Advanced Phosphitylation Technology for High-Purity Oligonucleotide Intermediates and Commercial Scale-Up
Introduction to Next-Generation Phosphitylation Technology
The landscape of oligonucleotide therapeutic manufacturing is undergoing a significant transformation, driven by the urgent need for safer, more scalable, and cost-effective synthetic routes. Patent CN1646452A introduces a groundbreaking process for preparing phosphitylated compounds, specifically 3'-O-phosphoramidites, which are critical building blocks for antisense drugs and siRNA therapeutics. This technology fundamentally shifts the paradigm away from traditional, hazardous activators like 1H-tetrazole towards novel acid-base complexes derived from sterically hindered amine bases. By leveraging activators such as Hünig's base (diisopropylethylamine) or collidine complexed with acids like trifluoroacetic acid (TFA), the process mitigates the explosion risks associated with nitrogen-rich heterocycles while simultaneously addressing the environmental burden of toxic, water-soluble pyridine salts. For R&D directors and process chemists, this represents a vital opportunity to enhance the safety profile of their manufacturing pipelines without compromising on the rigorous purity standards required for pharmaceutical intermediates.
Furthermore, the commercial implications of adopting this methodology extend far beyond mere safety compliance. The ability to utilize readily available, non-hazardous amine bases significantly simplifies the supply chain logistics for raw materials, reducing the dependency on specialized, high-cost reagents that often face availability bottlenecks. The patent data demonstrates that these hindered activators not only match but frequently exceed the yields of conventional methods, achieving conversion rates upwards of 90% in many embodiments. This efficiency gain is paramount for procurement managers seeking to optimize the cost of goods sold (COGS) for high-volume oligonucleotide production. As the demand for nucleic acid-based medicines continues to surge, establishing a robust, reliable pharmaceutical intermediate supplier network that utilizes such advanced, risk-mitigated chemistry becomes a strategic imperative for maintaining competitive advantage in the global biopharmaceutical market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nucleoside phosphoramidites has relied heavily on activators such as 1H-tetrazole, which, despite its efficacy, poses severe safety challenges due to its explosive nature. Handling tetrazole on a commercial scale requires extensive safety infrastructure, including blast-proof facilities and rigorous operational protocols, which inevitably drives up capital expenditure and operational costs. Moreover, alternative activators derived from unsubstituted pyridines, while less explosive, generate salts that are highly toxic and extremely water-soluble. This solubility characteristic creates a significant environmental liability, necessitating the installation of sophisticated and expensive wastewater treatment systems to prevent ecological contamination. These conventional pathways also often suffer from side reactions, such as the alkylation of lactam units in guanosine derivatives, which can compromise the purity of the final product and complicate downstream purification efforts. Consequently, the industry has long sought a viable alternative that balances reactivity with safety and environmental stewardship.
The Novel Approach
The innovative strategy outlined in CN1646452A circumvents these historical drawbacks by employing acid-base complexes formed from sterically hindered amines. Unlike the small, highly nucleophilic tetrazole anion, these bulky amine complexes exhibit reduced nucleophilicity, which paradoxically leads to fewer side reactions with sensitive functional groups on the nucleoside substrate. 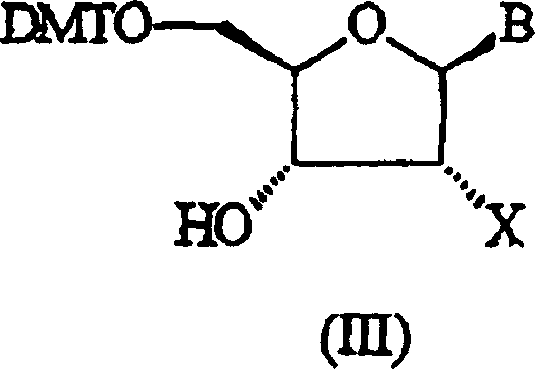 As illustrated by the general structure of the hydroxyl-containing compounds, the process is compatible with a wide array of protecting groups, including DMTr, TBDMS, and MOE, ensuring versatility across different oligonucleotide sequences. The use of solvents like methyl acetate or toluene, coupled with precipitation purification techniques using petroleum ether, further eliminates the need for chlorinated solvents like dichloromethane, aligning the process with green chemistry principles. This novel approach not only enhances the safety and sustainability of cost reduction in pharmaceutical intermediates manufacturing but also streamlines the isolation of high-purity products, making it an ideal candidate for large-scale industrial adoption.
