Advanced Synthesis of Adamantane Carboxamide Derivatives for Commercial Pharmaceutical Applications
The pharmaceutical landscape is constantly evolving with the demand for novel scaffolds that offer improved pharmacokinetic profiles and target specificity. Patent CN110790677B introduces a significant advancement in this domain by disclosing a series of N-(3-halogenated-4-substituted phenyl-2-azetidinone-1-yl) adamantane carboxamide compounds. These molecules represent a strategic fusion of three biologically active substructures: the lipophilic adamantane cage, the reactive 2-azetidinone ring, and the versatile hydrazide skeleton. This unique combination is designed to enhance membrane permeability and metabolic stability, addressing common challenges in drug development. The patent highlights a streamlined preparation method that operates under mild conditions, specifically utilizing an ice bath to control the exothermic nature of the acylation reaction. This approach not only simplifies the synthetic workflow but also ensures the integrity of the sensitive beta-lactam ring, which is crucial for maintaining biological activity.
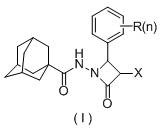
For R&D directors evaluating new chemical entities, the structural versatility offered by this patent is particularly compelling. The general formula allows for extensive modification at the phenyl ring, enabling the fine-tuning of electronic and steric properties to optimize binding affinity against Topoisomerase I. The inclusion of halogen atoms and various alkyl or alkoxy groups provides a robust platform for structure-activity relationship (SAR) studies. Furthermore, the inherent lipophilicity of the adamantane moiety is expected to improve the ADME (Absorption, Distribution, Metabolism, and Excretion) properties of the final drug candidates. By integrating these distinct pharmacophores into a single molecular entity, the invention opens new avenues for developing potent antitumor agents with potentially reduced toxicity profiles compared to existing therapies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis pathways for complex heterocyclic compounds often involve multiple protection and deprotection steps, leading to decreased overall yields and increased waste generation. Conventional methods for constructing the 2-azetidinone ring frequently require harsh reaction conditions, such as high temperatures or strong bases, which can compromise the stability of sensitive functional groups attached to the scaffold. Additionally, many prior art processes rely on expensive transition metal catalysts or specialized reagents that are difficult to source in bulk quantities, creating bottlenecks in the supply chain. The purification of intermediates in these multi-step sequences often necessitates column chromatography, a technique that is notoriously difficult to scale up for industrial manufacturing due to solvent consumption and throughput limitations. These factors collectively contribute to higher production costs and longer lead times, making it challenging to bring new drug candidates to clinical trials efficiently.
The Novel Approach
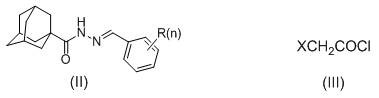
In stark contrast, the methodology described in patent CN110790677B offers a remarkably efficient route that bypasses many of these traditional hurdles. The core innovation lies in the direct reaction between 1-adamantanecarboxyl hydrazone and haloacetyl chloride, which proceeds smoothly in polar aprotic solvents like N,N-dimethylformamide. This one-pot transformation effectively constructs the target azetidinone ring while simultaneously installing the adamantane carboxamide group, significantly reducing the number of unit operations required. The reaction is conducted under ice bath conditions, typically ranging from 5 to 20 hours, which minimizes thermal degradation and side reactions. Workup is equally straightforward, involving the addition of ice water to precipitate the crude product, followed by simple filtration and recrystallization. This eliminates the need for complex extraction protocols or chromatographic purification, thereby enhancing the overall process mass intensity (PMI).
Mechanistic Insights into Acylation and Cyclization
The chemical mechanism underpinning this synthesis involves a sequential nucleophilic acyl substitution followed by an intramolecular cyclization. Initially, the nucleophilic nitrogen of the 1-adamantanecarboxyl hydrazone attacks the electrophilic carbonyl carbon of the haloacetyl chloride. This step forms an unstable acylhydrazine intermediate, releasing hydrogen chloride as a byproduct. The presence of the base or the inherent basicity of the reaction medium helps to neutralize the acid, driving the equilibrium forward. Subsequently, the remaining nitrogen atom within the hydrazine moiety performs a nucleophilic attack on the alpha-carbon bearing the halogen atom. This intramolecular displacement results in the closure of the four-membered azetidinone ring, expelling the halide ion. The rigidity of the adamantane group likely plays a role in pre-organizing the molecule or stabilizing the transition state, facilitating the formation of the strained beta-lactam ring under such mild thermal conditions.
From an impurity control perspective, the simplicity of this mechanism offers distinct advantages. Since the reaction avoids the use of heavy metal catalysts, the risk of metal contamination in the final API intermediate is virtually eliminated, a critical consideration for regulatory compliance. The primary impurities are likely to be unreacted starting materials or hydrolysis products, both of which are effectively removed during the recrystallization step using solvents like ethanol or ethyl acetate. The high selectivity of the cyclization step ensures that the desired regioisomer is formed predominantly, minimizing the formation of structural analogs that could complicate downstream purification. This clean reaction profile translates directly into higher purity specifications for the final product, reducing the burden on quality control laboratories and ensuring consistent batch-to-batch reproducibility essential for GMP manufacturing environments.
