Advanced Solid-Phase Manufacturing Strategies for High-Purity GLP-1 Receptor Agonists
Introduction to Next-Generation GLP-1 Agonist Manufacturing
The pharmaceutical landscape for Type 2 diabetes and obesity treatment has been revolutionized by Glucagon-like peptide-1 (GLP-1) receptor agonists, with Liraglutide and Semaglutide standing as cornerstone therapeutics. Patent CN113748125A introduces a sophisticated and economically viable methodology for the preparation of these critical peptides and their analogs, including D-Liraglutide and D-Semaglutide. Unlike traditional recombinant DNA techniques that rely on complex biological expression systems in Saccharomyces cerevisiae, this invention leverages a robust solid-phase peptide synthesis (SPPS) approach combined with strategic liquid-phase fragment preparation. The core innovation lies in the utilization of specific dipeptide building blocks, namely Fmoc-Arg(Pbf)-Gly-OH and Fmoc-Glu(OtBu)-Gly-OH, which streamline the elongation process and significantly mitigate the formation of difficult-to-remove impurities. This technical breakthrough offers a compelling alternative for reliable pharmaceutical intermediates supplier networks seeking to diversify their manufacturing capabilities beyond fermentation-based routes.
For R&D directors and process chemists, the significance of this patent extends beyond mere synthesis; it represents a paradigm shift in impurity control and process reproducibility. The document details a comprehensive Fmoc-based strategy that ensures high fidelity in amino acid sequencing while maintaining the integrity of sensitive side-chain functionalities. By anchoring the peptide chain to a 2-Chlorotrityl chloride (2-CTC) resin, the method facilitates mild cleavage conditions that are crucial for preserving the structural nuances of these large peptide molecules. Furthermore, the inclusion of detailed purification protocols involving multi-step preparative HPLC underscores the commitment to achieving substantially pure final products, a prerequisite for regulatory approval in major markets. This approach not only addresses the growing global demand for GLP-1 therapies but also provides a scalable framework for cost reduction in pharmaceutical intermediates manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Liraglutide and Semaglutide has been dominated by recombinant DNA technology, where the peptide precursor is expressed in yeast hosts such as Saccharomyces cerevisiae. While effective, this biological route presents inherent challenges that complicate the supply chain and increase production costs. The fermentation process requires stringent control over biological variables, and the subsequent downstream processing involves complex enzymatic modifications, such as acylation and N-terminal extension removal, which can introduce variability in yield and purity. Moreover, recombinant methods often struggle with specific impurity profiles related to host cell proteins and incomplete enzymatic conversions, necessitating extensive and costly purification steps. The reliance on biological systems also limits flexibility; modifying the sequence to create analogs like D-Liraglutide often requires re-engineering the entire expression vector, leading to prolonged development timelines and increased R&D expenditure for high-purity pharmaceutical intermediates.
The Novel Approach
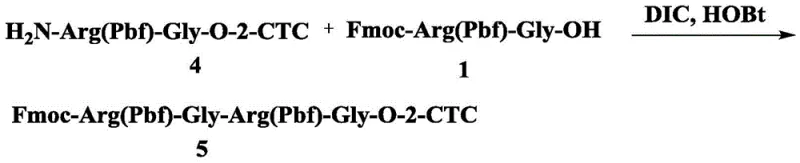
In contrast, the methodology outlined in CN113748125A employs a fully chemical synthetic route that offers superior control over the molecular architecture of the target peptides. By utilizing a solid-phase synthesis strategy anchored on 2-CTC resin, the process eliminates the biological variability associated with fermentation. The key differentiator is the strategic incorporation of pre-synthesized dipeptide fragments, specifically Fmoc-Arg(Pbf)-Gly-OH and Fmoc-Glu(OtBu)-Gly-OH, into the elongation cycle. This "dipeptide insertion" technique drastically reduces the number of coupling cycles required and minimizes the risk of racemization and deletion sequences that plague long linear syntheses. The result is a more streamlined process that enhances overall yield and simplifies the impurity profile, making it an ideal solution for commercial scale-up of complex pharmaceutical intermediates. This chemical approach provides manufacturers with the agility to rapidly adapt to market demands for various GLP-1 analogs without the lead time associated with biological strain development.
