Optimizing Palbociclib Intermediate II Production via Novel Palladium Catalysis
Optimizing Palbociclib Intermediate II Production via Novel Palladium Catalysis
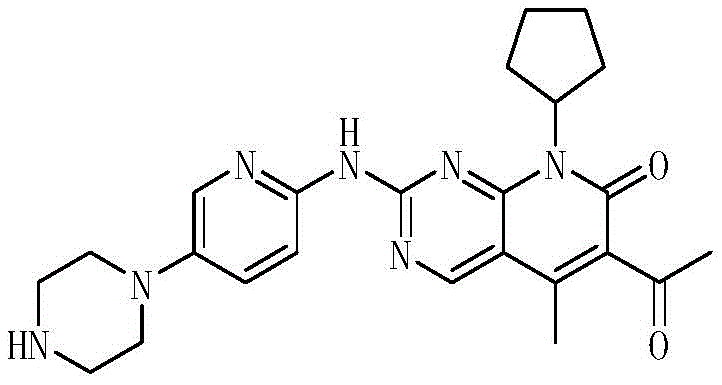
The global demand for CDK4/6 inhibitors continues to surge as breast cancer therapies evolve, placing immense pressure on the supply chains of critical pharmaceutical intermediates. Patent CN108283940B introduces a transformative preparation method for Palbociclib Intermediate II, addressing longstanding bottlenecks in catalytic efficiency and cost structure. This technology leverages a simplified palladium catalytic system comprising palladium acetate or chloride paired with the DPPP ligand, replacing complex and expensive predecessors. For R&D and procurement leaders, this represents a pivotal shift towards more robust and economically viable manufacturing pathways for high-value oncology drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Palbociclib Intermediate II has been plagued by reliance on sophisticated and fragile catalytic systems that hinder industrial scalability. Prior art, such as the methods described in WO 2008032157 and CN105541832A, frequently utilized catalysts like Pd(dppf)2Cl2 or Pd2dba3, which are not only prohibitively expensive but also highly sensitive to moisture and oxygen. These conventional routes often suffered from inconsistent quality control, with yields fluctuating between 60% and 75%, necessitating extensive purification steps that erode profit margins. Furthermore, the use of hazardous additives like DABCO in some legacy processes introduced significant safety risks and environmental compliance burdens, complicating the waste treatment protocols required for large-scale production facilities.
The Novel Approach
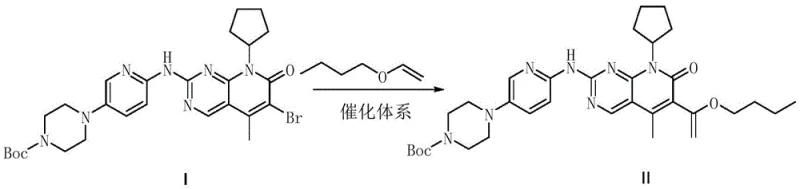
The innovative methodology disclosed in CN108283940B fundamentally reengineers the catalytic landscape by employing a cost-effective palladium source coupled with the DPPP ligand. This new system facilitates a smooth rearrangement reaction between Intermediate I and butyl vinyl ether, achieving superior conversion rates under milder conditions. By shifting away from air-sensitive ferrocene-based palladium complexes to simple palladium salts, the process drastically reduces raw material costs while enhancing operational stability. The reaction proceeds efficiently in common solvents like dioxane or n-butanol, eliminating the need for exotic reagents and streamlining the post-reaction workup to a simple filtration and crystallization sequence.
Mechanistic Insights into Pd/DPPP-Catalyzed Rearrangement
The core of this technological breakthrough lies in the synergistic interaction between the divalent palladium catalyst and the 1,3-bis(diphenylphosphino)propane (DPPP) ligand. Unlike bulkier phosphine ligands that may sterically hinder the catalytic cycle, DPPP provides an optimal bite angle that stabilizes the active palladium species during the vinyl ether insertion and subsequent aromatization steps. The patent data highlights that DPPP possesses a melting point of 63-65°C, which allows it to dissolve and complex effectively with the palladium center at the reaction temperature of 85-95°C. This thermal compatibility ensures a homogeneous catalytic environment, minimizing the formation of palladium black and other inactive species that typically degrade reaction performance in heterogeneous systems.
From an impurity control perspective, the simplicity of the catalytic components translates directly into a cleaner impurity profile for the final intermediate. Traditional methods involving complex ligands often leave behind difficult-to-remove organometallic residues that require rigorous scavenging treatments. In contrast, the DPPP system generates fewer side products, and the use of stable palladium salts reduces the risk of metal leaching variations. The resulting Intermediate II consistently demonstrates HPLC purity greater than 97%, significantly reducing the burden on downstream purification units. This high level of chemical fidelity is crucial for maintaining the strict quality standards required for GMP-grade API production, ensuring that the final drug substance meets regulatory specifications without extensive reprocessing.
