Advanced Solid-Phase Synthesis of C-Terminal Modified Peptides for Commercial Scale
The pharmaceutical industry is constantly seeking robust methodologies to enhance the efficiency of peptide drug development, particularly for complex conjugates. Patent CN106928313B introduces a transformative approach to the synthesis of C-terminal modified peptides, addressing long-standing bottlenecks in traditional manufacturing. This technology shifts the critical coupling of diamino linkers from the liquid phase to the solid phase, fundamentally altering the process economics and technical feasibility. By anchoring the diamino compound directly to the solid-phase synthetic resin at the outset, the method eliminates the need for difficult solution-phase amide bond formations that typically plague C-terminal modifications. This strategic pivot not only simplifies the operational workflow but also drastically improves the overall yield and purity profile of the final active pharmaceutical ingredient. For R&D teams evaluating process scalability, this patent offers a compelling alternative to legacy techniques, promising a more streamlined path from laboratory bench to commercial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of C-terminal modified peptides often relies on completing the peptide sequence on a resin, cleaving it, and then attempting to couple a diamino linker in the liquid phase. As illustrated in the prior art flow, this approach introduces significant complexity because the amide bond reaction must be performed twice in solution, greatly increasing post-treatment difficulties. The liquid-phase coupling of bifunctional linkers like hexamethylenediamine is notoriously inefficient, often leading to polymerization, incomplete reactions, and challenging purification scenarios. Furthermore, performing these modifications in solution requires large volumes of solvents and extensive workup procedures to remove excess reagents and byproducts, which drives up both cost and environmental impact. The cumulative effect of these inefficiencies is a substantial reduction in the yield of the target peptide, making the process economically unviable for large-scale manufacturing of high-value peptide drugs.
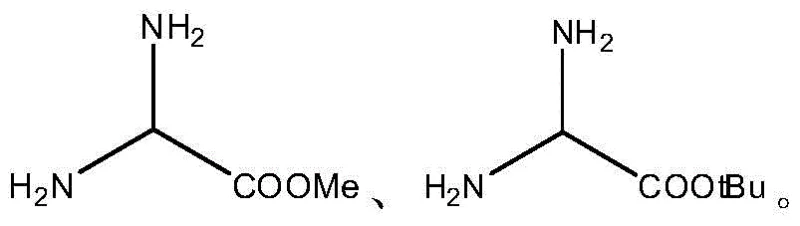
The Novel Approach
In stark contrast, the novel methodology described in the patent reverses the synthesis order by first coupling one end of the diamino compound to the solid-phase carrier. This allows the entire peptide chain to be assembled on the free amino group of the linker while it is still anchored to the resin, utilizing standard Fmoc solid-phase peptide synthesis strategies. By keeping the difficult linker coupling step on the solid support, the process leverages the advantages of pseudo-dilution and easy filtration to drive reactions to completion. The result is a fully protected polypeptide resin that can be cleaved and modified with much higher efficiency. This approach effectively solves the technical barrier of uniformly coupling bifunctional compounds to the solid phase, ensuring consistent loading and minimizing side reactions. The transition from liquid-phase linker attachment to solid-phase initialization represents a paradigm shift that enhances both the chemical robustness and the commercial viability of producing C-terminal modified peptides.
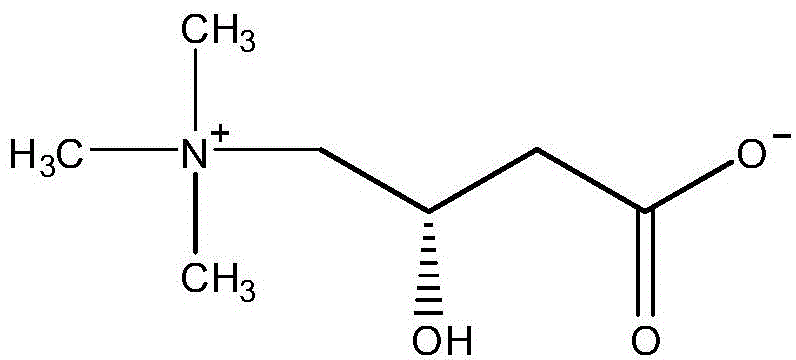
Mechanistic Insights into Solid-Phase Diamino Coupling
The core innovation lies in the precise control of the coupling between the diamino compound and the solid-phase carrier, specifically using resins like 2-Chlorotrityl Chloride Resin. The patent details a two-step blocking method employed during the coupling of the diamino compound and the first amino acid residue to prevent the generation of byproducts. After the initial attachment of the diamino linker to the resin, any unreacted chlorotrityl groups are capped, and crucially, the free amino group of the linker is protected or managed to ensure that subsequent amino acid coupling occurs only at the desired site. This meticulous control prevents the bifunctional diamino compound from acting as a cross-linker between resin beads, which would otherwise lead to insoluble aggregates and loss of material. The use of specific coupling systems, such as DIC/HOBt or HBTU/DIPEA, further optimizes the activation of carboxyl groups, ensuring high substitution degrees ranging from 0.1 to 1.2 mmol/g. This level of mechanistic precision is essential for maintaining the integrity of the growing peptide chain and ensuring that the final cleavage yields a homogeneous product.
