Advanced Manufacturing of Guadecitabine: Overcoming Synthetic Bottlenecks for Commercial Scale
Introduction to Next-Generation Guadecitabine Manufacturing
The landscape of oncology therapeutics is continuously evolving, with DNA methyltransferase inhibitors playing a pivotal role in treating myelodysplastic syndromes (MDS) and acute myeloid leukemia (AML). Guadecitabine, a second-generation dinucleotide of decitabine, offers superior pharmacokinetic properties compared to its parent compound, primarily due to its resistance to rapid degradation by cytidine deaminase. The patent CN109503689B discloses a groundbreaking method for preparing Guadecitabine that addresses critical bottlenecks in existing synthetic routes. This innovation is particularly significant for a reliable Guadecitabine supplier aiming to secure the global supply chain for this high-value API. The disclosed technology leverages a convergent synthesis strategy that not only enhances yield but also drastically improves the safety profile of the manufacturing process.
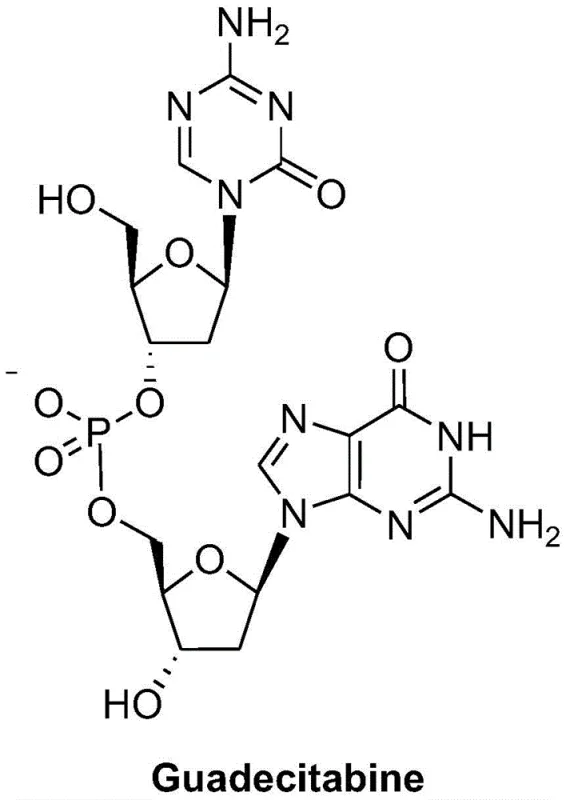
Understanding the molecular architecture of Guadecitabine is essential for appreciating the synthetic challenges involved. The molecule consists of a 5-aza-2'-deoxycytidine moiety linked via a phosphate diester bond to a 2'-deoxyguanosine unit. Traditional approaches to constructing this phosphodiester linkage often suffer from low regioselectivity or require harsh conditions that compromise the integrity of the sensitive nucleobases. The new methodology described in the patent introduces a refined protection-deprotection scheme that ensures high fidelity in bond formation. By optimizing the protecting groups on both the cytidine and guanosine fragments, the inventors have created a pathway that is amenable to rigorous quality control, ensuring the final product meets stringent purity specifications required for clinical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of Guadecitabine was fraught with significant technical and economic hurdles. One prominent method reported by Redkar utilized an enzymatic approach involving Lipozyme RM IM to protect the 5'-hydroxyl group. While enzymatic reactions are often praised for their specificity, the reliance on immobilized lipases introduces substantial cost volatility and supply chain fragility. Furthermore, the reaction conditions for these biocatalytic steps are notoriously strict and difficult to control on a multi-kilogram scale. Another existing route, detailed in patent WO2007041071, employed tris(dimethylamino)sulfonium difluorotrimethylsilicate (TAS-F) for deprotection. TAS-F is not only prohibitively expensive but also presents severe safety hazards due to its toxicity and the requirement for gasification during the reaction process. These factors collectively limit the potential for cost reduction in API manufacturing and pose unacceptable risks for large-scale industrial operations.
The Novel Approach
In stark contrast to the legacy methods, the process outlined in CN109503689B adopts a fully chemical synthesis route that bypasses the need for biological catalysts and hazardous fluorinating agents. The core of this novelty lies in the use of specifically designed intermediates: N-isobutyryl-3'-[1-(1-fluoro-1,1-diisopropylsiloxy)-1,1-diisopropylsiloxy]-2'-deoxyguanosine and 5-aza-4-(N,N-dimethylaminomethylidene)amino-5'-(4,4'-dimethoxytriphenyl)-2'-deoxycytidine. These precursors are coupled using a phosphorus reagent in a "one-pot" reaction, which dramatically simplifies the operational workflow. This convergent strategy allows for the efficient assembly of the dinucleotide backbone under mild conditions, typically at temperatures ranging from 0°C to 30°C. The elimination of complex purification steps associated with enzyme removal or toxic gas scrubbing results in a cleaner process with fewer by-products, directly translating to enhanced supply chain reliability and reduced production lead times.
