Scalable Synthesis of p-Trifluoromethylphenol: A Green Catalytic Route for Commercial Production
Scalable Synthesis of p-Trifluoromethylphenol: A Green Catalytic Route for Commercial Production
The global demand for fluorinated aromatic compounds continues to surge, driven by their critical role in the development of next-generation pharmaceuticals and agrochemicals. Among these valuable building blocks, p-trifluoromethylphenol stands out as a pivotal intermediate for synthesizing various bioactive molecules, including anti-cancer agents, anti-inflammatory drugs, and advanced herbicides. However, traditional manufacturing routes have long been plagued by severe environmental hazards, high energy consumption, and complex purification challenges. A groundbreaking technical solution disclosed in patent CN111559959A offers a transformative approach to this synthesis, leveraging solid acid catalysis to overcome these historical bottlenecks. This patent details a robust methodology that replaces corrosive liquid mineral acids with a regenerable strong acid cation exchange resin, fundamentally altering the economic and safety profile of producing this high-value chemical. For industry leaders seeking a reliable pharmaceutical intermediate supplier, understanding this technological shift is essential for securing a competitive edge in the supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of p-trifluoromethylphenol has relied heavily on classical diazotization protocols utilizing concentrated inorganic acids such as sulfuric acid or hydrochloric acid. These legacy processes impose significant operational burdens on manufacturing facilities, primarily due to the extreme reaction conditions required to maintain stability. Traditional diazotization typically necessitates cryogenic temperatures ranging from -5°C to 0°C to prevent the premature decomposition of the unstable diazonium salt intermediate. Maintaining such low temperatures on a commercial scale demands substantial energy input for refrigeration cycles, drastically inflating utility costs. Furthermore, the use of large quantities of strong mineral acids generates massive volumes of acidic wastewater, creating a heavy burden on waste treatment systems and complicating regulatory compliance. The separation of the product from the acidic matrix often requires multiple extraction steps using volatile organic solvents, which introduces additional safety risks related to flammability and toxicity while increasing the overall carbon footprint of the manufacturing process.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the innovative process described in CN111559959A utilizes a strong acid cation exchange resin as a solid proton source, enabling a paradigm shift towards greener chemistry. By employing this heterogeneous catalyst, the reaction can be conducted under much milder thermal conditions, specifically within a range of 0°C to 25°C, which is remarkably close to ambient room temperature. This elimination of deep freezing requirements not only reduces energy consumption but also simplifies the reactor design and operational control. The solid nature of the catalyst allows for easy separation via simple filtration or sieving immediately after the diazotization step, effectively bypassing the need for complex neutralization and extraction procedures associated with liquid acids. Moreover, the resin can be regenerated and recycled multiple times through a straightforward acid wash protocol, transforming a consumable reagent into a durable asset. This approach aligns perfectly with the strategic goals of cost reduction in agrochemical intermediate manufacturing by minimizing raw material waste and streamlining the workflow.
Mechanistic Insights into Resin-Catalyzed Diazotization
The core scientific breakthrough of this methodology lies in the unique interaction between the strong acid cation exchange resin and the sodium nitrite reagent. Unlike liquid acids that dissociate instantly and flood the reaction medium with protons, the solid resin functions as a controlled release system for hydrogen ions. As the sodium nitrite comes into contact with the resin beads, an ion exchange occurs where sodium ions displace protons on the resin surface, generating nitrous acid in situ at a moderated rate. This controlled generation is critical because it prevents the local accumulation of high concentrations of nitrous acid, which is prone to thermal decomposition and the formation of hazardous nitrogen oxides. The immediate consumption of the generated nitrous acid by the amino group of the p-trifluoromethylaniline ensures that the diazonium salt forms efficiently without significant side reactions. This mechanistic advantage is further enhanced by the electron-withdrawing nature of the trifluoromethyl group on the aromatic ring, which lowers the electron density of the amino group and stabilizes the intermediate, allowing the reaction to proceed smoothly even at elevated temperatures compared to traditional methods.
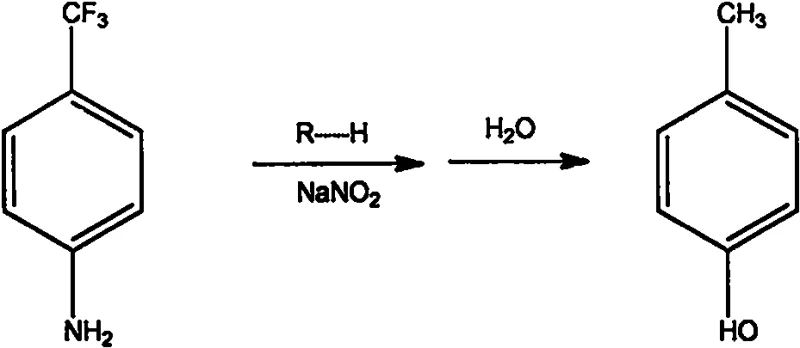
Following the formation of the diazonium salt, the process transitions seamlessly into the hydrolysis phase. The filtrate, now containing the dissolved diazonium species free from solid catalyst particles, is heated to temperatures between 95°C and 100°C. Under these conditions, the diazonium group is replaced by a hydroxyl group, yielding the target p-trifluoromethylphenol. The absence of inorganic salts and residual mineral acids in the filtrate is a distinct advantage, as it permits direct distillation for purification. The crude product can be isolated by first removing water via atmospheric distillation, followed by vacuum rectification to collect the pure phenol fraction. This streamlined purification pathway significantly reduces the impurity profile, ensuring that the final high-purity p-trifluoromethylphenol meets the stringent specifications required for sensitive pharmaceutical applications. The ability to control the reaction kinetics through the physical properties of the resin represents a sophisticated level of process engineering that enhances both yield consistency and product quality.
