Advanced Synthetic Route for High-Purity Sofosbuvir Intermediate Impurity KD003
Advanced Synthetic Route for High-Purity Sofosbuvir Intermediate Impurity KD003
The rigorous regulatory landscape governing modern pharmaceutical development necessitates the availability of highly characterized reference standards to ensure the safety and efficacy of active pharmaceutical ingredients. Patent CN111848554B introduces a sophisticated synthetic methodology specifically designed for the preparation of a critical sofosbuvir intermediate impurity, designated as KD003. This technical breakthrough addresses the persistent challenge of generating specific structural analogs required for impurity profiling and method validation in HPLC analysis. By establishing a robust pathway that leverages sequential protection and deprotection strategies, the invention enables the production of target molecules with exceptional purity profiles. For R&D directors and quality control managers, access to such well-defined impurities is not merely a regulatory checkbox but a fundamental requirement for comprehensive drug substance characterization. The methodology outlined in this patent represents a significant stride forward in the reliable supply of complex nucleoside analog standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing specific nucleoside impurities often suffer from poor regioselectivity, leading to complex mixtures that are difficult and costly to separate. In the absence of a differentiated protection strategy, direct acylation of polyhydroxylated lactones frequently results in random substitution patterns, generating a plethora of regioisomers that compromise the overall yield. Furthermore, conventional methods may rely on harsh reaction conditions that degrade the sensitive furanose ring structure, thereby introducing additional degradation products that complicate the purification process. The lack of control over the reaction trajectory often forces manufacturers to employ extensive chromatographic separations, which drastically increases production time and solvent consumption. These inefficiencies translate directly into higher costs and longer lead times, creating bottlenecks for supply chain managers who require consistent volumes of high-quality reference materials. Consequently, the industry has long sought a more precise synthetic route that minimizes byproduct formation from the outset.
The Novel Approach
The novel approach detailed in patent CN111848554B circumvents these historical limitations by implementing a stepwise orthogonal protection strategy that guarantees regiocontrol. By first masking the primary terminal hydroxyl group with a bulky silyl protecting group, the synthesis effectively directs subsequent acylation exclusively to the secondary ring hydroxyl position. This strategic ordering of reactions eliminates the formation of unwanted regioisomers, thereby streamlining the purification workflow and significantly enhancing the final product purity. The use of mild reagents such as tert-butyldimethylsilyl chloride and benzoyl chloride ensures that the core lactone scaffold remains intact throughout the transformation. This method not only improves the chemical yield but also enhances the operational safety and reproducibility of the process on a larger scale. For procurement teams, this translates to a more predictable manufacturing timeline and a substantial reduction in the cost of goods sold due to minimized waste and higher throughput efficiency.
Mechanistic Insights into Regioselective Protection and Acylation
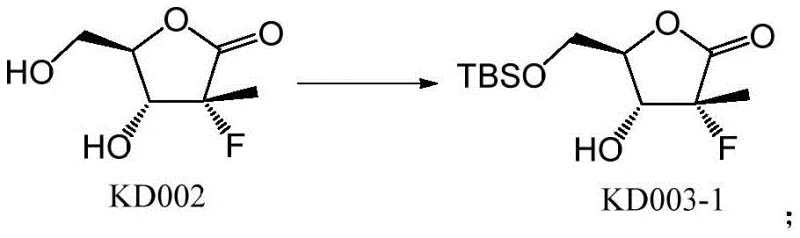
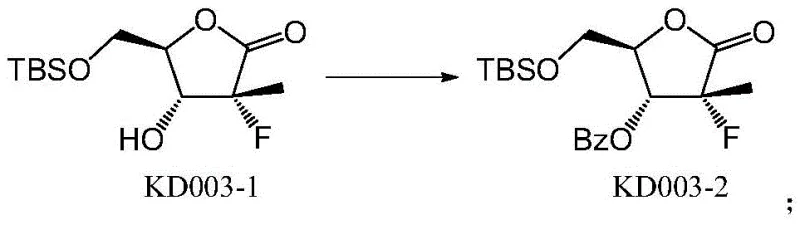
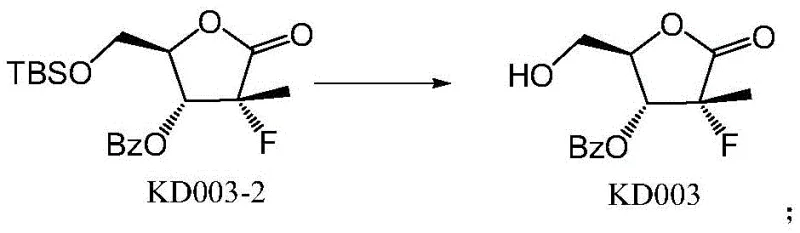
The core of this synthetic innovation lies in the precise manipulation of hydroxyl reactivity through steric and electronic differentiation. In the initial step, the primary hydroxyl group of the starting material KD002 exhibits higher nucleophilicity compared to the secondary ring hydroxyl, allowing for selective silylation using TBSCl in the presence of imidazole and pyridine. This reaction proceeds smoothly at room temperature to yield the mono-protected intermediate KD003-1 with high fidelity. Following this, the secondary hydroxyl group is activated for acylation; the addition of DMAP acts as a potent nucleophilic catalyst, facilitating the attack of the ring oxygen on the benzoyl chloride electrophile. This sequence ensures that the benzoyl group is installed solely at the desired position without affecting the silyl ether. The mechanistic elegance of this route ensures that the final deprotection step using TBAF cleanly reveals the primary alcohol without disturbing the newly formed ester linkage.
