Scalable Enzymatic Synthesis of Chiral Arformoterol Intermediate for Global Pharma Supply
Scalable Enzymatic Synthesis of Chiral Arformoterol Intermediate for Global Pharma Supply
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for synthesizing complex chiral intermediates, particularly for high-value respiratory medications like arformoterol. A significant breakthrough in this domain is detailed in patent CN111944855B, which discloses a novel method for synthesizing (R)-1-(4-(benzyloxy)-3-nitrophenyl)-2-bromoethanol, a critical chiral building block. This patent highlights a shift from traditional chemical resolution or harsh chemical reduction methods to a sophisticated biocatalytic approach using carbonyl reductase. By leveraging enzyme catalysis, the process achieves exceptional stereoselectivity and yield under mild conditions, addressing key pain points in modern API manufacturing such as safety, environmental impact, and cost efficiency. The strategic importance of this intermediate lies in its role as a precursor to (R,R)-formoterol, a potent beta-2 agonist used in asthma treatment, where chirality dictates pharmacological activity.
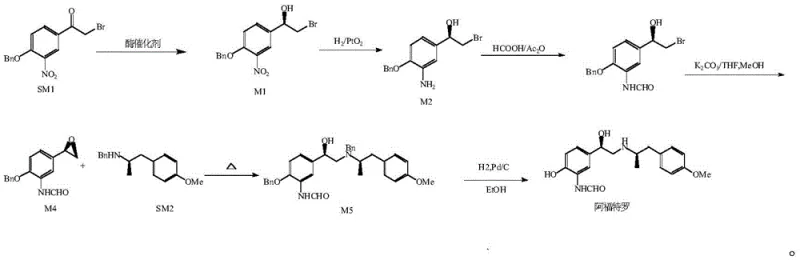
For procurement and supply chain leaders, understanding the downstream application is vital. As illustrated in the reaction pathway, the intermediate M1 is not merely a standalone chemical but a pivotal junction in the synthesis of life-saving asthma medication. The ability to produce this intermediate with high optical purity directly impacts the efficacy and safety profile of the final drug substance. Consequently, securing a reliable supply of this specific chiral intermediate through robust, patent-protected methodologies becomes a strategic imperative for pharmaceutical manufacturers aiming to optimize their production pipelines for beta-adrenoreceptor agonists.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral alcohol intermediates like M1 has relied heavily on chemical resolution or asymmetric chemical reduction using metal catalysts. These traditional approaches are fraught with significant operational and economic drawbacks that hinder efficient commercial scale-up. Chemical resolution methods, for instance, are inherently inefficient because they theoretically discard at least 50% of the material (the unwanted enantiomer), leading to poor atom economy and increased waste disposal costs. Furthermore, asymmetric chemical reductions often require the use of hazardous reducing agents such as borane or high-pressure hydrogen gas, necessitating specialized, explosion-proof equipment and rigorous safety protocols that drive up capital expenditure. The need for expensive chiral ligands and transition metal catalysts further inflates the raw material costs, while the removal of trace metal residues to meet stringent pharmaceutical purity standards adds complex purification steps to the workflow.
The Novel Approach
In stark contrast, the method disclosed in the patent data introduces a biocatalytic paradigm that elegantly circumvents these limitations by utilizing carbonyl reductase in an aqueous-organic solvent system. This novel approach operates under remarkably mild conditions, typically at temperatures between 20°C and 35°C and near-neutral pH levels, eliminating the need for extreme thermal or pressure inputs. The use of ethanol and water as the primary solvent system not only reduces the reliance on volatile organic compounds (VOCs) but also simplifies the workup procedure, as the product can be easily extracted and crystallized. Most critically, the enzymatic route demonstrates superior stereocontrol, consistently achieving enantiomeric excess (ee) values greater than 98%, which surpasses the typical 90% ee obtained via conventional chemical resolution. This leap in selectivity ensures that the downstream synthesis of arformoterol proceeds with minimal formation of toxic or inactive isomers, thereby enhancing the overall quality and safety of the final therapeutic agent.
Mechanistic Insights into Carbonyl Reductase Catalyzed Reduction
The core of this technological advancement lies in the specific activity and selectivity of the carbonyl reductase enzyme employed in the transformation of the ketone substrate SM1 to the chiral alcohol M1. The enzyme functions by facilitating the transfer of a hydride equivalent to the prochiral carbonyl carbon with high spatial precision, dictated by the enzyme's active site architecture. This biological catalyst effectively discriminates between the two faces of the planar carbonyl group, ensuring that reduction occurs exclusively to form the desired (R)-configuration. The patent specifies that the carbonyl reductase used possesses a specific activity of approximately 15 IU/mg, indicating a high turnover rate that allows for efficient conversion even at relatively low enzyme loadings (mass ratio of 0.2 to 2.0 relative to substrate). This high catalytic efficiency is crucial for industrial viability, as it minimizes the amount of biocatalyst required per batch, directly influencing the cost of goods sold (COGS).
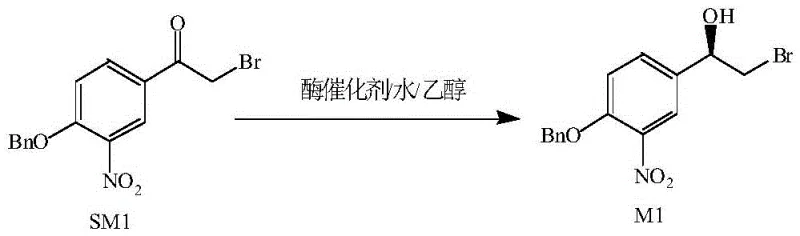
Beyond mere conversion, the mechanism inherently supports superior impurity control, a critical factor for R&D directors focused on regulatory compliance. The high specificity of the enzyme means that side reactions, such as the reduction of the nitro group or debromination, are virtually non-existent under the optimized conditions (pH 6.0-7.0). In chemical reductions, controlling chemoselectivity in the presence of sensitive functional groups like nitro and bromo substituents is often challenging and requires protecting group strategies that add steps and cost. Here, the biocatalyst naturally tolerates these functionalities, resulting in a crude product with chemical purity exceeding 98% before final crystallization. This "clean" reaction profile significantly reduces the burden on downstream purification units, allowing for simpler filtration and crystallization processes using n-heptane, which further enhances the overall process robustness and scalability.