As illustrated by the general structure of the hydroxyl-containing compounds, the process is compatible with a wide array of protecting groups, including DMTr, TBDMS, and MOE, ensuring versatility across different oligonucleotide sequences. The use of solvents like methyl acetate or toluene, coupled with precipitation purification techniques using petroleum ether, further eliminates the need for chlorinated solvents like dichloromethane, aligning the process with green chemistry principles. This novel approach not only enhances the safety and sustainability of cost reduction in pharmaceutical intermediates manufacturing but also streamlines the isolation of high-purity products, making it an ideal candidate for large-scale industrial adoption.
Mechanistic Insights into Acid-Base Complex Catalyzed Phosphitylation
The core of this technological advancement lies in the unique activation mechanism facilitated by the hindered amine acid-base complexes. Traditionally, it was hypothesized that less hindered, more nucleophilic activators would be superior for displacing the leaving group on the phosphitylating reagent. However, the patent data reveals a counter-intuitive finding: activators with significant steric bulk, such as those derived from Formula I and Formula II amines, achieve yields comparable to or higher than their less hindered counterparts. 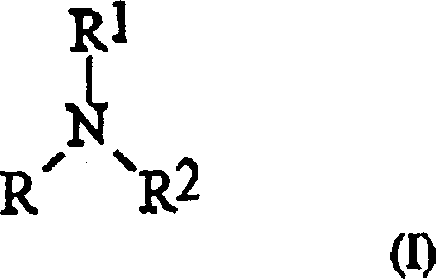
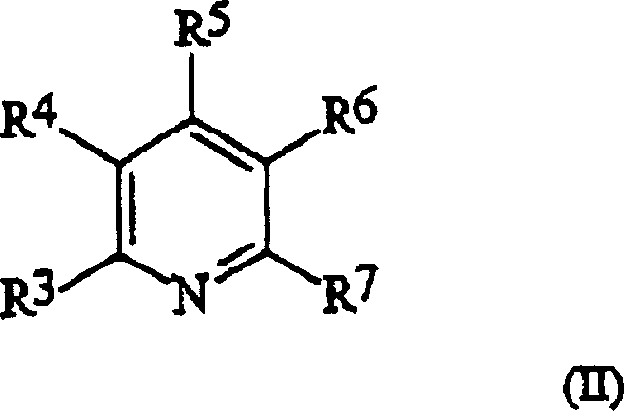 This phenomenon is attributed to the suppression of undesirable side reactions; the steric bulk prevents the activator from attacking electrophilic sites on the nucleoside base, such as the exocyclic amines or lactam carbonyls, which are common sites for degradation in conventional tetrazole-mediated reactions. The acid component, typically trifluoroacetic acid, protonates the amine to form a stable salt that effectively activates the phosphorus center without introducing the toxicity associated with heavy metal catalysts or the instability of tetrazoles.
This phenomenon is attributed to the suppression of undesirable side reactions; the steric bulk prevents the activator from attacking electrophilic sites on the nucleoside base, such as the exocyclic amines or lactam carbonyls, which are common sites for degradation in conventional tetrazole-mediated reactions. The acid component, typically trifluoroacetic acid, protonates the amine to form a stable salt that effectively activates the phosphorus center without introducing the toxicity associated with heavy metal catalysts or the instability of tetrazoles.
