Industrial Scale Synthesis of 6-Amino-7-Fluoro-1,4-Benzoxazine-3(4H)-One via Optimized Catalytic Hydrogenation
The global demand for high-performance herbicides continues to drive innovation in the synthesis of key agrochemical intermediates, specifically focusing on efficiency and environmental compliance. Patent CN102002013A introduces a groundbreaking preparation method for 6-amino-7-fluoro-1,4-benzoxazine-3(4H)-one, a critical building block for the synthesis of the protoporphyrinogen oxidase (PPO) inhibitor herbicide, flumioxazin. This technical disclosure represents a significant leap forward from legacy manufacturing processes, offering a streamlined pathway that addresses long-standing challenges in yield optimization and waste management. By leveraging a robust catalytic hydrogenation strategy followed by a controlled acid-cyclization, this method eliminates the need for hazardous reducing agents and complex multi-step purifications. For R&D directors and procurement specialists alike, understanding the nuances of this patented route is essential for securing a reliable supply chain of high-purity agrochemical intermediates. The following analysis dissects the chemical engineering principles behind this innovation, highlighting its potential to redefine cost structures and production scalability in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 6-amino-7-fluoro-1,4-benzoxazine-3(4H)-one has been plagued by inefficient and environmentally burdensome methodologies. Prior art, such as methods utilizing iron powder and acetic acid for reduction, suffers from severe drawbacks including the generation of substantial quantities of iron sludge, which complicates waste treatment and increases disposal costs significantly. Furthermore, these traditional reduction techniques often result in lower reaction conversion rates and inconsistent product yields, necessitating extensive downstream processing to achieve acceptable purity levels. Other existing methods involving palladium or platinum catalysts have struggled with practical industrial application due to excessive solvent loads and the difficulty in separating the catalyst from the product mixture, leading to potential metal contamination and increased production costs. These legacy processes create bottlenecks in commercial scale-up of complex agrochemical intermediates, forcing manufacturers to balance between yield losses and environmental regulatory compliance.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a direct catalytic hydrogenation of 2-(5-fluoro-2,4-dinitrophenoxy) acetic acid or its salts, followed by a precise acid-catalyzed cyclization. This method fundamentally shifts the paradigm by employing molecular hydrogen as a clean reducing agent, thereby eliminating the heavy metal waste associated with iron powder reduction. The process operates under mild reaction conditions, typically between 10°C and 50°C, which enhances safety profiles and reduces energy consumption compared to high-temperature alternatives. By optimizing the solvent system to include mixtures of methanol and water, the new technique ensures excellent solubility of the substrate while maintaining the catalyst in a state that allows for easy filtration and recovery. This streamlined workflow not only boosts overall reaction yield but also simplifies the isolation of the final product, making it an ideal candidate for cost reduction in agrochemical intermediate manufacturing.
Mechanistic Insights into Catalytic Hydrogenation and Cyclization
The core of this synthetic breakthrough lies in the selective reduction of the nitro groups to amino groups without compromising the integrity of the fluorine substituent or the ether linkage. The reaction initiates with the adsorption of 2-(5-fluoro-2,4-dinitrophenoxy) acetic acid onto the surface of the metal catalyst, typically 5% Pd/C or Raney Nickel, within a pressurized hydrogen environment. The hydrogen molecules dissociate on the catalyst surface and sequentially reduce the two nitro groups at the 2 and 4 positions of the benzene ring to form the corresponding diamine intermediate, 2-(5-fluoro-2,4-diaminobenzene oxygen) acetic acid. This step is critical, as over-reduction or dehalogenation must be strictly avoided to maintain the structural fidelity required for the subsequent herbicidal activity. The use of specific solvent ratios, such as methanol to water in a 2:1 to 1:2 ratio, plays a pivotal role in stabilizing the transition states and ensuring high selectivity.
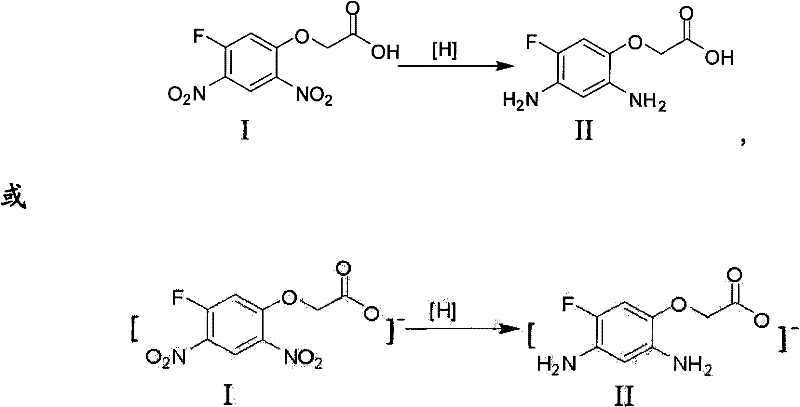
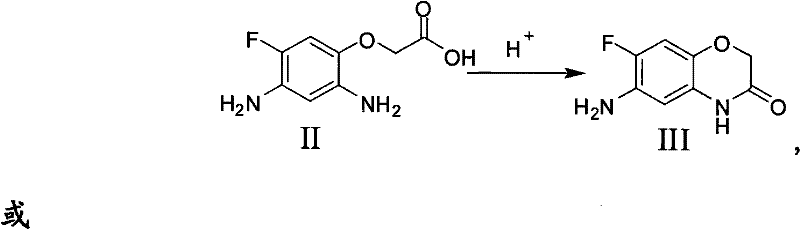
Following the successful formation of the diamine intermediate, the process transitions to the cyclization phase, which is driven by thermodynamic stability and acid catalysis. Upon adjusting the pH of the filtrate to an acidic range of 1 to 7, preferably using strong mineral acids like hydrochloric acid, the nucleophilic attack of the ortho-amino group on the carbonyl carbon of the acetic acid side chain is facilitated. This intramolecular condensation results in the formation of the seven-membered benzoxazine ring structure, releasing a molecule of water in the process. The precise control of pH during this stage is paramount for impurity control; maintaining the acidity prevents the premature polymerization of the sensitive diamine and directs the reaction exclusively towards the desired cyclic ketone. Finally, basifying the solution to a pH of 8-10 induces the precipitation of the target molecule, allowing for simple filtration and drying to obtain the high-purity final product.
