Advanced Synthesis of 1-Formyl-2-Substituted Benzimidazoles for High-Performance Industrial Applications
The chemical industry constantly seeks robust solutions for metal protection and efficient synthetic routes for heterocyclic compounds. Patent CN103333116A introduces a groundbreaking methodology for the preparation of 1-formyl-2-substituted benzimidazoles, a class of compounds exhibiting exceptional utility as industrial anti-corrosion agents. This technology addresses critical pain points in traditional synthesis, such as prolonged reaction times and low yields, by employing a streamlined acid-catalyzed cyclization followed by direct formylation. The resulting derivatives, where the substituent R can range from simple alkyl groups like methyl and ethyl to complex aryloxy moieties, demonstrate superior performance in preventing acid corrosion on metal surfaces. For R&D directors and procurement specialists, this patent represents a pivotal shift towards more sustainable and cost-effective manufacturing of high-value fine chemicals. 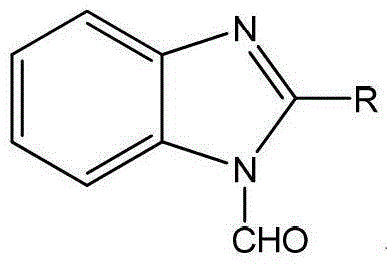
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of N-formylated benzimidazoles often relied on cumbersome and inefficient protocols that hindered large-scale commercial viability. A notable example found in existing literature involves the use of carbon dioxide and polymethylhydrosiloxane with iodopropane catalysis, a process that notoriously requires up to 24 hours to reach completion. Furthermore, such legacy methods typically suffer from suboptimal conversion rates, with reported yields hovering around a mere 58%, which drastically inflates the cost of goods sold due to material loss and extensive purification requirements. The reliance on specialized silanes and gas handling also introduces significant safety hazards and operational complexity, making scale-up a daunting task for supply chain managers who prioritize consistency and safety. These inefficiencies create a bottleneck in the production of reliable industrial anti-corrosion agent supplier materials, forcing manufacturers to seek alternative pathways.
The Novel Approach
In stark contrast, the methodology disclosed in CN103333116A leverages a straightforward two-step sequence that dramatically enhances operational efficiency and output quality. By utilizing readily available starting materials such as o-phenylenediamine and common organic acids, the process achieves complete cyclization within a concise 3 to 5-hour reflux period. The subsequent formylation step is equally rapid, requiring only 2 to 4 hours under mild reflux conditions in acetonitrile, thereby reducing the total reaction time to less than 9 hours. This acceleration is coupled with impressive yield improvements, consistently achieving between 71% and 79% across various substituents, which signifies a substantial reduction in raw material waste. The elimination of exotic reagents in favor of formic acid and hydrochloric acid simplifies the supply chain logistics and reduces the environmental footprint associated with hazardous waste disposal.
Mechanistic Insights into Acid-Catalyzed Cyclization and Formylation
The core of this synthetic strategy lies in the precise control of acid-catalyzed condensation reactions, which drive the formation of the benzimidazole ring system with high regioselectivity. In the first stage, the nucleophilic attack of the amine groups in o-phenylenediamine on the carbonyl carbon of the organic acid is facilitated by concentrated hydrochloric acid, promoting dehydration and ring closure to form the 2-substituted benzimidazole intermediate. The reaction kinetics are carefully monitored via TLC to ensure the complete consumption of the diamine starting material, preventing the carryover of unreacted amines which could complicate downstream purification. Following cyclization, the pH of the reaction mixture is critically adjusted to a range of 9-10 using sodium hydroxide, a step that induces the precipitation of the intermediate while keeping soluble impurities in the aqueous phase. This pH-controlled isolation is a key mechanism for impurity control, ensuring that the subsequent formylation step proceeds with a high-purity substrate.
The second mechanistic phase involves the N-formylation of the benzimidazole nitrogen using formic acid, which acts as both the reagent and the source of the formyl group. The reaction proceeds through an acyl substitution mechanism where the nucleophilic nitrogen attacks the carbonyl of the formic acid, followed by the elimination of water to establish the stable N-C=O bond. The use of acetonitrile as a solvent provides an optimal medium for this transformation, balancing solubility and boiling point to maintain efficient reflux temperatures without degrading the sensitive heterocyclic core. Post-reaction processing involves vacuum distillation to remove volatile components, followed by an ethanol wash that effectively scavenges residual solvents and trace acids. This rigorous purification protocol ensures that the final 1-formyl-2-substituted benzimidazole products meet stringent purity specifications required for high-performance anti-corrosion applications.
