound I.
- Perform acid-mediated Boc removal to obtain Compound II, followed by base-induced cyclization to form Compound III.
- Reduce Compound III to Compound IV using NaBH4 or catalytic hydrogenation, then resolve with D-malic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift to this one-pot methodology offers profound economic and logistical benefits that extend beyond simple yield improvements. The consolidation of multiple reaction steps into a single vessel significantly reduces the demand for reactor time and labor, as operators are not required to perform multiple isolation, filtration, and drying cycles. This streamlining of the workflow translates directly into lower operational expenditures and a reduced carbon footprint, aligning with corporate sustainability goals. Furthermore, the ability to recycle solvents like tetrahydrofuran and ethanol, which are used consistently throughout the process, mitigates the volatility of raw material costs and reduces the dependency on external solvent suppliers. The elimination of expensive catalysts like palladium in favor of more economical reducing agents further optimizes the bill of materials, making the final API intermediate more cost-competitive in the global market.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the drastic simplification of unit operations. By removing the need for intermediate isolation, the process eliminates the associated costs of filtration media, drying energy, and solvent swaps. The patent documentation highlights a potential reduction in production costs from approximately 8600 RMB/kg to roughly 3800 RMB/kg, indicating a massive efficiency gain. Even without guaranteeing specific figures, the qualitative mechanism of removing transition metals and reducing solvent diversity ensures a structurally lower cost base compared to legacy routes.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as 2,5-difluorobromobenzene, isopropyl magnesium chloride, and sodium borohydride ensures a robust and resilient supply chain. These reagents are widely available from multiple global vendors, reducing the risk of supply disruptions that can occur with specialized or proprietary catalysts. Additionally, the mild reaction conditions, which do not require extreme cryogenic cooling, allow for production in a wider range of facilities, increasing the flexibility of the manufacturing network and ensuring consistent delivery schedules for downstream API producers.
- Scalability and Environmental Compliance: From an environmental perspective, the reduction in wastewater generation is a critical advantage. Traditional multi-step processes generate significant aqueous waste during each workup, requiring costly treatment before discharge. This one-pot method minimizes aqueous effluent by limiting quenching and washing steps to the very end of the sequence. The simplified waste profile makes it easier to comply with stringent environmental regulations, reducing the liability and overhead associated with waste management. This scalability ensures that the process remains viable and compliant even as production volumes increase to meet commercial demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering a clear picture of the technology's capabilities. Understanding these nuances is vital for making informed decisions about technology transfer and process adoption.
Q: What is the primary advantage of the one-pot method in CN111302997A?
A: The primary advantage is the elimination of intermediate isolation steps. By maintaining the reaction in a single solvent system (typically THF) through Grignard formation, cyclization, and reduction, the process drastically reduces solvent consumption, wastewater generation, and operational time compared to traditional multi-step methods.
Q: How is high enantiomeric purity achieved in this synthesis?
A: High enantiomeric purity (>99% ee) is achieved through a final resolution step. The crude racemic amine (Compound IV) is salified with D-malic acid in ethanol, followed by recrystallization. This classical resolution technique effectively separates the desired (R)-enantiomer from the mixture.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly scalable. It avoids extreme cryogenic conditions (like -78°C) required by some prior art, operating instead at mild temperatures (0-25°C or mild heating). The use of common solvents like THF and ethanol, along with standard reagents like NaBH4 or Pd/C, facilitates easy adaptation to 100 MT scale reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Raatinib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the one-pot synthesis described in CN111302997A for the production of high-value oncology intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical efficiencies of this patent are fully captured in real-world manufacturing. Our facility is equipped with state-of-the-art reactors capable of handling Grignard chemistry and hydrogenation safely, supported by rigorous QC labs that enforce stringent purity specifications to meet the exacting standards of the pharmaceutical industry. We are committed to delivering (R)-2-(2,5-difluorophenyl)pyrrolidine derivatives with the consistency and quality required for clinical and commercial success.
We invite potential partners to engage with our technical team to explore how this advanced synthesis route can be tailored to your specific supply chain needs. By leveraging our expertise, you can secure a Customized Cost-Saving Analysis that quantifies the specific benefits for your project. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your development timeline is accelerated with a reliable and cost-effective supply of this critical intermediate.