How to Synthesize (R)-1-(4-(benzyloxy)-3-nitrophenyl)-2-bromoethanol Efficiently
Implementing this biocatalytic route requires precise control over reaction parameters to maximize yield and stereoselectivity, as outlined in the experimental data provided in the patent. The process begins with the preparation of a homogeneous reaction mixture where the substrate SM1 is dissolved in a mixed solvent of ethanol and water, with a preferred volume ratio ranging from 0.5 to 2.0. The concentration of the substrate is carefully maintained between 0.066 g/mL and 0.2 g/mL to balance reaction kinetics with solubility limits. Once the enzyme is introduced, the pH of the system must be buffered strictly within the range of 6.0 to 7.0, preferably using sodium dihydrogen phosphate and disodium hydrogen phosphate, to maintain optimal enzyme stability and activity throughout the reaction duration of 24 to 48 hours. Detailed standardized synthetic steps for replicating this high-efficiency protocol are provided in the guide below.
- Prepare a reaction mixture containing compound SM1, carbonyl reductase (0.2-2.0 mass ratio), and an ethanol-water solvent system (0.5-2.0 volume ratio).
- Adjust the system pH to 6.0-7.0 using phosphate buffers and maintain the reaction temperature between 20-35°C for 24-48 hours.
- Filter the reaction solution, extract the filtrate with ethyl acetate, concentrate, and crystallize using n-heptane to obtain the final product M1.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this enzymatic methodology represents a substantial opportunity for cost reduction in pharmaceutical intermediates manufacturing and risk mitigation. The elimination of hazardous reagents like borane and high-pressure hydrogen removes the need for specialized containment infrastructure and expensive safety monitoring systems, leading to significant operational expenditure savings. Furthermore, the use of commodity solvents like ethanol and water, coupled with the high atom economy of the enzymatic reaction, drastically reduces raw material costs and waste treatment fees associated with toxic organic solvents. The simplified downstream processing, characterized by straightforward filtration and crystallization rather than complex chromatographic separations, shortens the production cycle time and increases facility throughput, allowing for more flexible and responsive supply chain management.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the removal of expensive chiral metal catalysts and the avoidance of stoichiometric hazardous reducing agents. By replacing these high-cost inputs with a recyclable or low-loading biocatalyst system, the direct material costs are significantly lowered. Additionally, the high yield (approximately 80-84%) and superior purity reduce the loss of valuable starting materials and minimize the need for re-processing off-spec batches. The energy consumption is also markedly reduced due to the ambient temperature and pressure conditions, contributing to a lower carbon footprint and reduced utility costs for the manufacturing plant.
- Enhanced Supply Chain Reliability: From a supply continuity perspective, the reliance on readily available and stable raw materials enhances the resilience of the supply chain. Unlike specialized chemical reagents that may face geopolitical or logistical bottlenecks, the enzymes and solvents used in this process are widely sourced and easy to stockpile. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by equipment failure or utility fluctuations, ensuring a steady flow of critical intermediates to downstream API manufacturers. This reliability is essential for maintaining uninterrupted production schedules for essential respiratory medications.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up, moving seamlessly from laboratory gram-scale to multi-ton production without the engineering challenges associated with high-pressure hydrogenation reactors. The aqueous nature of the reaction medium aligns perfectly with green chemistry principles, facilitating easier wastewater treatment and compliance with increasingly stringent environmental regulations. By minimizing the generation of hazardous waste and avoiding heavy metal contamination, manufacturers can achieve sustainability goals while reducing the regulatory burden associated with environmental permitting and waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic synthesis route, based on the specific data and embodiments found in the patent literature. These insights are intended to clarify the operational feasibility and quality advantages of the technology for potential partners and licensees. Understanding these nuances is critical for making informed decisions about technology adoption and supply partnerships in the competitive landscape of chiral pharmaceutical intermediates.
Q: What are the advantages of the enzymatic method over chemical reduction for this intermediate?
A: The enzymatic method avoids hazardous reagents like borane and hydrogen, operates under mild conditions (30°C, neutral pH), and achieves significantly higher enantiomeric excess (>98% ee) compared to traditional chemical resolution methods.
Q: What is the expected yield and purity of the product M1?
A: According to the optimized process parameters in the patent data, the method consistently delivers yields around 80-84% with chemical purity exceeding 98% and optical purity (ee value) greater than 98%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes common solvents (ethanol/water) and avoids extreme pressure or temperature requirements, making it highly scalable and safer for commercial manufacturing compared to high-pressure hydrogenation routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-1-(4-(benzyloxy)-3-nitrophenyl)-2-bromoethanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development and manufacture of advanced respiratory therapies. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of materials regardless of their project phase. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the high enantiomeric excess required for arformoterol synthesis. We are committed to delivering not just a chemical product, but a comprehensive solution that guarantees supply security and regulatory compliance for your global operations.
We invite you to collaborate with us to leverage this advanced enzymatic technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this greener, more efficient route can optimize your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your path to market with confidence and precision.
