Advanced Solid-Phase Synthesis of Bremelanotide Acetate for Commercial Scale-Up
Introduction to Advanced Peptide Synthesis Technology
The pharmaceutical landscape for treating sexual dysfunction disorders has been significantly advanced by the development of Bremelanotide, a cyclic heptapeptide analogue of alpha-melanocyte stimulating hormone. However, the commercial viability of such complex peptides has historically been constrained by challenging synthesis routes that suffer from low yields and difficult purification profiles. Patent CN113880921B introduces a transformative methodology that addresses these critical bottlenecks through a refined Solid-Phase Peptide Synthesis (SPPS) strategy. By utilizing a specifically modified RinkAmide resin carrier treated with 4-trifluoromethylmandelic acid, this innovation fundamentally alters the physical chemistry of the reaction matrix. This technical breakthrough not only enhances the mechanical stability of the resin during the rigorous swelling and washing cycles but also optimizes the spatial network for amino acid coupling. For R&D directors and procurement specialists seeking a reliable API intermediate supplier, understanding this patented route is essential for securing a stable supply of high-purity Bremelanotide Acetate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis pathways for cyclic peptides like Bremelanotide have predominantly relied on liquid-phase cyclization or standard solid-phase methods that fail to account for steric constraints. In liquid-phase ring-closing reactions, the probability of intermolecular coupling is dangerously high, leading to the formation of unwanted polymers such as dimers and trimers which drastically reduce the overall synthesis yield. To mitigate these side reactions, manufacturers are often forced to operate at extremely low concentrations, a practice that generates excessive volumes of wastewater and complicates solvent recovery, thereby inflating production costs and environmental liabilities. Furthermore, standard solid-phase cyclization often encounters severe steric hindrance, preventing the peptide chain from folding effectively to bring the reactive termini into proximity. This results in incomplete cyclization, difficult downstream purification, and a final product that struggles to meet the stringent purity specifications required for clinical applications.
The Novel Approach
The patented methodology overcomes these historical deficiencies by engineering the solid support itself to facilitate the reaction. By modifying the RinkAmide resin with 4-trifluoromethylmandelic acid prior to peptide assembly, the process introduces bulky aromatic groups that increase the resin's affinity for organic solvents and expand its internal network structure. This expansion is critical as it allows reactants to penetrate deeper into the resin matrix, increasing the collision frequency of reactive groups and accelerating condensation rates. The result is a linear peptide resin that is primed for efficient on-resin cyclization, minimizing the formation of oligomeric by-products. This approach streamlines the entire workflow from C-terminal to N-terminal assembly, ensuring that the subsequent cleavage and purification steps begin with a crude product of significantly higher quality. 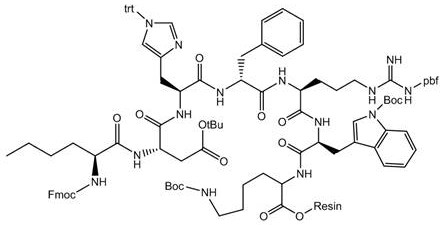
Mechanistic Insights into Resin Modification and Purification Dynamics
The core mechanistic advantage of this synthesis lies in the physicochemical modification of the solid support. The introduction of 4-trifluoromethylmandelic acid creates a more hydrophobic and sterically open environment within the resin beads. Experimental data indicates that this modification significantly increases the swelling degree of the resin in dichloromethane, expanding from approximately 5.8 mL/g for unmodified resin to much higher values depending on the modification ratio. This expanded network reduces the diffusion barrier for coupling reagents like HBTU and HOAT, ensuring that even sterically hindered amino acids such as N-methylated or bulky aromatic residues couple efficiently. Consequently, the deletion sequences that typically plague long peptide syntheses are minimized, leading to a crude peptide profile that is far easier to purify. The structural integrity of the growing peptide chain is maintained throughout the iterative deprotection and coupling cycles, preserving the stereochemistry essential for biological activity.
