Revolutionizing PT141 Production: Enzymatic Cyclization for Commercial Scale-Up
Revolutionizing PT141 Production: Enzymatic Cyclization for Commercial Scale-Up
The pharmaceutical industry is constantly seeking more efficient pathways for complex peptide synthesis, and the methodology disclosed in patent CN109306366B represents a significant leap forward for the production of PT141, also known as Bremelanotide. This alpha-melanocyte stimulating hormone analog is a critical cyclic heptapeptide currently in advanced clinical stages for treating sexual dysfunction. Traditionally, the synthesis of such cyclic peptides has been plagued by low yields and harsh reaction conditions during the crucial cyclization step. However, this new technical disclosure introduces a paradigm shift by replacing conventional chemical coupling reagents with a highly specific enzymatic cyclization strategy. By leveraging biocatalysts such as thermolysin, papain, and specifically Butlasse 1, the process achieves a dramatic improvement in both yield and purity profiles. For R&D directors and procurement specialists, this transition from chemical to enzymatic activation offers a compelling value proposition, promising not only superior product quality with purity levels exceeding 99% but also a more sustainable and cost-effective manufacturing route that aligns with modern green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the cyclization of linear peptide precursors to form the macrocyclic structure of PT141 has relied heavily on chemical activation strategies involving potent coupling reagents and activating agents. As detailed in the background of the patent, prior art methods often utilize reagents such as PyBOP and DIEA in organic solvents like DMF under cryogenic conditions, typically around -20°C. These chemical approaches suffer from inherent inefficiencies, including slow reaction kinetics that require extended standing times of up to 24 hours to reach completion. Furthermore, the use of strong chemical activators frequently leads to the formation of racemization byproducts and oligomerization impurities, which complicates downstream purification and drastically reduces the overall isolated yield. In comparative examples provided within the technical data, these conventional chemical cyclization routes consistently demonstrate yields in the range of merely 30% to 40%, creating a significant bottleneck for commercial scalability. The reliance on hazardous organic solvents and the generation of stoichiometric amounts of chemical waste further exacerbate the environmental burden and operational costs associated with these legacy processes.
The Novel Approach
In stark contrast to the harsh chemical environments of the past, the novel approach outlined in CN109306366B employs a sophisticated enzymatic cyclization strategy that operates under mild, aqueous conditions. This method involves dissolving the purified linear peptide in a phosphate-buffered saline (PBS) solution, preferably adjusted to a pH of 6.0, and introducing a specific protease catalyst. The process eliminates the need for expensive and hazardous coupling reagents during the ring-closing step, thereby simplifying the reaction workup and reducing the impurity load. Experimental data from the patent highlights the exceptional performance of Butlasse 1 enzyme, which facilitates the cyclization at a moderate temperature of 30°C, achieving yields as high as 88% with purity maintained above 99.5%. This biocatalytic route not only accelerates the reaction timeline but also ensures high stereoselectivity, effectively preserving the chiral integrity of the sensitive amino acid residues like D-Phe and Nle. The transition to this enzymatic platform represents a robust solution for reliable PT141 supplier networks aiming to enhance supply chain stability and reduce manufacturing complexity.
Mechanistic Insights into Enzyme-Catalyzed Peptide Cyclization
The core innovation of this synthesis lies in the precise mechanistic action of the selected proteases, which function as highly efficient biocatalysts for intramolecular amide bond formation. Unlike chemical activators that indiscriminately activate carboxyl groups, enzymes like thermolysin and Butlasse 1 possess specific active sites that recognize the unique sequence of the PT141 linear precursor, specifically targeting the junction between the Lysine and Aspartic Acid residues to form the lactam bridge. This substrate specificity is crucial for minimizing side reactions such as intermolecular polymerization, which is a common failure mode in dilute chemical cyclizations. The reaction mechanism proceeds through a nucleophilic attack facilitated by the enzyme's catalytic triad, stabilizing the transition state and lowering the activation energy required for ring closure. By operating in an aqueous buffer system at pH 6.0, the enzyme maintains its optimal conformational stability, ensuring consistent catalytic turnover throughout the batch. This biological precision allows for the use of higher substrate concentrations compared to chemical methods, directly contributing to the observed volumetric productivity gains and making the process highly attractive for cost reduction in peptide manufacturing.
