Scalable Production of m-Trifluoromethylphenol Using Recyclable Acidic Ionic Liquids
Scalable Production of m-Trifluoromethylphenol Using Recyclable Acidic Ionic Liquids
The synthesis of fluorinated phenolic compounds represents a critical challenge in modern fine chemical manufacturing, particularly for applications in pharmaceutical and agrochemical sectors where purity and process safety are paramount. Patent CN111362782B introduces a transformative methodology for the preparation of m-trifluoromethylphenol, utilizing m-trifluoromethylaniline as the starting material in conjunction with a specialized acidic ionic liquid and sodium nitrite. This innovation marks a significant departure from traditional diazotization protocols that rely heavily on corrosive mineral acids and energy-intensive cryogenic conditions. By employing an ionic liquid system, the process achieves diazotization at temperatures approaching normal ambient conditions, specifically within the range of 0-20°C, thereby enhancing operational safety and reducing the carbon footprint associated with deep cooling requirements. The reaction pathway involves the in situ generation of the diazonium species followed by direct hydrolysis in a toluene solvent system, streamlining the workflow by eliminating intermediate isolation steps. This approach not only simplifies the unit operations but also facilitates the recycling of the catalytic medium, aligning with green chemistry principles essential for sustainable industrial production.
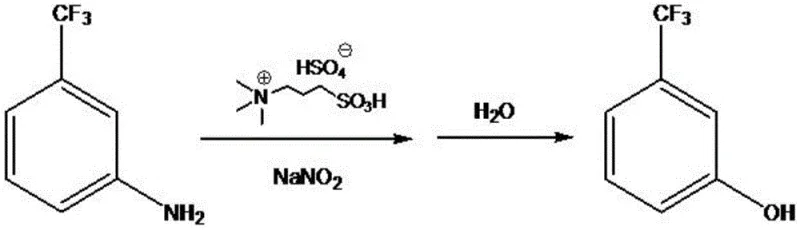
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of m-trifluoromethylphenol has relied on classical diazotization strategies involving strong inorganic acids such as concentrated sulfuric acid or hydrochloric acid. These traditional methods necessitate the generation of nitrous acid through the reaction of sodium nitrite with mineral acids, a process that is inherently unstable and requires rigorous temperature control typically between -5°C and 0°C to prevent premature decomposition of the diazonium intermediate. Such cryogenic conditions impose a substantial energy burden on manufacturing facilities, requiring specialized refrigeration equipment and increasing operational expenditures significantly. Furthermore, the use of large volumes of strong mineral acids generates considerable amounts of acidic wastewater, creating severe environmental disposal challenges and complicating downstream purification processes. The hydrolysis step in conventional routes often involves heterogeneous reaction conditions in organic-aqueous mixtures at elevated temperatures exceeding 90°C, which can lead to side reactions and the formation of complex impurity profiles that are difficult to remove, ultimately impacting the yield and quality of the final API intermediate.
The Novel Approach
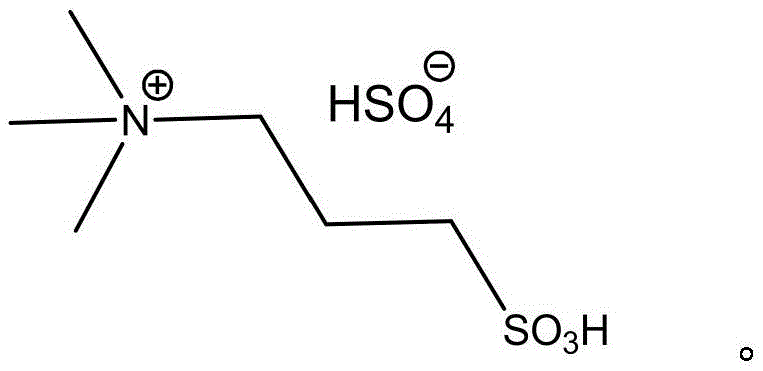
In stark contrast to these legacy techniques, the methodology disclosed in the patent leverages the unique physicochemical properties of acidic ionic liquids to create a more robust and efficient synthetic route. The use of N,N,N-trimethyl-N-sulfonic propyl ammonium bisulfate serves a dual function, acting simultaneously as an acid catalyst to facilitate diazotization and as a phase transfer catalyst to enhance the interaction between the organic amine and the aqueous nitrite source. This dual functionality allows the reaction to proceed smoothly at much milder temperatures, ranging from 0°C to 20°C, effectively removing the need for expensive cryogenic cooling infrastructure. The process design eliminates the isolation of the unstable diazonium salt; instead, upon completion of the diazotization, the temperature is simply raised to 30-35°C in the presence of toluene to effect hydrolysis. This telescoped operation reduces the number of processing steps, minimizes solvent usage, and significantly lowers the risk of handling hazardous intermediates, representing a paradigm shift towards safer and more cost-effective chemical manufacturing.