Following the on-resin cyclization and cleavage, the purification strategy employs a sophisticated dual-chromatography system designed to remove trace impurities and convert the peptide into its acetate salt form. The process begins with Preparative High-Performance Liquid Chromatography (HPLC) using octadecyl silica gel, which effectively separates the target peptide from most hydrophobic impurities and deletion sequences. Crucially, the second stage utilizes Anion Exchange Chromatography with a specialized mobile phase containing acetic acid, triisopropanolamine, and dimethylglyoxime. These additives create a synergistic effect that modulates the ionic strength and interaction between the peptide and the stationary phase. This unique mobile phase composition enhances separation selectivity, allowing for the precise removal of closely related impurities while simultaneously facilitating the salt exchange to the acetate form. 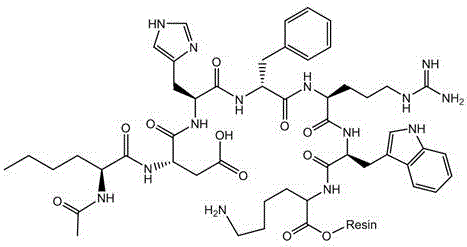
How to Synthesize Bremelanotide Acetate Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing clinical-grade Bremelanotide Acetate. The process initiates with the careful preparation of the modified resin, followed by the sequential coupling of Fmoc-protected amino acids in the order of Lys, Trp, Arg, D-Phe, His, Asp, and Nle. Each coupling step is optimized with specific reagents like DIC and HOBt to ensure high efficiency. Once the linear sequence is assembled and the N-terminus is acetylated, the side-chain protecting groups are removed, and the peptide is cyclized directly on the solid support. The final steps involve cleavage from the resin, alkaline hydrolysis of the ester linkage, and the aforementioned dual-column purification. For detailed operational parameters including flow rates, gradients, and reagent ratios, please refer to the standardized synthesis guide below.
- Modify RinkAmide resin with 4-trifluoromethylmandelic acid to enhance swelling and mechanical strength.
- Perform Fmoc-SPPS from C-terminal to N-terminal to assemble the linear heptapeptide sequence.
- Execute on-resin cyclization followed by cleavage, hydrolysis, and dual-column purification (HPLC + Anion Exchange).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers substantial strategic advantages beyond mere technical elegance. The primary benefit is the drastic simplification of the purification train, which directly translates to cost reduction in pharmaceutical manufacturing. By achieving a crude peptide purity of over 89% and a final purity exceeding 99.8%, the process minimizes the loss of valuable material during downstream processing. Traditional methods often suffer from significant yield erosion during purification due to the co-elution of impurities, but the enhanced selectivity of this anion exchange method preserves the bulk of the synthesized product. This efficiency means that less raw material is required to produce the same amount of finished API, effectively lowering the cost of goods sold (COGS) without compromising on quality standards.
- Cost Reduction in Manufacturing: The elimination of complex liquid-phase cyclization steps and the reduction in solvent consumption due to higher reaction concentrations significantly lower operational expenses. The modified resin's durability also extends its service life, reducing the frequency of resin replacement and associated waste disposal costs. Furthermore, the high yield of the cyclization step reduces the burden on the purification team, allowing for faster batch turnover and better utilization of chromatography columns.
- Enhanced Supply Chain Reliability: The robustness of the solid-phase method ensures consistent batch-to-batch quality, which is critical for maintaining regulatory compliance and avoiding production delays. The use of commercially available Fmoc-amino acids and standard coupling reagents mitigates the risk of raw material shortages. Additionally, the scalability of the process from laboratory to commercial production is well-documented, providing confidence that supply volumes can be ramped up to meet market demand without the need for extensive process re-validation.
- Scalability and Environmental Compliance: By avoiding the dilute conditions required for liquid-phase cyclization, this method significantly reduces the volume of organic waste generated per kilogram of product. The streamlined workflow also shortens the overall production cycle time, enhancing the agility of the supply chain. The ability to produce high-purity material with fewer processing steps aligns with green chemistry principles, reducing the environmental footprint of the manufacturing facility and simplifying regulatory reporting regarding solvent emissions and waste treatment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Bremelanotide Acetate using this advanced SPPS methodology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the technology's capabilities. Understanding these details is vital for partners evaluating the feasibility of integrating this intermediate into their own drug development pipelines.
Q: How does the modified resin improve synthesis yield?
A: The 4-trifluoromethylmandelic acid modification increases the resin's swelling degree and expands its network structure, allowing better reactant access and reducing steric hindrance during cyclization.
Q: What purification method ensures >99% purity?
A: A combination of Preparative HPLC followed by Anion Exchange Chromatography using a mobile phase containing triisopropanolamine and dimethylglyoxime achieves superior separation selectivity.
Q: Is this process suitable for large-scale production?
A: Yes, the solid-phase approach minimizes intermolecular polymerization side reactions common in liquid-phase cyclization, making it highly scalable with consistent quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bremelanotide Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which are equipped with state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our expertise in peptide chemistry allows us to navigate the complexities of cyclic peptide synthesis, offering solutions that optimize both yield and cost-efficiency for our global partners.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your time to market while ensuring the highest levels of product quality and supply security.
