Advanced Catalytic Route for 4,4'-Dichlorobenzenesulfone Manufacturing and Commercial Scale-Up
The chemical industry is constantly seeking more sustainable and efficient pathways for producing high-value intermediates, and a significant breakthrough has been documented in patent CN111302983A regarding the preparation of 4,4'-dichlorobenzenesulfone (DDS). This compound serves as a critical monomer for engineering plastics such as polysulfone and polyethersulfone, as well as a vital intermediate in pharmaceutical and agrochemical synthesis. The traditional methods for synthesizing DDS have long been plagued by environmental concerns and operational inefficiencies, but this new technology introduces a superparamagnetic nanoparticle supported ionic liquid catalyst that fundamentally changes the process dynamics. By leveraging the unique properties of magnetic separation, this method offers a streamlined approach that eliminates the need for complex post-reaction purification steps often associated with conventional Lewis acid catalysts. For R&D directors and procurement specialists alike, understanding this shift is crucial for optimizing supply chains and reducing the total cost of ownership for high-purity specialty chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 4,4'-dichlorobenzenesulfone has relied on several established routes, each carrying significant drawbacks that impact both economic viability and environmental compliance. The sulfuric acid method, while inexpensive in terms of raw materials, suffers from low conversion rates and extended reaction periods, resulting in poor product quality and lower melting points that are unacceptable for high-performance polymer applications. Alternatively, the chlorosulfonic acid method, although capable of producing good quality products, generates severe three-waste problems (wastewater, waste gas, and solid waste) and incurs high production costs due to the corrosive nature of the reagents and the energy-intensive neutralization processes required. Furthermore, methods utilizing anhydrous aluminum trichloride as a catalyst, while effective, produce substantial amounts of hazardous waste water and gas, creating a heavy burden on waste treatment facilities and increasing the overall carbon footprint of the manufacturing process.
The Novel Approach
In stark contrast to these legacy technologies, the novel approach described in the patent utilizes a sophisticated superparamagnetic nanoparticle supported ionic liquid catalyst to drive the Friedel-Crafts reaction between chlorobenzene and thionyl chloride. This method stands out because chlorobenzene serves a dual role as both the reactant and the solvent, thereby eliminating the need for additional organic solvents that would otherwise require recovery or disposal. The process is characterized by its simplicity and high efficiency, yielding a product with a purity content exceeding 99.2% and keeping the undesirable 2,4'-isomer content below 0.1%. The integration of magnetic nanoparticles allows for the catalyst to be effortlessly separated from the reaction mixture using an external magnetic field, a feature that drastically simplifies the work-up procedure compared to traditional filtration or distillation methods. This technological leap not only enhances the yield to over 90% but also aligns perfectly with modern green chemistry principles by minimizing waste generation and energy consumption.
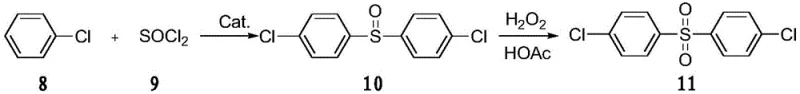
Mechanistic Insights into Magnetic Nanoparticle Catalyzed Friedel-Crafts Reaction
The core of this innovative synthesis lies in the intricate design of the catalyst, which combines the high surface area of superparamagnetic Fe3O4/SiO2 nanoparticles with the tunable acidity and stability of an ionic liquid functional group. The catalytic cycle begins with the activation of thionyl chloride by the Lewis acidic sites on the supported ionic liquid, facilitating the electrophilic attack on the chlorobenzene ring to form the 4,4'-dichlorodiphenyl sulfoxide intermediate. The magnetic core ensures that the active sites remain dispersed and accessible throughout the reaction, preventing the agglomeration that often deactivates heterogeneous catalysts. Following the initial sulfoxidation, the intermediate is subjected to an oxidation step using hydrogen peroxide in glacial acetic acid, which cleanly converts the sulfoxide to the final sulfone without introducing heavy metal contaminants. This two-stage mechanism is highly selective, effectively suppressing the formation of ortho-substituted isomers, which is a common challenge in electrophilic aromatic substitution reactions involving substituted benzenes.
From an impurity control perspective, the mechanism offers distinct advantages over homogeneous catalysis. The heterogeneous nature of the catalyst prevents the leaching of metal ions into the product stream, a critical factor for pharmaceutical intermediates where heavy metal limits are strictly regulated. The low solubility of the 4,4'-dichlorodiphenylsulfoxide in chlorobenzene at low temperatures (0-5°C) is exploited to induce crystallization, acting as a self-purification step that further enhances the purity of the intermediate before the final oxidation. This physical property, combined with the chemical selectivity of the catalyst, ensures that the final 4,4'-dichlorobenzenesulfone meets stringent specifications with a 2,4'-isomer content of less than 0.1%. Such precise control over the impurity profile is essential for downstream applications in polymerization, where even trace isomers can affect the thermal and mechanical properties of the resulting engineering plastics.