Impurity control is another critical aspect where this mechanism excels, particularly when dealing with complex modifying groups like Biotin or L-Carnitine. In traditional liquid-phase synthesis, the reaction between the C-terminal carboxyl of the peptide and the diamino linker often produces racemization or deletion sequences that are difficult to separate. By performing the chain elongation on the solid phase, the washing steps between each coupling cycle effectively remove excess reagents and soluble impurities before they can react further. The cleavage step utilizes a optimized mixture of TFA, TIS, EDT, and water, typically in a ratio of 85-95:2-5:2-5:1-5, which efficiently removes acid-labile protecting groups without degrading the sensitive peptide backbone. This controlled deprotection environment minimizes the formation of side products, resulting in a crude peptide with significantly higher purity, thereby reducing the load on downstream purification processes like preparative HPLC.
How to Synthesize C-Terminal Modified Peptide Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this technology in a GMP environment. The process begins with the swelling of the resin in DMF, followed by the controlled addition of the diamino compound and a base like DIPEA. A critical quality control point is the capping step using methanol and DIPEA to seal unreacted sites, ensuring that the subsequent peptide chain grows from a uniform starting point. Following the assembly of the peptide sequence, which can range from 1 to 20 amino acids, the resin is treated with a cleavage cocktail to release the fully protected intermediate. This structured approach ensures reproducibility and scalability, making it suitable for the production of clinical trial materials and commercial batches alike.
- Couple one end of a diamino compound (e.g., ethylenediamine, hexamethylenediamine) to a solid-phase synthetic resin such as 2-Chlorotrityl Chloride Resin.
- Employ Fmoc solid-phase peptide synthesis strategy to sequentially couple amino acids to the free amino group of the diamino linker on the resin.
- Cleave the fully protected polypeptide from the resin using a TFA-based cleavage reagent mixture.
- Remove protecting groups or couple with a carboxyl-containing modifying group (e.g., Biotin, L-Carnitine) to obtain the target C-terminal modified peptide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this solid-phase synthesis route offers tangible benefits in terms of cost structure and supply reliability. The primary advantage stems from the drastic simplification of the manufacturing process, which eliminates the need for multiple isolated liquid-phase coupling steps that are resource-intensive and time-consuming. By consolidating the synthesis onto a solid support, the consumption of organic solvents is significantly reduced, and the number of unit operations required for isolation and purification is minimized. This streamlining translates directly into lower operational expenditures and a reduced carbon footprint, aligning with modern green chemistry initiatives. Furthermore, the improved yield means that less starting material is required to produce the same amount of final API, effectively lowering the cost of goods sold (COGS) for high-value peptide therapeutics.
- Cost Reduction in Manufacturing: The elimination of complex liquid-phase linker coupling steps removes the need for expensive purification interventions typically required to separate linker byproducts. Since the solid-phase washing steps handle impurity removal continuously during synthesis, the burden on final chromatographic purification is substantially lightened. This reduction in downstream processing requirements leads to significant cost savings in terms of chromatography resin usage, solvent disposal, and labor hours. Additionally, the higher overall yield reported in the patent examples implies that less raw material is wasted, further enhancing the economic efficiency of the production line.
- Enhanced Supply Chain Reliability: The reagents required for this method, such as standard Fmoc-amino acids, 2-Chlorotrityl Chloride Resin, and common diamino alkanes, are widely available from established chemical suppliers. This reliance on commodity chemicals reduces the risk of supply chain disruptions associated with bespoke or hard-to-source reagents. The robustness of the solid-phase protocol also means that batch-to-batch variability is minimized, ensuring a consistent supply of high-quality intermediates. For supply chain planners, this predictability allows for more accurate forecasting and inventory management, reducing the need for safety stock and mitigating the risk of production delays.
- Scalability and Environmental Compliance: Scaling solid-phase peptide synthesis is a well-understood engineering challenge, and this method fits seamlessly into existing large-scale SPPS reactors. The reduction in solvent volume per kilogram of product simplifies waste treatment protocols, making it easier to comply with stringent environmental regulations. The process avoids the use of hazardous heavy metal catalysts often found in alternative coupling methods, thereby simplifying the regulatory filing process regarding residual impurities. This environmental and regulatory ease facilitates faster technology transfer from pilot plant to commercial manufacturing, accelerating time-to-market for new peptide drugs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this C-terminal modification technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is critical for R&D directors and procurement specialists evaluating the feasibility of adopting this synthesis route for their specific pipeline candidates.
Q: How does this solid-phase method improve yield compared to traditional liquid-phase coupling?
A: The patent data demonstrates that shifting the diamino linker coupling to the solid phase avoids difficult liquid-phase amide bond reactions. Experimental results show a total yield increase of approximately 3-fold for complex peptides like Epristeride compared to prior art methods.
Q: What types of diamino linkers are compatible with this synthesis strategy?
A: The method supports linear symmetrical diamino compounds such as ethylenediamine, propylenediamine, butylenediamine, hexylenediamine, and octylenediamine, allowing flexibility in spacer length for different drug conjugates.
Q: Does this process require specialized purification equipment?
A: While standard RP-HPLC is used for final polishing, the improved crude purity resulting from the solid-phase strategy reduces the burden on purification systems, making commercial scale-up more feasible with standard industrial chromatography setups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable C-Terminal Modified Peptide Supplier
NINGBO INNO PHARMCHEM stands at the forefront of peptide manufacturing, possessing the technical expertise to translate complex patent methodologies like CN106928313B into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of solid-phase linker coupling are realized in actual manufacturing outputs. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instruments to verify the identity and quality of every batch. Our commitment to technical excellence ensures that we can handle the nuances of diamino linker loading and Fmoc SPPS optimization required to deliver high-purity C-terminal modified peptides consistently.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic benefits specific to your target molecule. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this efficient manufacturing technology into your portfolio. Let us help you navigate the complexities of peptide synthesis and secure a reliable, cost-effective supply of your critical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