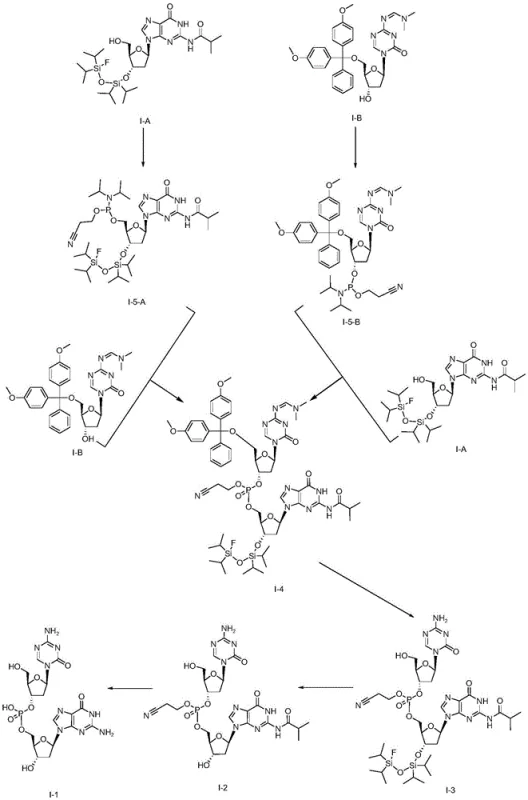
Mechanistic Insights into Phosphoramidite Coupling and Deprotection
The heart of this synthetic breakthrough is the phosphoramidite coupling mechanism, which facilitates the formation of the critical phosphodiester bond between the two nucleoside units. The reaction initiates with the activation of the phosphorus reagent, typically 2-cyanoethyl-N,N,N',N'-diisopropylphosphoramidite or its chloro-analog, by a catalyst such as pyridinium trifluoroacetate or 1H-tetrazole. This activation generates a highly reactive phosphite triester intermediate that rapidly couples with the free hydroxyl group of the acceptor nucleoside. The use of a "one-pot" protocol means that the activation and coupling occur sequentially in the same vessel without isolating the unstable phosphoramidite intermediate. This minimizes exposure to moisture and oxygen, which are detrimental to phosphorus chemistry, thereby maximizing the conversion rate. Following coupling, an oxidation step using tert-butyl hydroperoxide converts the trivalent phosphite to the stable pentavalent phosphate triester, locking the dinucleotide structure in place.
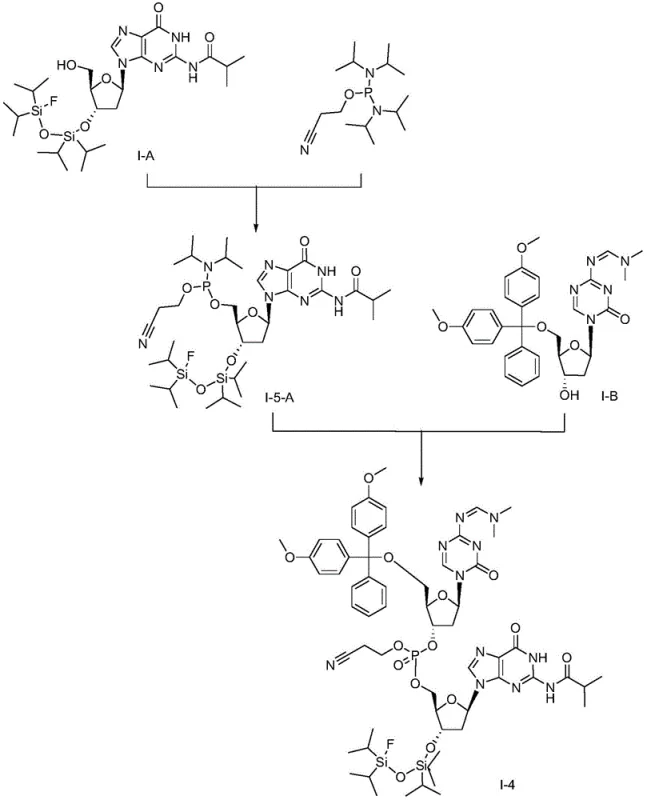
Following the coupling, the deprotection sequence is meticulously engineered to ensure orthogonality and high selectivity. The process begins with the removal of acid-labile protecting groups, such as the 4,4'-dimethoxytrityl (DMT) group, using mild organic acids like dichloroacetic acid in dichloromethane. This step exposes the necessary functional groups without affecting the silyl ethers. Subsequently, the silicon-protecting groups are cleaved using a fluoride source, such as hydrogen fluoride-pyridine complex, which is significantly safer and easier to handle than TAS-F. Finally, base-sensitive groups, including the N-isobutyryl and cyanoethyl moieties, are removed under alkaline conditions using ammonia or sodium hydroxide in methanol. This stepwise deprotection strategy prevents side reactions and ensures that the final Guadecitabine molecule is obtained with minimal impurities, a critical factor for regulatory approval and patient safety.