How to Synthesize 4,4'-Dichlorobenzenesulfone Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing 4,4'-dichlorobenzenesulfone with high reproducibility and scalability. The process is designed to be operationally simple, requiring standard reactor equipment but delivering superior results through the intelligent application of magnetic catalysis. The following guide summarizes the critical operational parameters and sequential steps necessary to achieve the reported yields and purity levels, serving as a foundational reference for process engineers looking to implement this technology. For a comprehensive, step-by-step laboratory procedure including exact stoichiometric ratios and safety precautions, please refer to the detailed instructions provided below.
- Conduct Friedel-Crafts reaction between chlorobenzene and thionyl chloride using the magnetic catalyst at 30°C, followed by hot filtration and low-temperature crystallization to isolate 4,4'-dichlorodiphenyl sulfoxide.
- Dissolve the isolated sulfoxide intermediate in glacial acetic acid and perform an oxidation reaction using hydrogen peroxide at 60-80°C.
- Cool the reaction mixture to precipitate the final 4,4'-dichlorobenzenesulfone product, filter, and dry to achieve purity exceeding 99.2%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this magnetic nanoparticle catalytic technology represents a strategic opportunity to optimize costs and enhance supply reliability. The elimination of auxiliary solvents and the ability to reuse the catalyst multiple times directly translate to a reduction in raw material consumption and waste disposal costs. Furthermore, the simplified work-up procedure, which relies on magnetic separation rather than complex filtration or extraction, significantly reduces processing time and labor requirements. These operational efficiencies contribute to a more resilient supply chain by shortening production cycles and reducing the dependency on scarce or expensive reagents typically associated with traditional sulfonation methods.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the dual function of chlorobenzene as both reactant and solvent, which removes the cost burden of purchasing, recovering, or disposing of additional organic solvents. Additionally, the catalyst's ability to be recovered via a magnetic field and reused for multiple cycles without significant loss of activity means that the effective cost of the catalyst per kilogram of product is drastically lowered over time. The high yield of over 90% ensures that raw material utilization is maximized, minimizing the financial loss associated with unreacted starting materials or by-products that would otherwise need to be separated and discarded.
- Enhanced Supply Chain Reliability: The reliance on readily available and commodity-grade raw materials such as chlorobenzene and thionyl chloride ensures a stable supply base that is less susceptible to market volatility compared to specialized reagents. The robustness of the catalyst and the simplicity of the reaction conditions (moderate temperatures around 30°C for the first step) reduce the risk of batch failures due to equipment malfunction or operator error. This reliability is crucial for maintaining consistent delivery schedules to downstream customers in the polymer and pharmaceutical sectors, where interruptions in the supply of key intermediates can halt entire production lines.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the heterogeneous nature of the catalyst, which avoids the heat transfer and mixing issues often encountered with viscous homogeneous reaction mixtures. The significant reduction in three-waste generation (wastewater, waste gas, and solid waste) aligns with increasingly stringent environmental regulations, reducing the liability and permitting hurdles associated with expanding production capacity. The use of hydrogen peroxide as a clean oxidant, which produces water as the only by-product in the second step, further enhances the environmental profile of the process, making it an attractive option for companies aiming to improve their sustainability metrics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the practical aspects of adopting this technology. Understanding these details is essential for evaluating the feasibility of integrating this process into existing manufacturing workflows.
Q: How is the catalyst recovered in this novel synthesis method?
A: The catalyst utilizes a superparamagnetic Fe3O4/SiO2 core, allowing for rapid and complete separation from the reaction mixture using an external magnetic field, eliminating the need for complex filtration or centrifugation steps typically required for homogeneous catalysts.
Q: What is the purity profile of the synthesized 4,4'-dichlorobenzenesulfone?
A: The process yields a product with exceptional purity, consistently exceeding 99.2% as determined by HPLC, with the critical 2,4'-isomer impurity maintained below 0.1%, which is crucial for downstream polymerization applications.
Q: Can the magnetic catalyst be reused without significant activity loss?
A: Yes, the robust nature of the supported ionic liquid catalyst allows it to be recovered, washed, and reused multiple times. Experimental data indicates that catalytic activity remains stable over at least five consecutive cycles without obvious reduction in yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Dichlorobenzenesulfone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in patent CN111302983A for the production of high-purity 4,4'-dichlorobenzenesulfone. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this magnetic nanoparticle catalyst are fully realized in a commercial setting. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications protocols, guaranteeing that every batch of DDS we produce meets the exacting standards required for engineering plastics and pharmaceutical applications, with isomer content tightly controlled below 0.1%.
We invite you to collaborate with us to leverage this innovative synthesis route for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this greener, more efficient process. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and sustainability in your supply chain.
