Advanced pH-Controlled Synthesis of Pyrazolone Derivatives for Scalable Agrochemical Production
Advanced pH-Controlled Synthesis of Pyrazolone Derivatives for Scalable Agrochemical Production
The global demand for high-performance agrochemical intermediates necessitates manufacturing processes that balance rigorous purity standards with economic efficiency. Patent CN101815705A introduces a transformative methodology for the synthesis of pyrazolone derivatives, specifically targeting the production of compounds represented by formula (3). These structures serve as critical building blocks in the development of next-generation pesticides and herbicides. The core innovation lies in a meticulously controlled three-step pH modulation strategy conducted within a water-organic solvent system. Unlike traditional acylation protocols that often struggle with selectivity issues, this approach leverages precise alkalinity adjustments to dictate reaction kinetics. By maintaining the aqueous phase pH within narrow windows—initially above 11.5, then dropping below 11.5, and finally stabilizing between 10.0 and 11.5—the process effectively suppresses the formation of di-acylated impurities. This technical breakthrough offers a compelling value proposition for reliable agrochemical intermediate suppliers seeking to optimize their production portfolios.
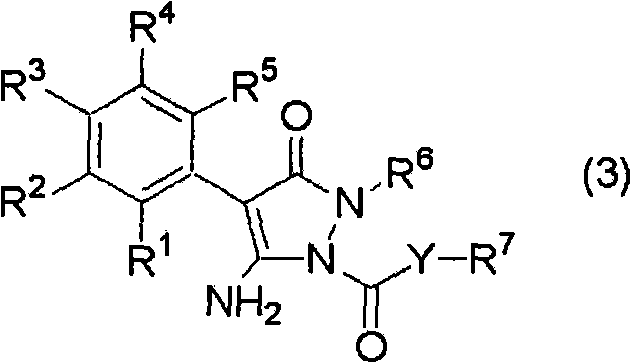
The structural integrity of the final product, as depicted in the associated chemical diagrams, is paramount for downstream biological efficacy. The ability to consistently produce high-purity pyrazolone derivatives ensures that subsequent formulation steps in pesticide manufacturing proceed without interference from reactive impurities. This patent data underscores a shift towards more sophisticated process chemistry where reaction conditions are dynamically managed rather than statically set. For R&D directors evaluating potential licensing opportunities or process upgrades, the detailed mechanistic control described in CN101815705A represents a significant leap forward in synthetic reliability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrazolone derivatives via acylation has been plagued by inherent selectivity challenges, as documented in prior art such as US Patent 6,294,567. Conventional techniques typically involve the dropwise addition of an acid halide to a mixture of the pyrazolone compound and a base, or vice versa, without rigorous real-time pH monitoring. In these uncontrolled environments, the local concentration of base can fluctuate wildly, leading to inconsistent deprotonation of the pyrazolone nitrogen. This inconsistency often results in the over-acylation of the substrate, generating di-acylated byproducts that are structurally similar to the desired mono-acylated product. Separating these impurities is notoriously difficult and costly, often requiring multiple recrystallization steps or complex chromatography, which drastically erodes overall yield. Furthermore, the exothermic nature of acylation reactions, if not managed through precise pH buffering, can lead to thermal runaways that degrade sensitive functional groups on the aromatic ring.
The Novel Approach
The methodology disclosed in CN101815705A fundamentally reengineers the reaction landscape by introducing a tri-phasic pH control protocol. Step A establishes a highly alkaline environment (pH > 11.5) to ensure complete deprotonation of the starting pyrazolone compound (1), creating a highly nucleophilic species ready for attack. Step B acts as a transition phase where the initial addition of the acid halide (2) consumes excess base, naturally lowering the pH to a threshold of 11.5. Crucially, Step C maintains the reaction within a 'Goldilocks zone' of pH 10.0 to 11.5 by co-feeding base and acid halide. This dynamic equilibrium ensures that there is always sufficient base to neutralize the generated acid (HX) but not so much that it promotes side reactions or hydrolysis of the acid halide. This precise control mechanism effectively eliminates the formation of the di-acyl impurity, which was observed at 3.0% in comparative examples versus only 1.39% in the optimized process, demonstrating a clear pathway for cost reduction in agrochemical manufacturing through improved material efficiency.