How to Synthesize Guadecitabine Efficiently
The practical implementation of this synthesis route involves precise control over stoichiometry and reaction conditions to achieve optimal yields. The process starts with the preparation of the key intermediates I-A and I-B, which serve as the building blocks for the final assembly. The coupling reaction is typically performed in anhydrous dichloromethane or acetonitrile under an inert nitrogen atmosphere to prevent hydrolysis of the activated phosphorus species. Careful monitoring via TLC or HPLC is essential to determine the exact endpoint of the coupling and oxidation steps. Once the fully protected dinucleotide intermediate I-4 is obtained, the deprotection cascade is initiated. Each deprotection stage requires specific workup procedures, such as aqueous extractions and silica gel chromatography, to isolate the pure intermediate before proceeding to the next step. For detailed operational parameters and specific molar ratios, refer to the standardized synthesis guide below.
- Prepare protected nucleoside intermediates I-A and I-B using silyl and trityl protecting groups respectively.
- Perform a one-pot phosphoramidite coupling reaction between I-A and I-B using a phosphorus reagent and catalyst to form intermediate I-4.
- Execute sequential deprotection: remove acid-sensitive groups, then silicon-protecting groups with fluoride, and finally base-sensitive groups to yield Guadecitabine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis method offers transformative benefits that extend beyond mere technical feasibility. The primary advantage lies in the substantial cost savings achieved by eliminating high-cost reagents. By removing the dependency on expensive immobilized enzymes and toxic fluorinating agents like TAS-F, the raw material bill of materials is significantly reduced. Furthermore, the simplified "one-pot" coupling reduces the number of unit operations, which lowers energy consumption and labor costs associated with intermediate isolation and purification. This efficiency gain allows for a more competitive pricing structure for the final API, making it accessible for broader clinical use while maintaining healthy margins for manufacturers.
- Cost Reduction in Manufacturing: The exclusion of biocatalysts and hazardous specialty chemicals directly lowers the input costs per kilogram of product. Additionally, the high selectivity of the chemical deprotection steps minimizes the loss of valuable intermediates, improving the overall mass balance of the process. This efficiency translates into a lower cost of goods sold (COGS), providing a strategic advantage in price-sensitive markets.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals rather than specialized biological enzymes mitigates the risk of supply disruptions. Enzymes often have limited shelf lives and require cold chain logistics, whereas the chemical reagents used in this process are stable and widely available from multiple global vendors. This diversification of the supply base ensures consistent production schedules and reduces the lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic gas generation make this process inherently safer and easier to scale from pilot plant to commercial tonnage. The reduced environmental footprint, characterized by lower hazardous waste generation, simplifies regulatory compliance and waste disposal management. This aligns with modern green chemistry principles and supports sustainable manufacturing goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Guadecitabine using this patented methodology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a transparent view of the technology's capabilities. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this supply source into their procurement portfolios.
Q: What are the main limitations of previous Guadecitabine synthesis methods?
A: Prior art methods often relied on expensive enzymatic catalysts like Lipozyme RM IM or highly toxic reagents such as TAS-F which required complex gasification procedures, posing safety and cost challenges for industrial scale-up.
Q: How does the new process improve safety and cost efficiency?
A: The novel route eliminates the need for expensive enzymes and toxic gas-phase reagents. It utilizes a convergent 'one-pot' coupling strategy under mild conditions, significantly simplifying operation and reducing raw material costs.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the process features high selectivity, short reaction times for deprotection steps, and uses readily available starting materials, making it highly robust and scalable for manufacturing high-purity pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Guadecitabine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes for life-saving oncology drugs like Guadecitabine. Our team of expert chemists has thoroughly analyzed the technology disclosed in CN109503689B and is well-positioned to implement this advanced manufacturing process. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demanding volume requirements of global pharmaceutical partners. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Guadecitabine meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage this innovative synthesis technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to technical excellence can drive value for your organization.
