Advanced Chiral Phosphoramides and N-Phosphonimines for Commercial Asymmetric Synthesis
The pharmaceutical industry's relentless pursuit of enantiomerically pure active ingredients has driven significant innovation in asymmetric synthesis methodologies. Patent CN101801984A introduces a transformative class of chiral phosphoramides connected with free amino groups and their corresponding chiral N-phosphonimines. This technology addresses critical limitations found in traditional chiral auxiliaries, specifically offering a robust alternative to the widely used but chemically sensitive N-sulfinylimines. By leveraging a phosphorus-based chiral scaffold, this invention enables the efficient construction of chiral amines, which are fundamental building blocks for a vast array of therapeutic agents. For R&D directors and procurement specialists, understanding this shift is vital, as it represents a pathway to more stable intermediates and streamlined supply chains for complex chiral drugs.
Historically, the field of asymmetric amine synthesis has been dominated by N-sulfinyl chemistry, pioneered by researchers such as Davis and Ellman. While effective, these traditional methods suffer from inherent chemical instabilities that pose significant risks during process development and scale-up. The N-sulfinyl group is notoriously sensitive to oxidation conditions, which can compromise the integrity of the intermediate during subsequent transformations. Moreover, the deprotection of N-sulfinyl groups often requires Brønsted-Lowry acids, conditions that can inadvertently destroy the chirality of the molecule or lead to racemization of the chiral sulfur center. Additionally, the preparation of these traditional auxiliaries frequently relies on expensive raw materials or environmentally hazardous disulfides, creating both cost and regulatory burdens for manufacturers seeking sustainable processes.
In stark contrast, the novel approach detailed in the patent utilizes a chiral phosphoramide backbone that exhibits remarkable chemical resilience. The synthesis begins with readily available chiral diamines, such as (1R,2R)-1,2-cyclohexanediamine, which are converted into N,N'-disubstituted derivatives. These intermediates are then phosphorylated to create a stable P-N chiral framework. Unlike their sulfur counterparts, these phosphoramides are not susceptible to oxidative degradation, allowing for greater flexibility in downstream synthetic steps. The resulting N-phosphonimines serve as potent electrophiles for asymmetric nucleophilic additions, facilitating the creation of diverse amine derivatives with high stereocontrol. This structural stability translates directly into operational reliability, reducing the risk of batch failures due to intermediate decomposition.
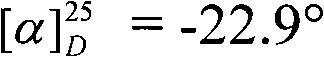
The mechanistic elegance of this system lies in the versatility of the phosphorus center and the modularity of the substituents. The synthesis of the core chiral phosphoramide involves a sequence of high-yielding transformations. Initially, the chiral diamine undergoes reductive amination or alkylation to protect the nitrogen atoms. Subsequently, reaction with phosphorus oxychloride (POCl3) in the presence of a base like triethylamine generates a phosphoryl chloride intermediate. This species is then converted to a phosphoryl azide using sodium azide, a reaction that proceeds efficiently at elevated temperatures (70°C). The final step involves catalytic hydrogenation over palladium on carbon to reduce the azide to the free primary amine, yielding the target chiral phosphoramide. This multi-step sequence is characterized by exceptional efficiency, with several steps reported to achieve quantitative yields, minimizing material loss and waste generation.
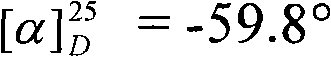
Once the chiral phosphoramide backbone is established, it serves as a precursor for the generation of chiral N-phosphonimines through condensation with aldehydes. This transformation is typically catalyzed by Lewis acids such as titanium tetrachloride (TiCl4) in the presence of a hindered base like DIPEA or triethylamine. The reaction proceeds smoothly at room temperature or slightly cooled conditions (0°C to room temperature), demonstrating excellent functional group tolerance. The resulting N-phosphonimines possess a P=N double bond that activates the adjacent carbon for nucleophilic attack, enabling the asymmetric construction of C-N bonds. This methodology supports a wide range of aromatic aldehydes, including those with electron-donating or electron-withdrawing substituents, as well as heteroaromatic systems, providing a broad scope for library synthesis and drug discovery applications.
How to Synthesize Chiral N-Phosphonimines Efficiently
The preparation of these high-value chiral intermediates follows a logical and scalable trajectory that integrates standard organic transformations with precise stereochemical control. The process begins with the modification of commercially available chiral diamines, followed by phosphorylation and functional group interconversion to install the reactive amine handle. The final condensation step with aldehydes is straightforward, requiring common laboratory reagents and mild conditions. This accessibility makes the technology particularly attractive for process chemists aiming to transition from milligram-scale discovery to kilogram-scale production without encountering significant engineering hurdles. The detailed standardized synthesis steps for implementing this protocol are outlined below.