Mechanistic Insights into pH-Controlled Acylation
To fully appreciate the sophistication of this synthesis, one must examine the interplay between the nucleophile and the electrophile under varying alkaline conditions. The reaction initiates with the pyrazolone compound (1), shown below, which possesses an acidic NH proton capable of deprotonation by strong bases like sodium hydroxide.
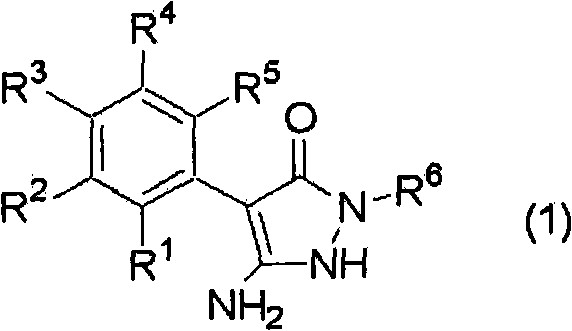
In the presence of the base, the pyrazolone ring forms an anionic species that is highly reactive towards the acid halide (2), depicted below. However, the reactivity is a double-edged sword; if the pH is too high, the resulting product can undergo a second acylation at the amino group or other nucleophilic sites. Conversely, if the pH drops too low, the starting material remains protonated and unreactive, leading to accumulation of unreacted acid halide which may hydrolyze in the aqueous phase.
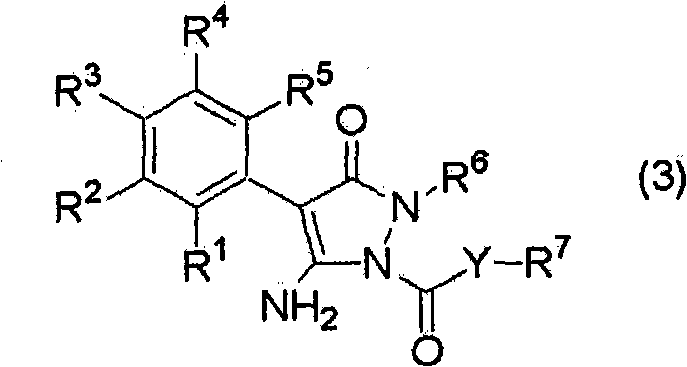
The patent's specific pH window of 10.0 to 11.5 during the main reaction phase (Step C) is chemically engineered to balance these competing factors. At this pH, the concentration of the pyrazolone anion is sufficient to drive the reaction forward at a practical rate, yet the basicity is moderated enough to prevent the deprotonation of the newly formed amide bond, thereby kinetically blocking the pathway to di-acylation. This mechanistic nuance is critical for high-purity agrochemical intermediate production, as it minimizes the generation of hard-to-remove impurities at the source rather than relying on downstream purification. The use of a water-organic solvent system further aids in heat dissipation and facilitates the separation of the inorganic salt byproducts, enhancing the overall robustness of the catalytic cycle.
How to Synthesize Pyrazolone Derivatives Efficiently
The operational execution of this synthesis requires strict adherence to the defined pH parameters to replicate the high yields reported in the patent data. The process begins with the preparation of a biphasic or homogeneous mixture depending on the solvent choice, followed by the sequential addition of reagents guided by pH meter readings rather than simple stoichiometric timing. This shift from time-based to condition-based addition is a hallmark of modern process chemistry, ensuring consistency regardless of minor variations in raw material quality. The following guide outlines the standardized procedure derived from the patent examples, serving as a foundational protocol for process engineers.
