Breakthrough in Agrochemical Intermediate Manufacturing: Protective-Group-Free Hydrogenation of Thiophene Derivatives
The global demand for high-performance agrochemicals continues to drive innovation in intermediate synthesis, particularly for heterocyclic compounds that serve as critical building blocks. Patent CN101535290A introduces a transformative methodology for the preparation of 2-alkyl-3-aminothiophene derivatives, specifically addressing the long-standing challenge of synthesizing these compounds without the economic burden of amino protecting groups. Traditionally, the presence of a free amino group on a thiophene ring during hydrogenation has been considered problematic due to potential catalyst poisoning and instability. However, this intellectual property demonstrates that direct catalytic reduction of 2-alkenyl-3-aminothiophene derivatives is not only feasible but highly efficient under specific high-temperature and high-pressure conditions. For R&D directors and procurement specialists seeking reliable agrochemical intermediate suppliers, this technology represents a pivotal shift towards leaner, more cost-effective manufacturing processes that eliminate unnecessary synthetic steps while maintaining high purity standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-alkyl-3-aminothiophene derivatives has been plagued by inefficiencies inherent to multi-step protection strategies. As illustrated in prior art, such as the methods described in Japanese Patent Laid-Open Publication No. 2000-327678, chemists were compelled to introduce protecting groups like formyl, acyl, or carbamate moieties onto the amino nitrogen before attempting reduction. This requirement stems from the belief that unprotected amines would destabilize the intermediate or deactivate the hydrogenation catalyst through strong coordination or sulfur-induced poisoning. Consequently, manufacturers faced a cumbersome workflow involving an initial protection step, followed by the reduction, and finally, a deprotection step to reveal the free amine. Each additional unit operation introduces yield losses, increases solvent consumption, and generates significant chemical waste, thereby inflating the overall cost of goods sold (COGS) and complicating the supply chain for high-purity agrochemical intermediates.
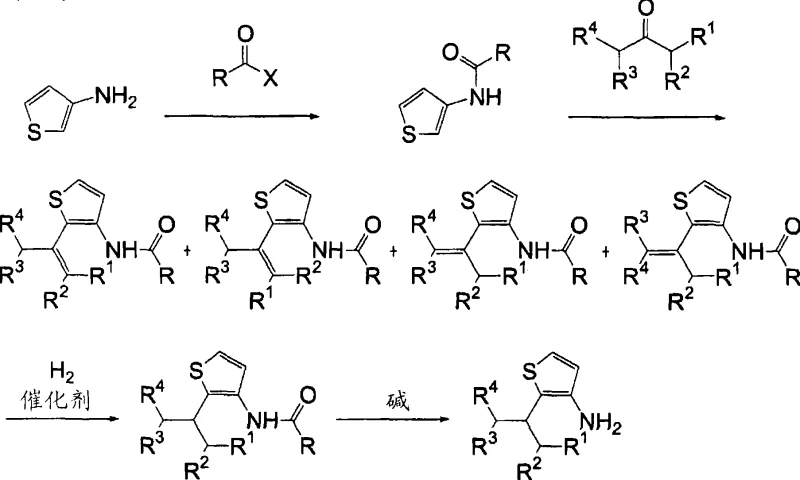
The Novel Approach
The methodology disclosed in CN101535290A fundamentally disrupts this status quo by validating a direct reduction pathway that bypasses the need for any amino protecting group. By utilizing a mixture of 2-alkenyl-3-aminothiophene isomers or their acid salts, the inventors achieved successful hydrogenation of the alkenyl side chain directly to the alkyl group. This approach leverages specific reaction conditions—namely elevated temperatures ranging from 150°C to 250°C and hydrogen pressures between 0.5 MPa and 3 MPa—to overcome the kinetic barriers and catalyst deactivation risks previously assumed to be insurmountable. The elimination of protection and deprotection steps results in a drastically simplified process flow, which translates directly into reduced operational expenditure and a smaller environmental footprint. For supply chain heads, this means fewer raw materials to source, shorter lead times for batch completion, and a more robust production schedule capable of meeting the rigorous demands of the global agrochemical market.
Mechanistic Insights into Catalytic Hydrogenation of Thiophene Derivatives
The core of this technological advancement lies in the careful selection of catalytic systems and reaction parameters that preserve the integrity of the thiophene ring while reducing the exocyclic double bond. Thiophene compounds are notoriously difficult substrates for catalytic hydrogenation because the sulfur atom can act as a potent catalyst poison, binding irreversibly to active metal sites and halting the reaction. Furthermore, harsh reduction conditions often risk hydrodesulfurization, leading to ring opening and the formation of undesirable byproducts. The patent details that catalysts based on palladium, platinum, rhodium, ruthenium, nickel, cobalt, chromium, copper, lead, or iron can be effectively employed, with palladium on carbon (Pd/C) showing particular promise. The mechanism likely involves the adsorption of the alkenyl moiety onto the catalyst surface, where it undergoes hydrogen addition, while the specific choice of solvent, such as 1-octanol, and the formation of acid salts may help modulate the basicity of the amino group, preventing excessive catalyst fouling.