Mechanistic Insights into Ionic Liquid-Catalyzed Diazotization
The core of this technological advancement lies in the specific structure and behavior of the ionic liquid catalyst, which fundamentally alters the reaction kinetics and thermodynamics of the diazotization process. Unlike free mineral acids that exist as discrete protons in solution, the acidic ionic liquid provides a structured acidic environment where the bisulfate anion acts as the proton donor while the bulky quaternary ammonium cation stabilizes the transition states. The acidity of this ionic medium is sufficiently strong to protonate the nitrite ion and generate the active nitrosating species, yet it is moderated enough to prevent the rapid, uncontrolled decomposition often seen with strong mineral acids. This controlled release of nitrous acid ensures that it reacts preferentially with the m-trifluoromethylaniline substrate rather than decomposing into nitrogen oxides, thereby improving atom economy. Additionally, the amphiphilic nature of the ionic liquid enhances the solubility of the organic amine in the reaction medium, effectively acting as a phase transfer catalyst that bridges the gap between the aqueous nitrite phase and the organic substrate, accelerating the rate of diazotization even at near-ambient temperatures.
From an impurity control perspective, the mild reaction conditions afforded by the ionic liquid system are instrumental in maintaining high product purity. In traditional high-temperature hydrolysis or uncontrolled low-temperature diazotization, side reactions such as coupling with unreacted amine or decomposition into tar-like byproducts are common. By maintaining the diazotization temperature between 0°C and 20°C and the hydrolysis temperature between 30°C and 35°C, the thermal stress on the sensitive diazonium intermediate is minimized. This precise thermal management prevents the formation of polymeric impurities and chlorinated or sulfonated byproducts that might arise from the use of hydrochloric or sulfuric acid respectively. The result is a cleaner reaction profile that simplifies the subsequent distillation and purification steps, ensuring that the final m-trifluoromethylphenol meets the stringent quality specifications required for downstream pharmaceutical applications without the need for extensive chromatographic purification.
How to Synthesize m-Trifluoromethylphenol Efficiently
The practical implementation of this synthesis route involves a straightforward sequence of mixing, reaction, and separation steps that are highly amenable to standard chemical processing equipment. The process begins with the preparation or procurement of the specific acidic ionic liquid, followed by the sequential addition of the amine substrate and the nitrite source under controlled stirring. The reaction is monitored to ensure complete conversion before the temperature is adjusted for the hydrolysis phase. A key feature of this protocol is the direct transition from diazotization to hydrolysis without intermediate workup, which saves time and reduces material loss. The detailed standardized synthesis steps, including specific molar ratios, addition rates, and workup procedures derived from the patent examples, are outlined in the technical guide below for R&D teams looking to replicate or scale this chemistry.
- Prepare the acidic ionic liquid catalyst by reacting trimethylamine with propane sultone, followed by acidification with sulfuric acid and vacuum drying.
- Mix m-trifluoromethylaniline with the ionic liquid and slowly add aqueous sodium nitrite at 0-20°C to perform diazotization without isolating the intermediate salt.
- Add toluene solvent directly to the reaction mixture, heat to 30-35°C for hydrolysis, separate the organic layer, and recover the ionic liquid from the aqueous phase using cation exchange resin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ionic liquid-based technology offers compelling economic and logistical benefits that extend beyond simple yield improvements. The most significant advantage lies in the drastic simplification of the process flow, which translates directly into reduced operational expenditures and enhanced throughput. By eliminating the need for cryogenic cooling systems and replacing corrosive mineral acids with a recyclable ionic medium, manufacturers can achieve substantial cost savings in both energy consumption and raw material procurement. The ability to recycle the ionic liquid catalyst multiple times without significant degradation means that the effective cost per kilogram of catalyst consumed is negligible compared to the continuous purchase of stoichiometric amounts of strong acids. Furthermore, the reduction in hazardous waste generation lowers the costs associated with environmental compliance and waste disposal, making the overall manufacturing process more sustainable and economically viable in the long term.