Mechanistic Insights into Dipeptide Fragment Coupling Strategy
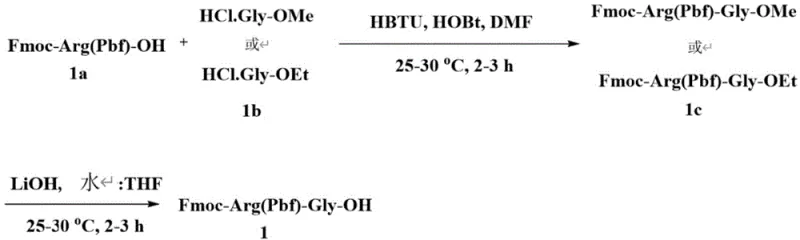
The core mechanistic advantage of this patent lies in the reduction of "Endo-Gly" impurities through the use of dipeptide fragments. In standard SPPS, coupling single amino acids sequentially can lead to incomplete reactions or side reactions, particularly at sterically hindered sites or sequences prone to aggregation. By pre-forming the Arg-Gly and Glu-Gly bonds in the liquid phase under controlled conditions, the patent ensures that these specific linkages are established with high fidelity before being introduced to the solid support. The liquid phase synthesis of fragments like Fmoc-Arg(Pbf)-Gly-OH utilizes coupling agents such as HBTU and HOBt in DMF, allowing for rigorous monitoring via TLC and purification via flash column chromatography prior to resin loading. This pre-validation of critical bond formations effectively removes a significant source of potential failure from the solid-phase cycle, thereby enhancing the overall purity of the crude peptide and reducing the burden on downstream purification processes.
Furthermore, the protection group strategy employed is meticulously designed to balance stability during synthesis with ease of removal during the final cleavage. The use of Pbf (2,2,4,6,7-pentamethyldihydrobenzofuran-5-sulfonyl) for arginine and OtBu (tert-butyl ester) for glutamic acid provides robust protection against the piperidine used for Fmoc deprotection cycles. The anchoring to 2-CTC resin is particularly noteworthy; unlike Wang or Rink amide resins that require strong trifluoroacetic acid (TFA) for cleavage, 2-CTC allows for the release of the protected peptide fragment using mild acidic conditions or specific cleavage cocktails. This orthogonality is crucial for preserving the acid-sensitive modifications introduced later in the synthesis, such as the fatty acid acylation in Liraglutide or the PEGylation in Semaglutide. The mechanistic precision ensures that the final global deprotection step yields a product with minimal side-chain damage, directly contributing to the high purity specifications required for clinical applications.
How to Synthesize Liraglutide Efficiently
The synthesis of Liraglutide via this patented route involves a systematic sequence of anchoring, elongation, side-chain modification, and cleavage. The process begins with the preparation of high-quality dipeptide fragments, which serve as the foundational building blocks for the peptide chain. These fragments are then anchored to the resin, followed by iterative cycles of Fmoc deprotection using 20% piperidine in DMF and coupling of subsequent amino acids or dipeptide units using activators like DIC and HOBt. A critical stage involves the selective deprotection of the Lysine side chain (using hydrazine for Dde removal) to facilitate the attachment of the gamma-glutamyl spacer and the palmitic acid tail. The final steps involve global deprotection and cleavage from the resin using a TFA-based cocktail, followed by a rigorous three-step HPLC purification protocol to achieve pharmaceutical-grade purity. For detailed operational parameters and stoichiometric ratios, please refer to the standardized guide below.
- Preparation of key dipeptide fragments Fmoc-Arg(Pbf)-Gly-OH and Fmoc-Glu(OtBu)-Gly-OH using liquid phase coupling.
- Anchoring the initial fragment to 2-CTC resin followed by sequential Fmoc-deprotection and coupling cycles.