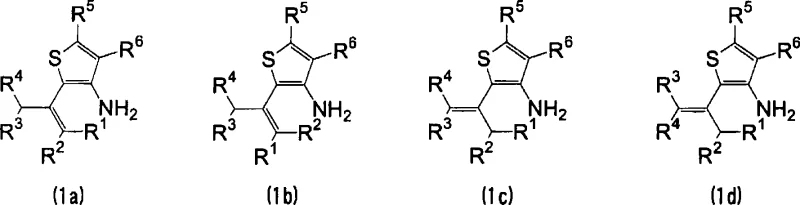
Impurity control in this synthesis is managed through the precise optimization of temperature and pressure, which minimizes side reactions such as polymerization or ring degradation. The patent examples highlight that conducting the reaction at temperatures around 200°C and pressures of 1.0 to 1.5 MPa yields the target 2-alkyl-3-aminothiophene with exceptional selectivity. For instance, the reduction of a mixture of 3-amino-2-{(E)-(4-methyl-2-penten-2-yl)}thiophene and its isomers resulted in yields exceeding 86% and even reaching 92% under optimized conditions. This high level of conversion indicates that the process is robust against the variability of starting material isomers, a common issue in industrial organic synthesis. By avoiding the introduction of foreign protecting groups, the impurity profile of the final product is inherently cleaner, reducing the burden on downstream purification units like distillation or crystallization and ensuring that the final API intermediate meets stringent quality specifications required by regulatory bodies.
How to Synthesize 3-Amino-2-(4-methylpentan-2-yl)thiophene Efficiently
To implement this synthesis effectively, operators must adhere to strict safety and procedural guidelines given the high-pressure and high-temperature nature of the reaction. The process begins with the dissolution of the alkenyl precursor in a high-boiling solvent, followed by the addition of the catalyst and sealing of the reactor. Detailed standardized operating procedures regarding catalyst loading, gas purging cycles to remove oxygen, and controlled heating ramps are essential for reproducibility and safety. The following guide outlines the critical phases of this transformation, emphasizing the parameters that drive high yield and selectivity in a commercial setting.
- Prepare the reaction mixture by dissolving the 2-alkenyl-3-aminothiophene derivative (or its acid salt) in a suitable high-boiling solvent such as 1-octanol.
- Add a heterogeneous metal catalyst, such as 5% palladium on carbon, to the reaction vessel and seal it for pressurized operation.
- Pressurize the reactor with hydrogen gas (0.5-3 MPa) and heat to elevated temperatures (150-250°C) to effect catalytic reduction of the alkenyl side chain.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this protective-group-free synthesis offers profound advantages for procurement managers and supply chain strategists looking to optimize cost structures. The most immediate benefit is the substantial reduction in raw material costs, as the expensive reagents required for introducing and removing protecting groups—such as acid chlorides, anhydrides, and deprotecting acids or bases—are entirely eliminated. This simplification also leads to a significant decrease in solvent usage and waste disposal costs, contributing to a more sustainable and economically viable manufacturing model. Furthermore, the shortened synthetic route reduces the total processing time per batch, allowing facilities to increase throughput and respond more agilely to market fluctuations without the need for capital-intensive expansion of production lines.
- Cost Reduction in Manufacturing: The elimination of protection and deprotection steps removes two entire unit operations from the manufacturing workflow, which directly correlates to lower labor costs, reduced energy consumption for heating and cooling, and decreased equipment occupancy time. By streamlining the synthesis to a single catalytic reduction step, the process efficiency is maximized, allowing for a more competitive pricing structure for the final agrochemical intermediate. This lean manufacturing approach ensures that cost savings are realized not just in material procurement but across the entire operational value chain, enhancing the overall margin potential for downstream herbicide or fungicide producers.
- Enhanced Supply Chain Reliability: Relying on fewer raw materials and simpler processing steps inherently reduces supply chain risk. The precursors required for this method are readily available, and the catalysts used are standard industrial commodities, minimizing the risk of bottlenecks associated with specialty reagents. Additionally, the robustness of the reaction conditions allows for consistent batch-to-batch quality, which is critical for maintaining long-term contracts with major agrochemical companies. This reliability ensures a steady flow of high-purity intermediates, preventing production delays that could ripple through the global supply network and affect the availability of finished crop protection products.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard high-pressure reactors and common solvents that are easily handled in large-scale facilities. The reduction in chemical waste generation aligns with increasingly stringent environmental regulations, reducing the liability and cost associated with effluent treatment. By avoiding the use of stoichiometric amounts of protecting group reagents, the atom economy of the process is significantly improved, making it an environmentally preferable choice for green chemistry initiatives. This compliance advantage facilitates smoother regulatory approvals and enhances the corporate sustainability profile of manufacturers adopting this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the feasibility and benefits of the technology for potential partners and licensees.
Q: Why is the removal of the amino protecting group significant in this synthesis?
A: Conventional methods require formyl or acyl protecting groups to prevent catalyst poisoning and side reactions, necessitating additional synthetic steps for protection and deprotection. This patent demonstrates that direct catalytic reduction is feasible without these groups, significantly shortening the process flow and reducing raw material consumption.
Q: What catalysts are effective for reducing thiophene derivatives without desulfurization?
A: The patent identifies palladium, platinum, rhodium, ruthenium, and nickel as effective catalysts. Specifically, 5% palladium on carbon showed high yields (up to 92%) under optimized conditions, proving that thiophene ring stability can be maintained during high-pressure hydrogenation.
Q: Can this process be scaled for commercial agrochemical production?
A: Yes, the method utilizes industrially advantageous catalytic reduction conditions compatible with standard high-pressure reactors. The use of common solvents like 1-octanol and robust catalysts supports large-scale manufacturing from 100 kgs to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Alkyl-3-Aminothiophene Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN101535290A for the production of next-generation agrochemical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial realities. Our state-of-the-art facilities are equipped with high-pressure hydrogenation reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-alkyl-3-aminothiophene derivative delivered meets the exacting standards required for global agricultural applications.
We invite forward-thinking R&D and procurement leaders to collaborate with us to leverage this cost-effective technology for their supply chains. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, along with specific COA data and route feasibility assessments. Let us help you secure a reliable supply of high-quality intermediates while optimizing your manufacturing economics through our expertise in process development and scale-up.