Impurity control is inherently built into the design of this synthetic pathway, as the orthogonal nature of the protecting groups prevents cross-reactivity. The silyl group is stable under the basic conditions used for benzoylation, while the ester bond formed is stable under the fluoride-mediated deprotection conditions. This orthogonality means that side reactions such as migration of the acyl group or premature cleavage of the silyl ether are virtually eliminated. Experimental data from the patent indicates that the intermediates KD003-1 and KD003-2 can be isolated with purities exceeding 99%, which sets a strong foundation for the final step. The final deprotection using tetrabutylammonium fluoride is conducted at controlled low temperatures to prevent base-catalyzed hydrolysis of the lactone ring. This meticulous attention to reaction parameters ensures that the final impurity standard KD003 is obtained with a purity profile suitable for use as a certified reference material in analytical laboratories.
How to Synthesize Sofosbuvir Intermediate Impurity KD003 Efficiently
The synthesis of KD003 is executed through a streamlined three-step protocol that balances reaction efficiency with ease of operation. The process begins with the dissolution of the starting lactone in dichloromethane, followed by the sequential addition of silylating agents to generate the first intermediate. Subsequent benzoylation is performed in a similar solvent system, utilizing catalytic amounts of DMAP to drive the reaction to completion without excessive heat. The final step involves a simple aqueous workup and chromatographic purification to isolate the target molecule. Detailed standardized operating procedures for each stage, including specific molar ratios and quenching protocols, are essential for maintaining batch-to-batch consistency. For a comprehensive guide on the exact reagent quantities and workup procedures, please refer to the structured synthesis guide below.
- Protect the terminal hydroxyl group of the starting lactone KD002 using TBSCl, imidazole, and pyridine in DCM to form the silylated intermediate KD003-1.
- Perform regioselective benzoylation on the ring hydroxyl group of KD003-1 using benzoyl chloride, triethylamine, and DMAP to yield the dibenzoyl-like intermediate KD003-2.
- Remove the terminal silyl protecting group from KD003-2 using TBAF in THF under controlled low temperatures to isolate the final impurity standard KD003.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers distinct advantages in terms of cost structure and supply chain resilience. The reliance on commodity chemicals such as pyridine, triethylamine, and benzoyl chloride means that raw material sourcing is straightforward and not subject to the volatility associated with exotic catalysts. This accessibility ensures that production schedules can be maintained without interruption, providing supply chain heads with the confidence of continuity. Furthermore, the high purity achieved at each intermediate stage reduces the burden on downstream purification processes, leading to significant savings in solvent usage and chromatography media. The robustness of the reaction conditions also implies that the process can be transferred to larger reactors with minimal re-optimization, facilitating rapid scale-up to meet market demand. These factors collectively contribute to a more economical manufacturing model that aligns with the cost-reduction goals of modern pharmaceutical procurement strategies.
- Cost Reduction in Manufacturing: The elimination of complex separation steps required for regioisomer removal directly lowers the operational expenditure associated with production. By achieving high selectivity in the initial steps, the process minimizes the loss of valuable starting materials to side products, thereby improving the overall mass balance. The use of standard solvents and reagents further drives down the cost of goods, as there is no need for specialized or expensive proprietary catalysts. This economic efficiency allows for competitive pricing of the final impurity standard, making it an attractive option for budget-conscious quality control departments seeking reliable reference materials without premium markups.
- Enhanced Supply Chain Reliability: The simplicity of the synthetic route ensures that manufacturing lead times are predictable and short, reducing the risk of stockouts for critical reference standards. Since the reagents involved are widely available from multiple global suppliers, the supply chain is not vulnerable to single-source bottlenecks or geopolitical disruptions. This diversification of the supply base enhances the resilience of the procurement strategy, ensuring that production can continue even if one vendor faces difficulties. For supply chain managers, this reliability is paramount in maintaining the continuous flow of materials required for ongoing drug development and regulatory compliance activities.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily manageable in large-scale industrial reactors. The avoidance of hazardous heavy metals or unstable intermediates simplifies waste treatment and disposal, aligning with increasingly stringent environmental regulations. The high atom economy of the protection-deprotection sequence means less chemical waste is generated per unit of product, supporting sustainability initiatives within the organization. This environmental compatibility not only reduces disposal costs but also enhances the corporate social responsibility profile of the manufacturing operation, appealing to eco-conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this sofosbuvir intermediate impurity. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation. Understanding these details helps stakeholders make informed decisions about integrating this material into their quality control workflows. For further technical discussions or custom synthesis requirements, our team is available to provide deeper insights.
Q: What is the primary advantage of this synthesis method for KD003?
A: The primary advantage is the ability to achieve exceptionally high purity (over 99%) through a regioselective protection strategy that minimizes side reactions and simplifies downstream purification.
Q: Can this process be scaled for commercial reference standard production?
A: Yes, the process utilizes common reagents like TBSCl and benzoyl chloride and avoids exotic catalysts, making it highly amenable to scale-up from gram to kilogram quantities for supply chain stability.
Q: How does this method control specific impurities?
A: By sequentially protecting the terminal hydroxyl before benzoylating the ring hydroxyl, the method prevents the formation of regioisomers and over-acylated byproducts that typically plague direct acylation attempts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sofosbuvir Intermediate Impurity Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity reference standards play in the development and approval of life-saving medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with precision. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of KD003 meets the highest international standards. Our commitment to quality is backed by a deep understanding of organic synthesis and a dedication to customer success in the competitive pharmaceutical landscape.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project needs. Whether you require specific COA data for audit purposes or route feasibility assessments for new analogs, we are ready to support your R&D and supply chain objectives. Partner with us to secure a stable, high-quality supply of essential pharmaceutical intermediates that drive your innovation forward.