- Side-chain modification with fatty acids or PEG linkers, followed by global deprotection and multi-step HPLC purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the transition from recombinant to chemical synthesis for GLP-1 agonists offers profound strategic benefits. The primary advantage is the decoupling of production from biological constraints, which significantly enhances supply chain reliability. Chemical synthesis facilities can be scaled up or down with greater flexibility than fermentation plants, allowing manufacturers to respond more agilely to fluctuating market demands without the long lead times associated with bioreactor validation and batch cultivation. This flexibility translates into reduced lead time for high-purity pharmaceutical intermediates, ensuring that downstream drug product manufacturers can maintain consistent inventory levels even during periods of surging global demand for diabetes and obesity treatments. Additionally, the chemical route mitigates the risk of biological contamination, a critical quality attribute that simplifies regulatory compliance and reduces the complexity of quality control testing.
- Cost Reduction in Manufacturing: The implementation of dipeptide fragment coupling significantly optimizes the manufacturing cost structure by improving overall process efficiency. By reducing the total number of coupling cycles and minimizing the formation of hard-to-remove impurities, the process lowers the consumption of expensive reagents and solvents per kilogram of final product. Furthermore, the simplified impurity profile reduces the load on preparative HPLC purification, which is often the most cost-intensive step in peptide manufacturing. The elimination of complex enzymatic steps and the associated biological reagents further drives down the variable costs, resulting in substantial cost savings that can be passed down the supply chain or reinvested into capacity expansion.
- Enhanced Supply Chain Reliability: Adopting a fully chemical synthetic route diversifies the sourcing options for key therapeutic peptides, reducing dependency on a limited number of biotechnology vendors. Chemical intermediates such as Fmoc-protected amino acids and coupling reagents are widely available from multiple global suppliers, creating a resilient supply network that is less susceptible to single-point failures. This redundancy ensures continuous availability of critical materials, safeguarding production schedules against disruptions. Moreover, the scalability of solid-phase synthesis allows for rapid ramp-up of production capacity, enabling suppliers to meet urgent procurement needs and secure long-term supply agreements with confidence.
- Scalability and Environmental Compliance: The described process is inherently designed for industrial scale-up, utilizing standard reactor equipment and conditions that are easily transferable from pilot to commercial scale. The use of DMF and other common organic solvents allows for established recovery and recycling protocols, aligning with modern environmental sustainability goals. By optimizing reaction concentrations and minimizing waste generation through higher yields, the process supports greener manufacturing practices. This alignment with environmental regulations not only reduces disposal costs but also enhances the corporate sustainability profile of the manufacturing entity, a factor increasingly weighted in vendor selection criteria by multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the solid-phase synthesis of GLP-1 receptor agonists as described in the patent literature. These insights are derived from the specific process parameters and advantageous effects detailed in the documentation, providing clarity on purity, scalability, and methodological robustness. Understanding these aspects is crucial for stakeholders evaluating the feasibility of integrating this synthetic route into their existing manufacturing portfolios or sourcing strategies for next-generation metabolic therapies.
Q: How does the dipeptide fragment strategy improve purity in GLP-1 synthesis?
A: By utilizing pre-formed dipeptide fragments like Fmoc-Arg(Pbf)-Gly-OH, the process significantly reduces the formation of Endo-Gly impurities that typically arise from single amino acid couplings in long peptide chains.
Q: What resin is preferred for the initial anchoring step in this protocol?
A: The protocol specifies the use of 2-Chlorotrityl chloride (2-CTC) resin, which allows for mild acidic cleavage conditions that preserve acid-labile side-chain protecting groups until the final global deprotection step.
Q: Is this synthetic route scalable for industrial production?
A: Yes, the method is designed for industrial scalability, utilizing standard SPPS reagents and conditions (25-30°C) that can be adapted from gram-scale laboratory synthesis to multi-kilogram commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Liraglutide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodologies described in CN113748125A for the global supply of GLP-1 receptor agonists. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent, high-quality supply regardless of volume requirements. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, including the advanced HPLC methods necessary to separate complex peptide impurities. We are committed to leveraging these innovative dipeptide fragment strategies to deliver cost-effective and reliable solutions for the manufacturing of Liraglutide, Semaglutide, and their analogs, positioning us as a trusted partner in your supply chain.
We invite procurement leaders and R&D teams to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume and purity requirements. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized solid-phase synthesis capabilities can enhance your project's economic viability. By collaborating with NINGBO INNO PHARMCHEM, you gain access to a robust supply network dedicated to advancing the availability of life-saving metabolic therapies through superior chemical manufacturing excellence.