From an impurity control perspective, this mechanism offers distinct advantages for producing high-purity oligonucleotide intermediates. The reduced nucleophilicity of the hindered amine minimizes the formation of phosphite triesters and other phosphorylated byproducts that are difficult to separate. Furthermore, the lower water solubility of the resulting amine salts simplifies the aqueous workup process, allowing for cleaner phase separation and reducing the load on downstream purification columns. The reaction conditions, typically conducted at mild temperatures ranging from 0°C to 40°C, further preserve the integrity of acid-labile protecting groups like the dimethoxytrityl (DMTr) group. This precise control over the reaction environment ensures that the final phosphoramidite product meets the stringent quality specifications required for automated DNA synthesis, thereby reducing the failure rate of coupling cycles in the final oligonucleotide assembly and enhancing the overall efficiency of the therapeutic manufacturing process.
How to Synthesize 3'-O-Phosphoramidites Efficiently
Implementing this phosphitylation protocol requires careful attention to the preparation of the activator complex and the control of reaction stoichiometry to maximize yield and purity. The process begins with the in situ or pre-formed generation of the acid-base activator, typically using a molar ratio of roughly 1:1 between the hindered amine and the acid, although slight excesses of the base (e.g., 1.3 equivalents) can be beneficial. The hydroxyl-containing nucleoside is dissolved in an anhydrous aprotic solvent, and moisture is rigorously excluded to prevent hydrolysis of the sensitive phosphoramidite bond. Following the addition of the phosphitylating reagent, the activator solution is introduced under vigorous stirring to ensure homogeneous mixing and efficient activation. The reaction is allowed to proceed for approximately 12 hours at ambient temperature, after which the product can be isolated via a straightforward precipitation technique. For a comprehensive understanding of the specific operational parameters and safety guidelines, the detailed standardized synthesis steps are provided in the guide below.
- Prepare the activator solution by mixing a hindered amine base (e.g., diisopropylethylamine) with an acid (e.g., trifluoroacetic acid) in a suitable solvent like THF or methyl acetate at room temperature.
- Dissolve the 5'-O-protected nucleoside in an anhydrous solvent, remove a portion of the solvent under reduced pressure, and add the phosphitylating reagent such as 2-cyanoethyl-N,N,N',N'-tetraisopropylphosphorodiamidite.
- Add the prepared activator solution to the reaction mixture with vigorous stirring, allow the reaction to proceed for approximately 12 hours, and purify the resulting phosphoramidite via precipitation in petroleum ether or hexane.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this hindered amine-based phosphitylation process offers substantial strategic benefits that directly impact the bottom line and operational resilience. The primary advantage lies in the drastic simplification of the raw material supply chain; replacing explosive tetrazole and toxic pyridine salts with common, commercially abundant amines like Hünig's base and collidine reduces sourcing risks and stabilizes pricing. These reagents are widely available from multiple global suppliers, eliminating single-source dependencies that often plague the procurement of specialized fine chemicals. Furthermore, the elimination of hazardous reagents lowers the regulatory burden and insurance costs associated with storing and transporting dangerous goods, contributing to significant overhead savings. The process's compatibility with greener solvents and its ability to utilize precipitation for purification rather than resource-intensive chromatography further drive down operational expenditures, making it a highly attractive option for commercial scale-up of complex pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic impact of switching to this technology is profound, primarily driven by the removal of expensive and hazardous activators. Tetrazole and its derivatives command a premium price due to their specialized synthesis and handling requirements, whereas hindered amines are commodity chemicals produced at massive scales for various industries. Additionally, the avoidance of chlorinated solvents like dichloromethane reduces waste disposal costs, as the effluent streams are less toxic and easier to treat. The high yields reported in the patent, often exceeding 90%, mean that less starting material is wasted, directly improving the atom economy of the process. By minimizing the formation of difficult-to-remove impurities, the need for extensive recycling or reprocessing is diminished, leading to a leaner, more cost-efficient production cycle that enhances overall profitability without sacrificing product quality.