- Cost Reduction in Manufacturing: The replacement of concentrated sulfuric and hydrochloric acids with a reusable ionic liquid eliminates the recurring cost of purchasing large volumes of corrosive reagents. Since the ionic liquid acts as a catalyst and is recovered from the aqueous phase via cation exchange resin regeneration, the material cost for the acid component is amortized over many batches. Additionally, operating at near-ambient temperatures (0-20°C) removes the heavy energy load associated with maintaining sub-zero reaction conditions, leading to significantly lower utility bills for refrigeration and heating. The simplified workup, which avoids complex neutralization and salt filtration steps typical of mineral acid processes, further reduces labor and processing time, contributing to a leaner and more cost-efficient production model.
- Enhanced Supply Chain Reliability: The raw materials required for this process, specifically m-trifluoromethylaniline and sodium nitrite, are commodity chemicals with stable global supply chains, ensuring consistent availability for production planning. The robustness of the ionic liquid catalyst, which tolerates slight variations in temperature and mixing better than traditional nitrous acid systems, reduces the risk of batch failures due to process upsets. This reliability ensures a steady output of high-purity m-trifluoromethylphenol, allowing supply chain managers to maintain tighter inventory controls and meet delivery commitments to downstream pharmaceutical clients without the buffer stocks often required for less predictable chemical processes.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the homogeneous nature of the initial reaction mixture and the manageable exotherm profile at mild temperatures. The absence of large quantities of inorganic salts in the waste stream simplifies wastewater treatment, as the primary aqueous waste contains the recoverable ionic liquid rather than dissolved sulfate or chloride salts. This aligns with increasingly strict environmental regulations regarding industrial effluent, reducing the regulatory burden on manufacturing sites. The closed-loop potential of the ionic liquid system supports a circular economy approach, enhancing the corporate sustainability profile of the manufacturer and appealing to eco-conscious partners in the global supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ionic liquid catalyzed synthesis. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a reliable foundation for feasibility assessments. Understanding these details is crucial for R&D directors evaluating the technical fit and procurement officers calculating the total cost of ownership for this advanced manufacturing route.
Q: What are the primary advantages of using acidic ionic liquids over traditional mineral acids in diazotization?
A: Acidic ionic liquids replace hazardous concentrated sulfuric or hydrochloric acids, allowing the reaction to proceed at near-room temperature (0-20°C) rather than cryogenic conditions (-5 to 0°C). Furthermore, the ionic liquid acts as a dual catalyst (acid and phase transfer agent) and can be recycled from the aqueous phase, significantly reducing chemical waste and operational costs.
Q: How does this process improve safety compared to conventional methods?
A: Traditional diazotization requires unstable nitrous acid generated at very low temperatures to prevent decomposition. This novel method utilizes the stabilizing effect of the ionic liquid, enabling the reaction at milder temperatures (0-20°C) and eliminating the need for extreme cooling infrastructure, thereby reducing energy consumption and thermal runaway risks.
Q: Is the ionic liquid catalyst recoverable for industrial scale-up?
A: Yes, the process is designed for sustainability. After hydrolysis, the reaction mixture separates into an organic layer containing the product and an aqueous layer containing the ionic liquid. The aqueous phase can be treated with acidic cation exchange resin to regenerate the ionic liquid, which is then reused in subsequent batches without significant loss of catalytic activity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable m-Trifluoromethylphenol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates like m-trifluoromethylphenol play in the development of next-generation pharmaceuticals and agrochemicals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the ionic liquid diazotization method can be seamlessly transferred from the lab to the plant. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our capability to handle complex fluorinated chemistries safely and efficiently makes us an ideal partner for companies seeking to secure their supply chain for critical building blocks.
We invite you to collaborate with us to explore how this advanced synthesis route can optimize your production costs and improve your product quality. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your project timelines and commercial goals effectively.
