Advanced Synthesis of JAK Inhibitor Intermediates for Commercial Scale-Up
The pharmaceutical industry is constantly seeking more efficient pathways to produce complex small molecules, particularly for high-demand therapeutic areas like immunology. Patent CN110903254A introduces a groundbreaking synthetic method for heterocyclic intermediates applied to JAK inhibitor drugs, specifically targeting compounds like Upadacitinib. This technology addresses critical bottlenecks in traditional manufacturing by utilizing a novel coupling and ring-closing strategy that bypasses expensive protecting groups. By shifting from conventional silylated reagents to more accessible alkynyl alcohols, this process offers a robust solution for producing high-purity pharmaceutical intermediates. The implications for supply chain stability and cost efficiency are profound, providing a competitive edge for manufacturers aiming to optimize their production of immune-modulating therapies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrrolopyrazine cores required for JAK inhibitors relied heavily on trimethylsilylacetylene as a key building block. While effective, this reagent carries a substantial cost burden due to the expensive silicon protecting group involved in its structure. Furthermore, the downstream processing necessitates additional steps to remove this silicon moiety, which increases waste generation and complicates the purification workflow. These factors collectively inflate the cost of goods sold and introduce potential points of failure in large-scale operations. The reliance on such specialized reagents also poses supply chain risks, as availability can fluctuate based on petrochemical feedstock dynamics. Consequently, manufacturers face pressure to find alternatives that maintain yield and purity without the associated economic penalties.
The Novel Approach
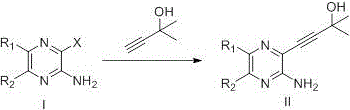
The innovative route disclosed in the patent circumvents these issues by employing 2-methyl-3-butyn-2-ol as a superior substitute for trimethylsilylacetylene. This reagent is not only significantly more affordable, reportedly costing about one-tenth of the traditional alternative, but it also streamlines the reaction sequence. The hydroxyl group in the new reagent facilitates a smoother cyclization process under basic conditions, eliminating the need for harsh deprotection steps. This strategic substitution enhances atom economy and reduces the environmental footprint of the synthesis. By simplifying the operational complexity, the new method enables faster turnaround times and more predictable manufacturing outcomes. This represents a pivotal shift towards more sustainable and economically viable production of complex heterocyclic intermediates.
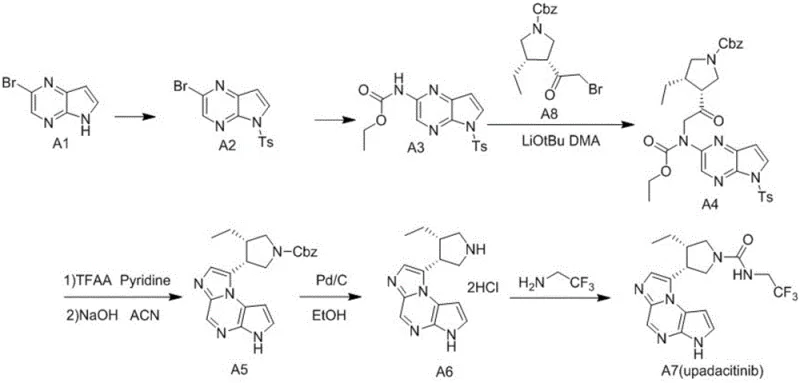
Mechanistic Insights into Pd-Catalyzed Sonogashira Coupling and Cyclization
The core of this technological advancement lies in the efficient execution of a transition metal-catalyzed Sonogashira coupling reaction. In this step, a halogenated pyrazine derivative reacts with the alkynyl alcohol in the presence of palladium and copper catalysts. The mechanism involves the oxidative addition of the palladium catalyst to the carbon-halogen bond, followed by transmetallation with the copper-acetylide species formed in situ. This precise orchestration ensures high regioselectivity and minimizes the formation of homocoupling byproducts. The use of mild bases and controlled temperatures further preserves the integrity of sensitive functional groups on the pyrazine ring. Understanding this catalytic cycle is crucial for R&D teams aiming to replicate the high yields reported in the patent examples while maintaining strict impurity control.
Following the coupling, the intermediate undergoes a base-promoted intramolecular cyclization to form the fused bicyclic system. This transformation is driven by the nucleophilic attack of the nitrogen atom on the activated alkyne, facilitated by the adjacent hydroxyl group. The reaction conditions, typically involving inorganic bases like sodium hydroxide or potassium tert-butoxide, are optimized to ensure complete conversion without degrading the product. This step effectively constructs the pyrrolo[2,3-b]pyrazine scaffold which is essential for the biological activity of the final JAK inhibitor. The robustness of this cyclization under various conditions highlights the versatility of the method for different substrate analogues. Such mechanistic clarity provides confidence in the scalability and reproducibility of the process for commercial applications.