How to Synthesize N-(3-halo-4-substituted phenyl-2-azetidinone-1-yl) adamantane carboxamides Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing these high-value intermediates with excellent yields. The process begins by dissolving equimolar amounts of the hydrazone derivative and the haloacetyl chloride in a suitable organic solvent, with N,N-dimethylformamide being the preferred choice due to its ability to dissolve both reactants effectively. The mixture is then cooled to an ice bath temperature to manage the exotherm and maintain selectivity. Reaction progress is monitored using thin-layer chromatography (TLC) until the starting material is fully consumed, typically requiring between 8 to 14 hours depending on the specific substituents. Upon completion, the reaction mixture is poured into a large volume of ice water, causing the product to precipitate as a solid. This crude solid is collected via suction filtration, washed thoroughly with water to remove soluble salts and residual solvent, and finally purified by recrystallization to achieve the desired pharmaceutical grade purity.
- Dissolve 1-adamantanecarboxyl hydrazone and haloacetyl chloride in an organic solvent such as N,N-dimethylformamide.
- Maintain the reaction mixture under ice bath conditions for 5 to 20 hours while monitoring progress via TLC.
- Quench the reaction with ice water to precipitate the solid product, followed by filtration, washing, and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of this synthetic route are substantial. The elimination of expensive catalysts and the reliance on commodity chemicals like haloacetyl chlorides and DMF significantly lowers the raw material costs associated with production. Furthermore, the simplified workup procedure reduces the consumption of auxiliary solvents and energy, contributing to a greener and more cost-effective manufacturing process. The high yields reported across various derivatives, often exceeding 80%, ensure that material throughput is maximized, reducing the cost per kilogram of the final intermediate. This efficiency is crucial for maintaining competitive pricing in the global market for pharmaceutical intermediates, allowing companies to allocate resources to other critical areas of drug development.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by removing the need for precious metal catalysts and complex purification steps like column chromatography. The use of inexpensive solvents and the ability to isolate the product via simple precipitation drastically cuts down on operational expenses. Additionally, the high atom economy of the reaction means less waste is generated, lowering disposal costs and environmental fees. These factors combine to create a highly economical production model that supports margin improvement without compromising on quality standards.
- Enhanced Supply Chain Reliability: Sourcing reliability is greatly improved as all key reagents, including adamantane derivatives and haloacetyl chlorides, are readily available from multiple global suppliers. The robustness of the reaction conditions means that production is less susceptible to variations in raw material quality or minor fluctuations in processing parameters. This stability ensures consistent output and reduces the risk of batch failures that can disrupt supply schedules. Consequently, manufacturers can maintain tighter delivery windows and build stronger partnerships with downstream API producers who depend on timely intermediate availability.
- Scalability and Environmental Compliance: The scalability of this method is evident in its straightforward engineering requirements, which do not demand specialized high-pressure or high-temperature equipment. The mild ice bath conditions are easily replicable in large-scale reactors, facilitating a smooth transition from laboratory benchtop to pilot plant and full commercial production. Moreover, the reduced solvent usage and absence of toxic heavy metals align well with increasingly stringent environmental regulations. This compliance minimizes the regulatory burden and facilitates faster approval processes for new manufacturing sites, ensuring long-term sustainability of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these adamantane carboxamide derivatives. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation. Understanding these details is essential for stakeholders looking to integrate this technology into their existing pipelines or evaluate its potential for new drug discovery programs. The clarity provided here aims to bridge the gap between academic research and industrial application.
Q: What is the primary biological activity of these adamantane carboxamide compounds?
A: These compounds exhibit significant Topoisomerase I (Top1) inhibitory activity, making them valuable leads for anticancer drug development.
Q: How does this synthesis method improve upon conventional routes?
A: The process utilizes a direct one-pot reaction under mild ice bath conditions, eliminating the need for complex multi-step sequences or expensive transition metal catalysts.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the use of common solvents like DMF and simple workup procedures involving ice water precipitation ensures high scalability and operational safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Adamantane Carboxamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in patent CN110790677B for the development of next-generation anticancer therapeutics. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative chemistry into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can grow seamlessly from early-stage development to full-scale manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of high-purity pharmaceutical intermediates meets the exacting standards required by global regulatory bodies.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific drug development needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and timeline. By partnering with us, you gain access to specific COA data and comprehensive route feasibility assessments that will accelerate your time to market. Let us help you navigate the complexities of chemical manufacturing so you can focus on delivering life-saving medicines to patients worldwide.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
