Advanced Synthesis of Bisthiazol-4-yl Disulfide Derivatives for Scalable Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex heterocyclic scaffolds, particularly those containing disulfide bridges which are prevalent in bioactive molecules. A significant breakthrough in this domain is detailed in Chinese Patent CN111233783A, which discloses a highly efficient synthetic route for bisthiazol-4-yl disulfide derivatives. This innovation addresses long-standing challenges in heterocycle synthesis by utilizing elemental sulfur as a direct sulfur source, thereby bypassing the need for pre-functionalized thiol precursors. The methodology represents a paradigm shift towards greener and more atom-economical processes, leveraging a radical-mediated cyclization strategy that operates under relatively mild thermal conditions. For R&D directors and process chemists, this patent offers a compelling alternative to traditional routes, promising enhanced purity profiles and simplified downstream processing. The ability to construct the disulfide bond concurrently with the thiazole ring formation in a single pot is a notable feat of modern synthetic design, reducing the overall step count and potential for material loss. As we delve deeper into the technical specifics, it becomes evident that this approach not only solves specific chemical problems but also aligns with broader industry goals regarding sustainability and cost-efficiency in the production of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of symmetrical disulfide bonds has relied heavily on the oxidation of thiols, a process that often necessitates the handling of malodorous and unstable mercaptan starting materials. Furthermore, alternative strategies involving the cross-coupling of alkyl halides with disodium disulfide or the use of disulfur dichloride (S2Cl2) present significant safety and operational hazards. The use of S2Cl2, in particular, is fraught with difficulties due to its corrosive nature and the generation of acidic byproducts that require rigorous neutralization and waste treatment protocols. These conventional pathways frequently suffer from limited substrate scope, where sensitive functional groups may not tolerate the harsh oxidative or acidic conditions required for bond formation. Additionally, the multi-step nature of many traditional syntheses, often requiring the isolation of intermediate thiols before oxidation, leads to increased production time and lower overall yields. For procurement managers, these inefficiencies translate directly into higher raw material costs and more complex supply chain logistics, as specialized and potentially hazardous reagents must be sourced and stored with extreme care. The environmental footprint of these older methods is also considerable, given the generation of sulfur-containing waste streams that demand specialized disposal procedures, thereby increasing the total cost of ownership for the manufacturing process.
The Novel Approach
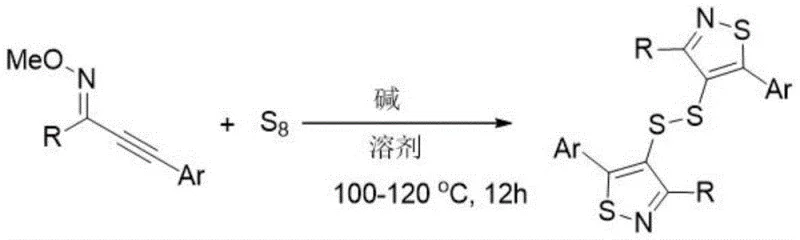
In stark contrast, the novel approach outlined in the patent data utilizes alkynyl oxime ethers as the primary substrate, reacting them directly with elemental sulfur in the presence of a non-nucleophilic base such as DBU. This method elegantly circumvents the need for smelly thiols or corrosive sulfur chlorides by employing odorless, cheap, and abundant elemental sulfur (S8) as the sulfur source. The reaction proceeds through a radical mechanism that simultaneously constructs the thiazole rings and the central disulfide linkage, effectively merging multiple synthetic transformations into a single operational step. This telescoping of reactions significantly simplifies the workflow, as there is no need to isolate unstable intermediate species, which often degrade or polymerize upon standing. The use of a mixed solvent system comprising NMP and water further enhances the green chemistry profile of the process, facilitating easier product isolation and reducing the reliance on purely organic volatile solvents. From a commercial perspective, this streamlined approach drastically reduces the complexity of the manufacturing protocol, allowing for faster batch turnover times and minimizing the risk of operator exposure to hazardous chemicals. The broad substrate compatibility demonstrated in the patent examples suggests that this methodology can be applied to a wide array of derivatives, making it a versatile tool for the rapid generation of diverse chemical libraries for drug discovery programs.