How to Synthesize 6-Amino-7-Fluoro-1,4-Benzoxazine-3(4H)-One Efficiently
Executing this synthesis requires strict adherence to the optimized parameters regarding catalyst loading, hydrogen pressure, and solvent composition to maximize throughput. The process begins by charging the autoclave with the dinitro substrate and the chosen solvent system, followed by the addition of the heterogeneous catalyst. It is imperative to purge the system thoroughly with inert gas before introducing hydrogen to mitigate explosion risks and ensure catalyst activity. Once the hydrogenation is complete, indicated by the stabilization of hydrogen pressure, the mixture must be filtered immediately under a nitrogen blanket to protect the newly formed amino groups from oxidation. The subsequent cyclization step demands careful monitoring of pH levels to ensure complete ring closure without degrading the product. Detailed standardized synthesis steps see the guide below.
- Hydrogenation: React 2-(5-fluoro-2,4-dinitrophenoxy) acetic acid or its salt with hydrogen gas using a Pd/C or Raney Nickel catalyst in a methanol/water solvent system.
- Filtration: Remove the solid hydrogenation catalyst from the reaction mixture under nitrogen atmosphere to prevent oxidation of the sensitive diamine intermediate.
- Cyclization: Adjust the filtrate pH to 1-7 using a strong acid like hydrochloric acid to induce intramolecular ring closure, then basify to precipitate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere chemical yield. The shift away from stoichiometric reducing agents like iron powder to catalytic hydrogenation fundamentally alters the cost basis of production by removing the need for expensive waste disposal services associated with heavy metal sludge. Additionally, the ability to use common, commodity-grade solvents like methanol and water reduces raw material volatility and ensures consistent availability, thereby enhancing supply chain reliability. The simplified workup procedure, which relies on pH adjustment and filtration rather than complex chromatographic separations, drastically shortens the production cycle time, allowing for faster turnover and improved responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The elimination of iron powder and the associated acidic waste streams significantly lowers the operational expenditure related to environmental compliance and waste treatment facilities. Furthermore, the high selectivity of the catalyst minimizes the formation of by-products, reducing the loss of valuable starting materials and lowering the cost per kilogram of the final active intermediate. The recovery and reuse of the heterogeneous catalyst further contribute to long-term cost savings, making the process economically superior to traditional batch methods.
- Enhanced Supply Chain Reliability: By utilizing robust and widely available raw materials such as 2-(5-fluoro-2,4-dinitrophenoxy) acetic acid, manufacturers can mitigate the risks associated with sourcing specialized or unstable reagents. The mild reaction conditions reduce the wear and tear on reactor equipment, leading to less unplanned downtime and more predictable production schedules. This stability is crucial for maintaining continuous supply lines to downstream herbicide formulators who depend on just-in-time delivery models.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with parameters that translate effectively from laboratory autoclaves to industrial-sized reactors without significant re-engineering. The reduction in hazardous waste generation aligns perfectly with increasingly stringent global environmental regulations, future-proofing the manufacturing site against potential regulatory crackdowns. This green chemistry approach not only improves the corporate sustainability profile but also facilitates easier permitting for capacity expansion projects.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational feasibility. Understanding these details is vital for technical teams evaluating the integration of this process into existing production lines.
Q: What are the critical advantages of this hydrogenation method over traditional iron powder reduction?
A: Unlike traditional iron powder reduction which generates massive amounts of iron sludge waste and suffers from lower yields, this catalytic hydrogenation method utilizes clean hydrogen gas, significantly reducing three-waste discharge and simplifying downstream purification processes.
Q: How does the new process address the issue of catalyst separation found in prior art?
A: The process optimizes solvent systems (methanol/water mixtures) and reaction conditions to ensure the catalyst remains easily filterable, avoiding the difficult separation issues associated with previous high-temperature or specific solvent protocols.
Q: Is this synthesis route suitable for large-scale commercial production of herbicide intermediates?
A: Yes, the method operates under mild temperatures (10-50°C) and moderate hydrogen pressures, utilizing cheap and readily available raw materials, making it highly scalable and economically viable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Amino-7-Fluoro-1,4-Benzoxazine-3(4H)-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global agrochemical supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 6-amino-7-fluoro-1,4-benzoxazine-3(4H)-one performs consistently in your downstream synthesis of flumioxazin. Our dedication to process excellence means we can offer a stable supply of this key intermediate, supporting your long-term production goals.
We invite you to collaborate with us to optimize your sourcing strategy and achieve significant operational efficiencies. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your business growth and enhance your competitive edge in the herbicide market.