- Enhanced Supply Chain Reliability: Supply chain continuity is critical in the pharmaceutical sector, where delays can halt clinical trials or commercial launches. This new method enhances reliability by utilizing reagents that are not subject to the strict transportation regulations imposed on explosives or highly toxic substances. The stability of the acid-base complexes allows for more flexible logistics, including longer storage times and broader shipping options. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, reducing the rejection rate of incoming batches. This resilience ensures a steady flow of high-purity nucleoside phosphoramidites to downstream synthesis facilities, mitigating the risk of production stoppages and enabling manufacturers to meet tight delivery schedules with greater confidence and consistency.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to multi-ton production often reveals hidden bottlenecks, particularly regarding heat management and waste treatment. This phosphitylation method is inherently scalable because it avoids exothermic hazards associated with tetrazole decomposition and utilizes mild reaction temperatures. The precipitation purification step is particularly advantageous for large-scale operations, as it replaces batch chromatography with continuous filtration, significantly increasing throughput. From an environmental standpoint, the process aligns with increasingly stringent global regulations on volatile organic compounds (VOCs) and hazardous waste. By reducing the toxicity of the waste stream and eliminating the need for specialized incineration of nitrogen-rich explosives, manufacturers can achieve better environmental compliance scores, which is increasingly important for maintaining corporate social responsibility standings and securing contracts with eco-conscious biopharma partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this phosphitylation technology. These answers are derived directly from the experimental data and claims presented in patent CN1646452A, providing a factual basis for decision-making. Understanding the nuances of activator selection, solvent compatibility, and purification strategies is essential for R&D teams evaluating this process for adoption. The insights provided here aim to clarify how this method compares to existing industry standards and highlight the specific operational advantages it offers for the synthesis of oligonucleotide therapeutics.
Q: Why are hindered amine activators preferred over tetrazole for phosphitylation?
A: Conventional activators like 1H-tetrazole are known to be explosive and hazardous, requiring special safety precautions. Additionally, pyridine-based activators often result in toxic, highly water-soluble salts that necessitate expensive wastewater treatment. The patented method uses hindered amine acid-base complexes which are less toxic, less water-soluble, and safer to handle on an industrial scale while maintaining high yields.
Q: What kind of yields can be expected with this novel phosphitylation process?
A: Experimental data from the patent indicates that yields are surprisingly high, often matching or exceeding conventional methods. For instance, reactions using Hünig's base/TFA complexes achieved yields ranging from 83% to 98% across various nucleoside substrates, demonstrating that the increased steric hindrance of the activator does not negatively impact reaction efficiency.
Q: How does this method improve the purification of oligonucleotide intermediates?
A: The process allows for effective purification through precipitation. By dissolving the crude phosphitylated compound in a solvent like toluene or methyl acetate and adding a precipitation solvent such as petroleum ether or hexane, high-purity products can be isolated. This avoids complex chromatographic steps for bulk production and facilitates easier scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nucleoside Phosphoramidites Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of oligonucleotide therapies depends on access to high-quality, consistently supplied intermediates. Our technical team has extensively analyzed the advancements described in CN1646452A and integrated similar safe, high-efficiency phosphitylation protocols into our own manufacturing platforms. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet stringent purity specifications regardless of order volume. Our rigorous QC labs employ state-of-the-art analytical methods, including HPLC and NMR, to verify the identity and purity of every batch, guaranteeing that our nucleoside phosphoramidites perform flawlessly in your automated synthesis cycles. We are committed to being a partner that not only supplies chemicals but also delivers technical value through process optimization and risk mitigation.
We invite you to engage with our technical procurement team to discuss how our capabilities align with your specific project needs. Whether you are in the early stages of process development or ramping up for commercial launch, we can provide a Customized Cost-Saving Analysis tailored to your current supply chain configuration. We encourage you to request specific COA data and route feasibility assessments to verify our capacity to support your long-term goals. By partnering with us, you gain access to a reliable supply of critical intermediates produced with the latest safety and efficiency standards, empowering your organization to accelerate the delivery of life-saving nucleic acid medicines to patients worldwide.