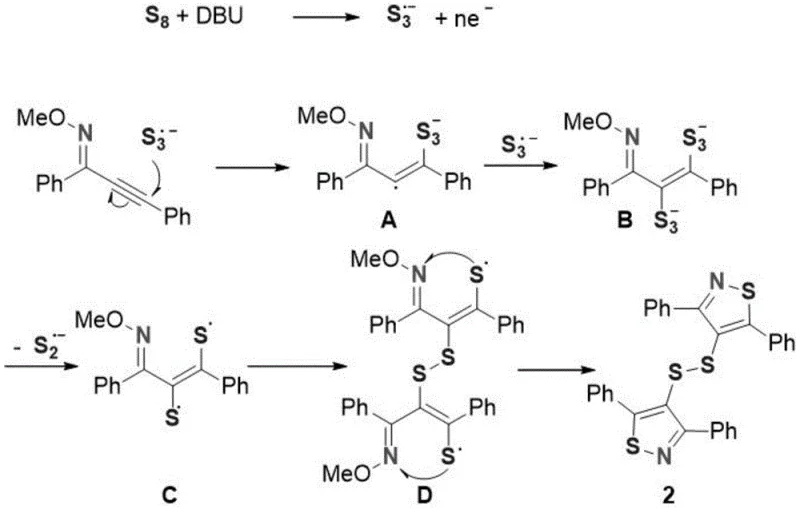
Mechanistic Insights into DBU-Promoted Radical Cyclization
The mechanistic pathway proposed for this transformation provides fascinating insights into the reactivity of elemental sulfur under basic conditions. The process initiates with the interaction between the octasulfur ring (S8) and the organic base DBU, which generates a trisulfur radical anion species (S3·-). This highly reactive radical species then attacks the electron-deficient carbon-carbon triple bond of the alkynyl oxime ether substrate, forming a vinyl radical anion intermediate. This initial addition step is crucial as it sets the stereochemistry and regiochemistry for the subsequent ring closure. Following this, a second equivalent of the trisulfur radical anion reacts with the intermediate to generate a dianionic species, which subsequently undergoes homolytic cleavage to release a disulfide radical. This sequence of events highlights the unique ability of the base-activated sulfur system to function as both a nucleophile and a radical source, driving the reaction forward without the need for external transition metal catalysts. The absence of heavy metals is a significant advantage for pharmaceutical applications, as it eliminates the stringent requirement for residual metal testing and purification steps that are often costly and time-consuming. Understanding this radical cascade allows process chemists to fine-tune reaction parameters such as temperature and base loading to maximize the formation of the desired disulfide-bridged product while suppressing potential side reactions.
Following the formation of the disulfide radical intermediate, the mechanism proceeds through an intermolecular radical dimerization event, which links two thiazole precursors together via the sulfur-sulfur bond. This dimerization is followed by an intramolecular cyclization where the nitrogen atom of the oxime ether attacks the adjacent carbon, displacing the methoxy group to close the thiazole ring. This final demethoxycyclization step is thermodynamically driven and results in the formation of the stable aromatic bisthiazole system. The entire cascade is remarkably efficient, converting simple starting materials into complex heterocyclic architectures with high atom economy. For quality control teams, the predictability of this mechanism means that the impurity profile is likely to be consistent and manageable, primarily consisting of unreacted starting materials or mono-cyclized byproducts that can be easily removed during standard workup procedures. The detailed elucidation of this mechanism, as depicted in the patent documentation, provides a solid theoretical foundation for scaling the reaction, ensuring that critical process parameters are maintained to prevent the accumulation of reactive radical species that could lead to safety incidents or product degradation.
How to Synthesize Bisthiazol-4-yl Disulfide Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the stoichiometry and thermal conditions specified in the patent to ensure optimal conversion and yield. The general procedure involves charging a reaction vessel with the alkynyl oxime ether substrate, elemental sulfur powder, and the organic base DBU in a solvent mixture of N-methyl-2-pyrrolidone (NMP) and water. The reaction mixture is then heated to a temperature between 100°C and 120°C and stirred for a duration of approximately 12 hours to allow the radical cascade to reach completion. Monitoring the reaction progress via thin-layer chromatography (TLC) is recommended to determine the exact endpoint, although the patent indicates that 12 hours is generally sufficient for most substrates. Upon completion, the workup procedure is straightforward, involving filtration to remove any insoluble sulfur residues, followed by aqueous washing and organic extraction. The crude product is typically purified using silica gel column chromatography with a petroleum ether and ethyl acetate gradient to afford the pure bisthiazol-4-yl disulfide derivative as a yellow solid. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results.
- Combine alkynyl oxime ether substrate with elemental sulfur (2 equivalents) and DBU base (3 equivalents) in an NMP-H2O solvent system.
- Stir the reaction mixture at a temperature range of 100°C to 120°C for approximately 12 hours to facilitate radical cyclization.