How to Synthesize 1-Formyl-2-Substituted Benzimidazole Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing these valuable compounds with minimal operational friction. The process begins with the condensation of o-phenylenediamine and a selected organic acid in the presence of an acid catalyst, followed by a neutralization and filtration step to isolate the intermediate. The second phase involves reacting this intermediate with formic acid under reflux, followed by solvent removal and recrystallization or washing to yield the final product. This standardized approach allows for easy adaptation to different R-groups, enabling the production of a diverse library of derivatives tailored to specific corrosion inhibition needs. Detailed standard operating procedures for scaling this reaction from laboratory to pilot plant are essential for maintaining quality consistency.
- Reflux o-phenylenediamine with an organic acid and concentrated hydrochloric acid for 3-5 hours, then adjust pH to 9-10 to precipitate 2-substituted benzimidazole.
- Reflux the intermediate with acetonitrile and formic acid for 2-4 hours, evaporate the solvent, and wash with ethanol to obtain the final 1-formyl product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers transformative benefits regarding cost structure and logistical reliability. The shift away from expensive silane reagents and complex gas handling systems to commodity chemicals like acetic acid and formic acid results in a drastic simplification of the raw material portfolio. This simplification not only lowers the direct material costs but also mitigates supply risk, as these commodities are globally available with stable pricing models. Furthermore, the significant reduction in reaction time from days to less than a single shift allows for higher asset turnover, meaning manufacturing facilities can produce more batches per month without capital expansion. The simplified workup, which relies on basic filtration and distillation rather than chromatography, further reduces operational expenditures related to labor and consumables.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and specialized silanes removes the need for costly metal scavenging steps and expensive reagent procurement. By utilizing high-yield reactions that exceed 70% efficiency, the process minimizes the loss of valuable starting materials, directly translating to lower cost per kilogram of the final active ingredient. The energy consumption is also optimized due to the shorter reflux times, contributing to overall utility savings in large-scale production environments.
- Enhanced Supply Chain Reliability: Sourcing o-phenylenediamine and common carboxylic acids is far more secure than relying on niche organosilicon suppliers, ensuring continuous production even during market fluctuations. The robustness of the reaction conditions, which tolerate standard industrial equipment without requiring specialized pressure vessels, enhances the flexibility of manufacturing sites. This reliability is crucial for maintaining steady inventory levels of high-purity benzimidazole derivatives for downstream customers in the agrochemical and pharmaceutical sectors.
- Scalability and Environmental Compliance: The aqueous workup and lack of heavy metal waste streamline the effluent treatment process, making it easier to comply with increasingly strict environmental regulations. The process generates minimal secondary pollution, aligning with green chemistry principles and reducing the liability associated with hazardous waste disposal. This environmental compatibility facilitates smoother regulatory approvals for new manufacturing lines and supports corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 1-formyl-2-substituted benzimidazoles. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this chemistry into their existing portfolios.
Q: How does this synthesis method compare to prior art in terms of efficiency?
A: Unlike previous methods requiring 24 hours and yielding only 58%, this patented process completes in 5-9 hours with yields exceeding 70%, significantly improving throughput.
Q: What are the primary industrial applications of these benzimidazole derivatives?
A: These compounds serve as highly effective anti-corrosion agents for metal pickling equipment and act as versatile intermediates in pharmaceutical synthesis.
Q: Does the process involve hazardous heavy metal catalysts?
A: No, the method utilizes common organic acids and formic acid, eliminating the need for expensive transition metal catalysts and simplifying waste treatment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Formyl-2-Substituted Benzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthesis routes for high-performance chemical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to industrial reactor is seamless and reproducible. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch. Our capability to handle complex heterocyclic syntheses positions us as a preferred partner for global enterprises seeking stability and quality in their supply chains.
We invite you to collaborate with us to leverage this advanced technology for your specific application needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing excellence can drive value for your organization.