- Synthesize N,N'-dibenzyl-1,2-cyclohexanediamine via reductive amination of 1,2-cyclohexanediamine with benzaldehyde and sodium borohydride.
- React the diamine with phosphorus oxychloride (POCl3) and triethylamine to form the phosphoryl chloride intermediate.
- Convert the phosphoryl chloride to phosphoryl azide using sodium azide in DMF at 70°C, followed by catalytic hydrogenation to yield the free amino chiral phosphoramide.
- Condense the chiral phosphoramide with various aromatic aldehydes using TiCl4 and DIPEA in dichloromethane to generate the target chiral N-phosphonimines.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this chiral phosphoramide technology offers compelling strategic benefits centered around cost efficiency and supply security. The elimination of sensitive sulfur chemistry removes the need for specialized handling protocols associated with toxic disulfides and odorous byproducts, thereby simplifying waste management and reducing environmental compliance costs. Furthermore, the high yields observed in the backbone synthesis, particularly the quantitative conversion in the reductive amination and hydrogenation steps, ensure maximal atom economy. This efficiency directly correlates to reduced raw material consumption, lowering the overall cost of goods sold (COGS) for the final chiral intermediates.
- Cost Reduction in Manufacturing: The synthetic route avoids the use of expensive and specialized sulfur-based reagents that are typical in traditional N-sulfinylimine chemistry. By utilizing commodity chemicals such as phosphorus oxychloride, sodium azide, and simple aromatic aldehydes, the material costs are significantly optimized. Additionally, the robustness of the intermediates reduces the likelihood of batch rejection due to decomposition, further enhancing cost predictability. The ability to perform reactions at room temperature also minimizes energy consumption associated with heating or cryogenic cooling, contributing to substantial cost savings in utility expenses.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including 1,2-cyclohexanediamine and benzaldehyde derivatives, are globally sourced commodities with stable supply lines. This reduces the risk of supply disruptions often associated with niche chiral pool reagents. The chemical stability of the phosphoramide intermediates allows for longer shelf lives and easier transportation compared to oxidation-sensitive sulfinyl compounds. Consequently, manufacturers can maintain leaner inventory levels while ensuring continuous production capability, effectively reducing lead time for high-purity chiral building blocks required for tight project timelines.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability, with reaction conditions that are easily transferable from laboratory glassware to industrial reactors. The absence of heavy metal catalysts in the condensation step (using TiCl4 which is manageable) and the use of standard solvents like dichloromethane and ethanol facilitate straightforward workup and purification. Moreover, the avoidance of toxic sulfur waste streams aligns with increasingly stringent global environmental regulations, positioning manufacturers favorably for audits and sustainability certifications. This green chemistry profile enhances the commercial viability of the intermediates in regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral phosphoramide technology. These insights are derived directly from the experimental data and structural advantages described in the patent literature, providing clarity for stakeholders evaluating this synthetic route. Understanding these nuances is essential for making informed decisions about process adoption and vendor selection.
Q: What are the advantages of N-phosphonimines over traditional N-sulfinylimines?
A: N-phosphonimines offer superior stability against oxidation compared to N-sulfinylimines. Furthermore, their deprotection does not require harsh acidic conditions that risk racemization, and their synthesis avoids toxic disulfides.
Q: What yields can be expected in the synthesis of the chiral phosphoramide backbone?
A: The patent data indicates exceptionally high yields for the backbone synthesis, with steps such as the reductive amination and the final hydrogenation achieving quantitative (100%) yields under optimized conditions.
Q: Is this technology suitable for large-scale pharmaceutical manufacturing?
A: Yes, the process utilizes standard reagents like POCl3 and Pd/C and operates at mild temperatures (room temperature to 70°C), making it highly scalable and compatible with existing industrial infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Phosphoramides Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced chiral intermediates play in accelerating drug development pipelines. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to full-scale manufacturing is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral phosphoramides and N-phosphonimines meets the exacting standards required by the global pharmaceutical industry. Our commitment to quality ensures that your synthesis projects proceed without interruption due to impurity profiles or stereochemical inconsistencies.
We invite you to collaborate with us to leverage this innovative technology for your next chiral amine project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific target molecules. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our supply of high-purity chiral phosphoramides can optimize your manufacturing economics and secure your supply chain.