How to Synthesize p-Trifluoromethylphenol Efficiently
Implementing this advanced synthesis route requires precise adherence to the optimized parameters established in the patent data to ensure maximum efficiency and safety. The process begins with the careful pretreatment of the cation exchange resin to remove any oligomers or inorganic contaminants that could affect catalytic activity. Once prepared, the resin is mixed with the amine substrate, and the diazotization agent is introduced under controlled stirring. The reaction mixture is maintained within the specified temperature window to facilitate complete conversion while preserving the integrity of the diazonium intermediate. Upon completion, the solid catalyst is recovered for regeneration, and the liquid phase undergoes thermal hydrolysis. The detailed standardized synthesis steps, including specific mass ratios, timing, and regeneration protocols, are outlined in the comprehensive guide below for technical teams ready to adopt this superior methodology.
- Pretreat the strong acid cation exchange resin by washing with water and soaking in 4-5% hydrochloric acid, then rinse to neutrality.
- Mix p-trifluoromethylaniline with the resin and add aqueous sodium nitrite at 0-25°C to perform the diazotization reaction.
- Filter the mixture to recover the resin, heat the filtrate to 95-100°C for hydrolysis, and purify the product via vacuum distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this resin-based synthesis technology offers profound strategic benefits that extend far beyond simple chemical transformation. The shift from consumable liquid acids to a recyclable solid catalyst fundamentally alters the cost structure of production, driving down variable expenses associated with raw material consumption. By eliminating the need for expensive cryogenic cooling infrastructure and reducing the reliance on volatile organic solvents for extraction, manufacturers can achieve substantial cost savings in capital expenditure and operational overhead. The simplified workflow also translates into shorter batch cycles and reduced downtime for cleaning and maintenance, enhancing overall plant throughput. Furthermore, the environmental benefits of this green chemistry approach mitigate regulatory risks and lower waste disposal fees, contributing to a more sustainable and resilient supply chain capable of meeting the evolving demands of global markets.
- Cost Reduction in Manufacturing: The implementation of a regenerable cation exchange resin drastically reduces the recurring cost of acid reagents, as the catalyst can be restored to its active form and reused across multiple production batches without significant loss of efficiency. This circular usage model eliminates the continuous purchase of large volumes of concentrated mineral acids and the associated costs of neutralizing and treating acidic effluent. Additionally, the removal of organic solvent extraction steps cuts down on solvent procurement costs and the energy required for solvent recovery and distillation. The cumulative effect of these efficiencies results in a significantly leaner cost base, allowing for more competitive pricing strategies in the marketplace while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain stability by reducing dependency on specialized low-temperature equipment and hazardous chemical logistics. Operating at near-ambient temperatures minimizes the risk of equipment failure due to thermal stress and simplifies the training requirements for operational staff. The ease of catalyst separation via filtration ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by off-spec material. Moreover, the use of widely available commercial resins and standard reagents like sodium nitrite ensures that raw material sourcing remains secure and unaffected by niche supply constraints, guaranteeing continuous availability of high-purity intermediates for downstream customers.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is inherently safer and more straightforward due to the absence of exothermic runaway risks associated with rapid acid mixing. The solid-liquid separation is easily managed using standard industrial filtration units, facilitating seamless scale-up from kilograms to multi-ton capacities. From an environmental perspective, the process generates minimal hazardous waste, as the primary byproduct is saline water rather than toxic organic sludge or spent acid. This alignment with green chemistry principles simplifies permitting processes and ensures long-term compliance with increasingly strict environmental regulations, safeguarding the facility's license to operate and enhancing its corporate social responsibility profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the operational realities and benefits. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into existing manufacturing lines or for procurement specialists assessing the long-term value proposition of suppliers utilizing this method.
Q: What are the primary advantages of using cation exchange resin over mineral acids in this synthesis?
A: Using strong acid cation exchange resin eliminates the need for corrosive concentrated sulfuric or hydrochloric acids, simplifies the separation process through filtration, and allows the catalyst to be regenerated and reused multiple times, significantly reducing waste and operational costs.
Q: How does this method improve safety compared to traditional diazotization processes?
A: The resin acts as a controlled proton source, preventing the rapid accumulation of unstable nitrous acid. This allows the reaction to proceed safely at near-room temperatures (0-25°C) rather than requiring hazardous cryogenic conditions (-5 to 0°C), thereby lowering energy consumption and explosion risks.
Q: Is organic solvent extraction required for the purification of the crude product?
A: No, this novel process avoids the use of organic solvents like toluene or dichloromethane for extraction. The crude product is obtained directly from the aqueous filtrate after removing the solid resin, followed by simple distillation, which enhances environmental compliance and reduces solvent recovery costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Trifluoromethylphenol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the rigorous demands of the global pharmaceutical and agrochemical industries. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the resin-catalyzed diazotization process are translated into reliable, high-volume supply solutions. We are committed to delivering high-purity p-trifluoromethylphenol that adheres to stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our facility is equipped to handle complex fluorinated chemistries with the utmost safety and precision, guaranteeing a consistent supply of this vital intermediate for your drug discovery and development programs.
We invite you to collaborate with us to leverage these technological advancements for your specific project needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized processes can enhance your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to drive efficiency and innovation in your supply chain, ensuring you have access to the highest quality materials with the reliability you expect from a world-class manufacturer.