How to Synthesize Pyrrolopyrazine Intermediate Efficiently
Implementing this synthesis requires careful attention to reaction parameters to maximize efficiency and safety. The process begins with the preparation of the halogenated starting material, followed by the critical coupling step under inert atmosphere to prevent catalyst oxidation. Subsequent workup procedures involve standard extraction and purification techniques that are well-established in pharmaceutical manufacturing. Detailed standardized synthetic steps see the guide below for specific stoichiometry and temperature profiles derived from the patent examples. Adhering to these protocols ensures that the final intermediate meets the rigorous quality standards required for downstream drug substance production. This structured approach minimizes variability and supports consistent batch-to-batch performance.
- Perform a Sonogashira coupling reaction between a halogenated pyrazine derivative and 2-methyl-3-butyn-2-ol using Pd/Cu catalysts.
- Execute a base-promoted cyclization and deprotection step to form the bicyclic pyrrolo[2,3-b]pyrazine core structure.
- Purify the final intermediate through crystallization or column chromatography to meet stringent pharmaceutical purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement and supply chain leaders, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical feasibility. The primary advantage stems from the drastic reduction in raw material costs, driven by the substitution of expensive silylated reagents with commodity chemicals. This shift directly impacts the bottom line, allowing for more competitive pricing structures in a crowded therapeutic market. Additionally, the simplified process flow reduces the demand for specialized equipment and extensive purification resources. These efficiencies translate into shorter manufacturing cycles and improved responsiveness to market demand fluctuations. Ultimately, this technology empowers organizations to build more resilient and cost-effective supply chains for critical immunology medications.
- Cost Reduction in Manufacturing: The replacement of trimethylsilylacetylene with 2-methyl-3-butyn-2-ol eliminates the cost premium associated with silicon protecting groups. This change alone drives a substantial decrease in the bill of materials, as the new reagent is widely available and priced significantly lower. Furthermore, the removal of dedicated deprotection steps reduces solvent consumption and waste disposal costs. The overall process efficiency means less energy is required per kilogram of product, contributing to lower utility expenses. These cumulative savings enhance the profit margin potential for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Sourcing 2-methyl-3-butyn-2-ol is far less risky compared to specialized silylated acetylenes, which often have limited supplier bases. The new reagent is a bulk chemical with stable global availability, reducing the likelihood of production stoppages due to material shortages. This reliability ensures continuous operation of manufacturing facilities and consistent delivery to clients. Moreover, the simplified synthesis reduces the number of critical intermediates that need to be stocked, lowering inventory carrying costs. A more robust supply chain mitigates the risk of delays and strengthens partnerships with downstream drug developers.
- Scalability and Environmental Compliance: The process is designed with industrial scale-up in mind, utilizing reaction conditions that are easily managed in large reactors. The higher atom economy means less chemical waste is generated per unit of product, aligning with increasingly strict environmental regulations. Reduced solvent usage and simpler workup procedures lower the burden on waste treatment facilities. This environmental friendliness not only ensures compliance but also enhances the corporate sustainability profile. Scalability is further supported by the robustness of the catalytic system, which maintains performance even as batch sizes increase to commercial tonnage.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this patented synthesis method. They are derived from a detailed analysis of the patent specifications and experimental data provided. These insights are intended to clarify the practical implications of adopting this technology for your specific manufacturing needs. Reviewing these answers can help stakeholders make informed decisions about process integration and resource allocation. The responses reflect the consensus on the method's viability and advantages in the current pharmaceutical landscape.
Q: What is the primary cost advantage of this new synthetic route?
A: The method replaces expensive trimethylsilylacetylene with 2-methyl-3-butyn-2-ol, which is significantly cheaper and eliminates the need for silicon protecting group removal steps.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent highlights simple operation conditions, easily available raw materials, and high atom economy, making it highly favorable for commercial scale-up.
Q: What specific JAK inhibitors can benefit from this intermediate?
A: This intermediate is specifically applicable to the synthesis of Upadacitinib and other related JAK1 inhibitor drugs used for treating rheumatoid arthritis and psoriatic arthritis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrrolopyrazine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we understand the critical importance of efficient and scalable synthesis routes for complex pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative patents like CN110903254A can be successfully translated into reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest industry standards. Our commitment to technical excellence allows us to navigate the complexities of heterocyclic chemistry with precision and reliability. Partnering with us means gaining access to a supply chain that is both robust and responsive to your evolving project requirements.
We invite you to discuss how this cost-effective synthesis route can be integrated into your supply chain for JAK inhibitor development. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs. Please contact us to request specific COA data and route feasibility assessments for this intermediate. By collaborating early, we can ensure a seamless transition from development to commercial supply, securing your position in the competitive immunology market. Let us help you optimize your production strategy with our proven expertise in fine chemical manufacturing.