- Filter the reaction mixture, wash with saturated brine, extract with ethyl acetate, dry over sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing and supply chain management perspective, this novel synthetic route offers substantial advantages over legacy technologies, primarily driven by the simplification of the raw material basket and the reduction of processing complexity. The substitution of expensive and hazardous sulfurating agents with commodity-grade elemental sulfur represents a direct material cost saving, as sulfur is one of the most abundant and inexpensive chemicals available globally. Furthermore, the elimination of transition metal catalysts removes a significant cost center associated with catalyst procurement, recovery, and the analytical testing required to certify low metal residuals in the final API intermediate. The simplified one-pot nature of the reaction reduces the demand for reactor time and labor, as there are fewer unit operations such as intermediate isolations, drying, and transfers between vessels. This consolidation of steps inherently lowers the energy consumption of the process, contributing to a reduced carbon footprint and lower utility costs for the manufacturing facility. For supply chain heads, the robustness of the method implies a more reliable production schedule with fewer variables that could cause batch failures or delays, ensuring a steady flow of material to downstream customers. The use of common solvents like NMP and ethyl acetate further mitigates supply risk, as these are widely produced and less subject to the geopolitical volatility that can affect specialized reagents.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming from the use of low-cost elemental sulfur and the avoidance of expensive catalytic systems. By removing the need for precious metal catalysts, the process eliminates the capital expenditure associated with catalyst recovery units and the ongoing cost of catalyst replenishment. Additionally, the high yields reported in the patent examples, reaching up to 89%, indicate excellent material efficiency, meaning less raw material is wasted as byproducts or lost during purification. The simplified workup procedure, which avoids complex distillation or crystallization steps for intermediates, further reduces the operational expenditure related to energy and solvent usage. These factors combine to create a manufacturing process with a significantly lower cost of goods sold (COGS), providing a competitive edge in pricing negotiations with pharmaceutical clients who are increasingly pressure-sensitive regarding API costs.
- Enhanced Supply Chain Reliability: The reliance on readily available and stable starting materials greatly enhances the resilience of the supply chain. Elemental sulfur and alkynyl oxime ethers are commercially accessible from multiple global suppliers, reducing the risk of single-source dependency that can plague specialized chemical syntheses. The stability of the reagents allows for bulk purchasing and long-term storage without significant degradation, enabling manufacturers to build strategic inventory buffers against market fluctuations. Moreover, the robustness of the reaction conditions, which tolerate a wide range of functional groups, means that variations in raw material quality are less likely to result in batch rejection, thereby improving overall supply continuity. This reliability is critical for maintaining just-in-time delivery schedules for major pharmaceutical partners, ensuring that clinical trial timelines or commercial launch dates are not compromised by manufacturing bottlenecks or raw material shortages.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram or tonne scale is facilitated by the absence of exothermic hazards typically associated with thiol oxidations or the use of reactive sulfur chlorides. The reaction operates at moderate temperatures and atmospheric pressure, requiring only standard glass-lined or stainless steel reactors without the need for specialized high-pressure equipment. From an environmental compliance standpoint, the process generates minimal hazardous waste, as the primary byproduct is likely inorganic salts that can be treated in standard wastewater facilities. The reduction in solvent usage and the elimination of toxic sulfur gases contribute to a safer working environment and lower emissions, helping manufacturers meet increasingly stringent environmental regulations. This alignment with green chemistry principles not only reduces regulatory risk but also enhances the corporate sustainability profile, which is becoming a key differentiator in vendor selection processes for environmentally conscious multinational corporations.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this technology, we have compiled a set of frequently asked questions based on the patent specifications and industry standards. These queries address common concerns regarding reaction safety, product purity, and the feasibility of adapting this method for specific derivative synthesis. Understanding these details is essential for technical teams evaluating the integration of this route into their existing manufacturing portfolios. The answers provided are derived directly from the experimental data and mechanistic proposals within the patent document, ensuring accuracy and relevance for decision-makers.
Q: What are the primary advantages of using elemental sulfur in this synthesis?
A: Elemental sulfur serves as an odorless, inexpensive, and readily available sulfur source, eliminating the need for hazardous reagents like disulfur dichloride or unstable thiols found in conventional methods.
Q: What is the typical yield range for this bisthiazole disulfide synthesis?
A: According to the patent data, the process achieves yields ranging significantly, with optimized conditions reaching up to 89%, demonstrating high efficiency across various substituted substrates.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method utilizes simple operational procedures, avoids complex intermediate separations, and employs common solvents, making it highly amenable to commercial scale-up and process optimization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bisthiazol-4-yl Disulfide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodology described in patent CN111233783A for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to rapidly adapt this novel radical cyclization route for your specific project needs, ensuring a seamless transition from laboratory discovery to commercial supply. Our team of experienced process chemists is adept at optimizing reaction parameters to maximize yield and purity, leveraging our state-of-the-art facilities to handle complex heterocyclic syntheses with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply requirements are met with consistency and reliability. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs equipped with advanced analytical instrumentation to verify the identity and potency of every batch. Whether you require custom synthesis of specific bisthiazole derivatives or scale-up of the general disulfide scaffold, our capabilities are aligned to support your development timeline and commercial goals.
We invite you to engage with our technical procurement team to discuss how this innovative sulfur-based synthesis can drive value for your organization. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener, more efficient route. We encourage you to request specific COA data and route feasibility assessments to validate the performance of this method against your current supply chain benchmarks. Let us collaborate to optimize your intermediate sourcing strategy, ensuring a secure, cost-effective, and sustainable supply of critical building blocks for your next-generation therapeutics.
