Advanced Synthesis of tert-Butyl 3-(Hydroxymethyl)Cyclohexanecarboxylate for Pharmaceutical Scale-Up
The pharmaceutical industry constantly seeks robust, scalable, and cost-effective pathways for synthesizing complex intermediates that serve as the backbone for next-generation therapeutics. A recent breakthrough detailed in patent CN112321433B introduces a highly efficient method for producing tert-butyl 3-(hydroxymethyl)cyclohexanecarboxylate, a critical building block with demonstrated potential in antibacterial, anti-inflammatory, and anti-tumor applications. This innovation addresses long-standing challenges in organic synthesis by replacing hazardous, multi-step protocols with a streamlined two-step sequence that prioritizes safety and yield. For R&D directors and procurement specialists alike, this patent represents a pivotal shift towards greener chemistry, utilizing readily available starting materials like cyclohexane-1,3-dicarboxylic acid to bypass the exorbitant costs and supply chain bottlenecks associated with traditional precursors. By leveraging selective mono-esterification followed by chemoselective reduction, this methodology not only enhances purity profiles but also significantly mitigates the environmental footprint typically associated with fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those documented in the Journal of Organic Chemistry (1989), rely on a convoluted four-step sequence that poses severe operational and economic risks for large-scale production. As illustrated in the reaction scheme below, the legacy process begins with the hydrolysis of dimethyl cyclohexane-1,3-dicarboxylate using barium hydroxide, a step notoriously difficult to control due to the formation of inseparable double-hydrolysis byproducts. Furthermore, the subsequent conversion to an acid chloride necessitates the use of thionyl chloride, a corrosive reagent that generates toxic fumes and requires specialized containment infrastructure. The final reduction step employs lithium aluminum hydride at cryogenic temperatures (-40°C), which not only escalates energy consumption but also introduces significant safety hazards regarding exothermic runaway reactions. These cumulative inefficiencies result in low overall yields and a complex impurity profile that demands extensive downstream purification, rendering the conventional route economically unviable for modern commercial supply chains.

The Novel Approach
In stark contrast, the methodology disclosed in patent CN112321433B offers a streamlined alternative that fundamentally reimagines the synthetic logic. The new route initiates with the direct reaction of inexpensive cyclohexane-1,3-dicarboxylic acid with di-tert-butyl dicarbonate (Boc2O) under basic catalysis, achieving selective mono-protection to form 3-(tert-butoxycarbonyl)cyclohexanecarboxylic acid. This intermediate is then subjected to a controlled reduction using borane-tetrahydrofuran complexes, which exhibit exceptional chemoselectivity by reducing the free carboxylic acid to a primary alcohol while leaving the sterically hindered tert-butyl ester intact. This strategic avoidance of acid chloride formation and the use of milder reducing agents eliminates the need for cryogenic conditions and corrosive reagents. The result is a process that is not only operationally simpler but also inherently safer, providing a reliable foundation for the commercial scale-up of complex pharmaceutical intermediates without compromising on product quality or regulatory compliance.
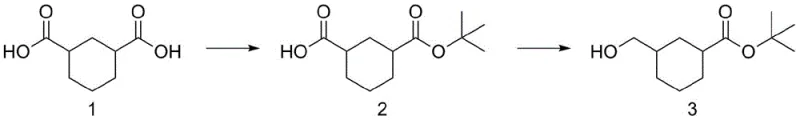
Mechanistic Insights into Selective Mono-Esterification and Reduction
The success of this novel pathway hinges on the precise control of reaction kinetics during the initial esterification phase. When cyclohexane-1,3-dicarboxylic acid reacts with di-tert-butyl dicarbonate in the presence of a nucleophilic catalyst like 4-dimethylaminopyridine (DMAP), the reaction proceeds through a mixed anhydride intermediate that favors mono-substitution. The steric bulk of the tert-butyl group, combined with the electronic deactivation of the remaining carboxylic acid after the first substitution, naturally inhibits di-esterification, thereby minimizing the formation of the di-tert-butyl ester byproduct. Experimental data within the patent indicates that optimizing the base catalyst is critical; while inorganic bases like sodium carbonate provide modest yields, organic bases such as DMAP significantly enhance conversion rates, likely by facilitating the formation of a more reactive acyl-pyridinium species that accelerates the attack of the carboxylate anion on the Boc anhydride. This mechanistic nuance ensures high selectivity, simplifying the isolation of the desired mono-acid intermediate and reducing the burden on purification resources.
Following the formation of the mono-protected intermediate, the subsequent reduction step leverages the distinct reactivity profiles of borane complexes towards different carbonyl functionalities. Unlike powerful hydride donors such as lithium aluminum hydride, which indiscriminately reduce both esters and acids, borane-tetrahydrofuran exhibits a kinetic preference for reducing carboxylic acids over esters. This selectivity is attributed to the initial coordination of the electron-deficient boron atom with the lone pairs of the carboxylic acid oxygen, forming an acyloxyborane intermediate that is rapidly reduced to the alcohol. Crucially, the tert-butyl ester moiety remains inert under these mild conditions (0°C to 25°C), preserving the protecting group for downstream synthetic utility. This chemoselectivity is the cornerstone of the process's efficiency, allowing for the direct transformation of the acid functionality without the need for additional protection-deprotection sequences, thereby shortening the overall synthetic timeline and improving the atom economy of the manufacturing process.