How to Synthesize Palbociclib Intermediate II Efficiently
The implementation of this synthesis route is designed for seamless integration into existing pharmaceutical manufacturing infrastructure, requiring only standard glass-lined or stainless steel reactors capable of handling mild heating and inert atmospheres. The process begins with the charging of solvents and reagents under nitrogen protection, followed by a controlled heating phase that drives the rearrangement to completion within a short timeframe. Detailed operational parameters, including precise molar ratios and temperature ramps, are critical for maximizing yield and minimizing batch-to-batch variability. For a comprehensive breakdown of the standardized operating procedures and critical process parameters, please refer to the technical guide below.
- Charge reactor with solvent (dioxane or n-butanol), Intermediate I, butyl vinyl ether, palladium catalyst (acetate or chloride), DPPP ligand, and base under nitrogen.
- Heat the reaction mixture to 85-95°C and maintain for 2-5 hours until HPLC indicates complete conversion of the starting material.
- Perform hot filtration through diatomaceous earth, concentrate filtrate, and crystallize the residue from ethanol to isolate high-purity Intermediate II.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented catalytic system offers tangible strategic benefits that extend beyond mere technical feasibility. The substitution of high-cost ligands with commercially abundant DPPP creates a buffer against volatile precious metal markets, stabilizing the cost of goods sold (COGS) for long-term supply contracts. Additionally, the robustness of the reaction conditions reduces the dependency on specialized handling equipment, allowing for broader sourcing options among contract manufacturing organizations (CMOs) and enhancing overall supply chain resilience against disruptions.
- Cost Reduction in Manufacturing: The economic impact of switching to the DPPP ligand system is profound, primarily driven by the substantial price differential between DPPP and alternative ligands like DPEPhOS. Market analysis indicates that DPPP is significantly more affordable, which directly lowers the variable cost per kilogram of the intermediate produced. Furthermore, the elimination of expensive, sensitive catalysts reduces the capital expenditure required for storage and handling infrastructure, while the simplified workup procedure minimizes solvent consumption and waste disposal fees, contributing to a leaner and more competitive cost structure.
- Enhanced Supply Chain Reliability: Supply continuity is paramount in the pharmaceutical sector, and this method secures the supply chain by utilizing reagents that are widely available from multiple global vendors. Unlike proprietary or complex catalysts that may have single-source bottlenecks, palladium chloride, palladium acetate, and DPPP are commodity chemicals with established production capacities. This diversification of the supplier base mitigates the risk of stockouts and ensures that production schedules can be maintained even during periods of global logistical strain, providing a reliable foundation for meeting clinical and commercial demand.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, avoiding the use of flammable and explosive reagents that often limit batch sizes in traditional synthesis routes. The absence of hazardous additives simplifies the environmental health and safety (EHS) profile of the manufacturing site, reducing the regulatory burden associated with volatile organic compound (VOC) emissions and hazardous waste treatment. This alignment with green chemistry principles not only facilitates faster regulatory approvals but also supports corporate sustainability goals, making the manufacturing process more attractive to environmentally conscious stakeholders and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis pathway. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the operational advantages and quality outcomes associated with this technology.
Q: Why is the DPPP ligand preferred over DPEPhOS in this synthesis?
A: DPPP (1,3-bis(diphenylphosphino)propane) offers a significant cost advantage, being approximately 60% cheaper than DPEPhOS, while possessing a lower melting point (63-65°C) that facilitates efficient complexation with the palladium catalyst at standard reaction temperatures.
Q: What are the purity specifications achievable with this new catalytic system?
A: The patented method consistently achieves HPLC purity levels exceeding 97%, with specific examples demonstrating purity up to 98.6%, ensuring the intermediate meets stringent requirements for downstream API synthesis.
Q: How does this method improve industrial scalability compared to prior art?
A: By eliminating unstable catalysts like Pd2dba3 and flammable additives like DABCO, and utilizing common solvents like dioxane, the process simplifies safety protocols and waste treatment, making it highly suitable for multi-kilogram to ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Palbociclib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of this patent can be fully realized in a GMP-compliant environment. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Palbociclib Intermediate II meets the exacting standards required for oncology drug development.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can enhance your project's economics and timeline. By requesting a Customized Cost-Saving Analysis, you can gain a detailed projection of the potential efficiencies this method offers for your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and accelerate your path to market.