- Step A: Mix water, organic solvent, pyrazolone compound (1), and base to adjust the aqueous phase pH between 11.5 and 12.5.
- Step B: Add acid halide (2) to the mixture until the aqueous phase pH drops to 11.5 or below.
- Step C: Continue adding acid halide (2) and base simultaneously to maintain the aqueous phase pH strictly between 10.0 and 11.5.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this pH-controlled synthesis route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the substantial reduction of waste and the optimization of raw material utilization. By suppressing the formation of di-acylated byproducts, the process inherently increases the effective yield of the desired intermediate without requiring expensive additional purification steps. This efficiency translates directly into lower cost of goods sold (COGS) and a reduced environmental footprint, aligning with increasingly stringent global regulations on chemical manufacturing emissions. Furthermore, the use of common solvents like xylene and methanol, combined with aqueous base, ensures that the supply chain for raw materials remains robust and resistant to market volatility.
- Cost Reduction in Manufacturing: The elimination of complex purification sequences required to remove di-acyl impurities significantly lowers operational expenditures. Traditional methods often necessitate energy-intensive recrystallizations or chromatographic separations to meet purity specs, whereas this novel route delivers a cleaner crude product. The qualitative improvement in selectivity means that less starting material is wasted on side products, maximizing the throughput of the reactor vessels. Additionally, the mild reaction temperatures (0-50°C) reduce energy consumption for heating and cooling, contributing to a more sustainable and cost-effective production profile.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as sodium hydroxide, hydrochloric acid, and standard organic solvents mitigates the risk of supply disruptions associated with exotic reagents. The process is designed to be tolerant of slight variations in feedstock, provided the pH control logic is maintained, which enhances the resilience of the manufacturing line. For reliable agrochemical intermediate suppliers, this robustness ensures consistent delivery schedules and reduces the likelihood of batch failures that could disrupt downstream pesticide production lines for major agricultural clients.
- Scalability and Environmental Compliance: The water-organic solvent system is inherently safer and easier to scale than purely organic processes, as the aqueous phase acts as a heat sink to manage the exotherm of the acylation reaction. This thermal stability allows for larger batch sizes and continuous processing opportunities, facilitating the commercial scale-up of complex agrochemical intermediates. Moreover, the simplified workup procedure, involving acidification and filtration, generates less hazardous waste solvent compared to methods requiring extensive organic extractions, supporting corporate sustainability goals and regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and claims within CN101815705A, providing clarity for technical teams evaluating the feasibility of adoption. Understanding these nuances is essential for integrating this technology into existing manufacturing frameworks.
Q: How does the new pH-controlled method improve upon conventional acylation processes?
A: Conventional methods often suffer from uncontrolled pH drops leading to di-acylation byproducts. This novel process maintains a specific pH window (10.0-11.5) during the critical reaction phase, significantly suppressing side reactions and improving selectivity.
Q: What are the primary advantages for large-scale agrochemical manufacturing?
A: The process utilizes a water-organic solvent biphasic or homogeneous system which simplifies heat management and workup. The high selectivity reduces the load on purification steps, lowering overall production costs and environmental waste.
Q: Can this synthesis route be adapted for different R-group substitutions?
A: Yes, the patent specifies that R groups on the phenyl ring and the acyl halide can vary (halogens, alkyls, alkenyls), making this a robust platform technology for generating a library of pyrazolone derivatives for various pesticide applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrazolone Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires a partner with deep technical expertise and scalable infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate pH-control mechanisms described in CN101815705A are faithfully reproduced at an industrial level. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify the absence of di-acylated impurities in every batch. Our commitment to quality assurance guarantees that the pyrazolone derivatives supplied meet the exacting standards required for global agrochemical registration and formulation.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific supply chain needs. By leveraging our process optimization capabilities, we can provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this high-selectivity method. We encourage you to request specific COA data and route feasibility assessments to validate the performance of our materials against your current benchmarks, ensuring a seamless integration into your production workflow.