How to Synthesize tert-Butyl 3-(Hydroxymethyl)Cyclohexanecarboxylate Efficiently
To implement this synthesis effectively, manufacturers must adhere to specific solvent systems and stoichiometric ratios that maximize yield while maintaining safety. The patent outlines a robust protocol where the initial esterification is conducted in a biphasic or mixed solvent system, typically comprising toluene and tert-butanol, which solubilizes both the organic substrate and the inorganic or organic base catalysts. Following the isolation of the mono-acid intermediate, the reduction is performed under an inert atmosphere to prevent moisture interference with the borane reagent. While the patent provides specific experimental examples, the general workflow emphasizes temperature control and reagent addition rates to manage exotherms. For a comprehensive understanding of the exact operational parameters, including specific molar ratios and workup procedures, please refer to the standardized synthesis guide below.
- React cyclohexane-1,3-dicarboxylic acid with di-tert-butyl dicarbonate (Boc2O) in a mixed solvent system (e.g., toluene/tert-butanol) using a base catalyst like DMAP to form 3-(tert-butoxycarbonyl)cyclohexanecarboxylic acid.
- Reduce the resulting mono-acid intermediate using a borane-tetrahydrofuran complex at controlled temperatures (0°C to 25°C) to selectively convert the free carboxylic acid into a hydroxymethyl group while preserving the tert-butyl ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route translates into tangible strategic advantages that extend beyond simple unit cost metrics. By shifting away from the legacy process, organizations can mitigate a wide array of operational risks associated with hazardous material handling and complex waste streams. The elimination of thionyl chloride removes the need for expensive scrubbing systems and specialized corrosion-resistant reactors, directly lowering capital expenditure requirements for production facilities. Furthermore, the use of ambient or near-ambient temperatures in the reduction step drastically reduces energy consumption compared to the cryogenic conditions (-40°C) required by traditional methods. These factors collectively contribute to a more resilient and cost-efficient supply chain, ensuring consistent availability of high-quality intermediates even during periods of raw material volatility.
- Cost Reduction in Manufacturing: The transition to cyclohexane-1,3-dicarboxylic acid as a starting material represents a significant departure from the high-cost dimethyl esters used in prior art. This commodity chemical is widely available and priced substantially lower than specialized precursors, driving down the raw material cost basis. Additionally, the simplified two-step sequence reduces labor hours and solvent usage, while the high selectivity of the reaction minimizes the loss of valuable material to byproducts. The removal of expensive heavy metal catalysts or hazardous chlorinating agents further decreases the cost of goods sold (COGS) by eliminating the need for costly disposal and remediation processes associated with toxic waste.
- Enhanced Supply Chain Reliability: Dependence on reagents like thionyl chloride and lithium aluminum hydride often introduces supply chain fragility due to their classification as controlled or hazardous substances with strict transportation regulations. The new method utilizes reagents like di-tert-butyl dicarbonate and borane-THF, which are standard inventory items for most fine chemical manufacturers, ensuring a steady and uninterrupted flow of materials. This reliability is crucial for maintaining production schedules and meeting tight delivery windows for downstream API manufacturers. The robustness of the reaction conditions also means that production is less susceptible to disruptions caused by equipment failure or utility fluctuations, providing a more predictable lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns perfectly with modern green chemistry principles. The absence of corrosive gas evolution and the use of milder reaction conditions simplify the scale-up process from pilot plant to commercial tonnage. Waste streams are significantly cleaner, lacking the heavy metal residues or acidic byproducts that complicate effluent treatment. This ease of compliance not only reduces the administrative burden on EHS teams but also future-proofs the manufacturing site against increasingly stringent environmental regulations. The ability to scale this process without encountering the thermal hazards associated with large-scale hydride reductions makes it an ideal candidate for continuous manufacturing or large-batch production campaigns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on why this method is becoming the preferred choice for forward-thinking chemical enterprises. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this intermediate into their broader portfolio of active pharmaceutical ingredients.
Q: Why is the new synthesis route superior to the conventional literature method?
A: The conventional method relies on hazardous reagents like thionyl chloride and lithium aluminum hydride, and suffers from difficult separation of double-hydrolysis byproducts. The new route utilizes mild Boc-protection and selective borane reduction, eliminating corrosive reagents and simplifying purification.
Q: What is the optimal reducing agent for this transformation?
A: While lithium aluminum hydride and sodium borohydride were tested, the patent data indicates that borane-tetrahydrofuran solution provides the highest yield and best chemoselectivity, effectively reducing the carboxylic acid without cleaving the tert-butyl ester.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process uses commercially available raw materials like cyclohexane-1,3-dicarboxylic acid and operates under mild conditions (0°C to reflux), making it highly operable and safe for scaling up to multi-kilogram or ton-level production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable tert-Butyl 3-(Hydroxymethyl)Cyclohexanecarboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and unwavering commitment to quality. Our team has extensively analyzed the synthetic pathways outlined in CN112321433B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production necessary to bring this efficient route to market. We understand that delivering high-purity intermediates is not just about following a recipe; it involves rigorous process optimization, stringent purity specifications, and the operation of rigorous QC labs to ensure every batch meets the exacting standards required by global regulatory bodies. Our facility is equipped to handle the specific solvent systems and reducing agents required for this synthesis safely and efficiently.
We invite you to collaborate with us to leverage this advanced technology for your drug development programs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this novel route for your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments tailored to your project timelines. Let us help you secure a sustainable, cost-effective supply of this critical intermediate, enabling you to focus on what matters most: bringing life-saving therapies to patients faster and more reliably.