Furthermore, the enzymatic approach offers superior control over the impurity profile, a critical parameter for R&D directors focused on regulatory compliance and drug safety. Chemical cyclization often generates difficult-to-remove impurities resulting from epimerization at the activation site or incomplete reactions that require extensive chromatographic purification. In the enzymatic pathway, the mild reaction conditions (0-100°C, preferably 30°C) prevent thermal degradation and racemization of the sensitive peptide backbone. The patent data indicates that the crude product obtained after enzymatic cyclization possesses a significantly cleaner profile, which streamlines the subsequent reverse-phase high-performance liquid chromatography (RP-HPLC) purification step. The use of a mobile phase consisting of 0.1% acetic acid and acetonitrile allows for the efficient separation of the target cyclic peptide from any remaining linear precursor or minor enzymatic byproducts. This enhanced purity control ensures that the final API intermediate meets stringent quality specifications, reducing the risk of batch rejection and ensuring a consistent supply of high-purity PT141 for downstream formulation.
How to Synthesize PT141 Efficiently
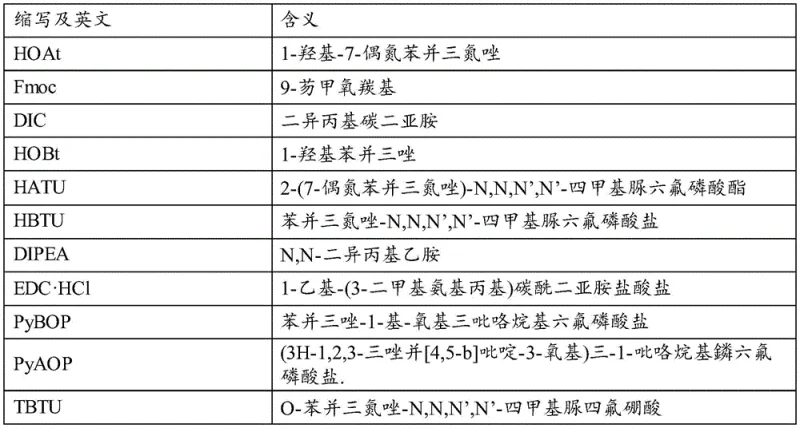
The synthesis of PT141 via this novel enzymatic route begins with the construction of the linear peptide chain using standard Fmoc solid-phase peptide synthesis (SPPS) protocols on a Wang resin. The process involves the sequential coupling of protected amino acids, including Fmoc-Lys(Boc)-OH, Fmoc-Trp(Boc)-OH, and Fmoc-Asp(OtBu)-OH, utilizing coupling systems such as DIC/HOBt or DIPEA/HATU to ensure high coupling efficiency at each step. Once the full linear sequence is assembled and N-terminally acetylated, the peptide is cleaved from the resin using a TFA-based cocktail containing scavengers like thioanisole and EDT to remove side-chain protecting groups. The resulting crude linear peptide is then precipitated, dried, and dissolved in a PBS buffer for the critical enzymatic cyclization step. For a comprehensive understanding of the specific operational parameters, reagent ratios, and purification techniques required to replicate this high-yield process, please refer to the detailed standardized synthesis guide provided below.
- Perform Fmoc solid-phase synthesis to construct the linear peptide sequence Ac-Nle-Asp-His-D-Phe-Arg-Trp-Lys on a Wang resin.
- Cleave the linear peptide from the resin using a TFA-based cocktail to remove protecting groups and obtain the crude linear peptide.
- Dissolve the linear peptide in PBS buffer (pH 6.0) and add Butlasse 1 enzyme at 30°C to catalyze the cyclization reaction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this enzymatic cyclization technology translates into tangible strategic advantages that extend beyond simple yield improvements. The elimination of stoichiometric chemical coupling reagents for the cyclization step significantly reduces the raw material bill of materials, as enzymes are used in catalytic amounts rather than in excess. This shift also mitigates the supply risk associated with specialized phosphonium or uronium salts, which can be subject to market volatility and long lead times. Moreover, the transition to an aqueous buffer system for the key ring-closing reaction drastically simplifies waste management protocols, reducing the volume of hazardous organic solvent waste that requires costly disposal. The mild reaction temperatures further contribute to energy efficiency, lowering the utility costs associated with cryogenic cooling or high-temperature heating typically required in traditional peptide synthesis. These factors collectively drive substantial cost savings and enhance the overall economic viability of large-scale PT141 production.
- Cost Reduction in Manufacturing: The enzymatic process fundamentally alters the cost structure by removing the need for expensive activating agents like PyBOP or HATU during the cyclization phase, which are typically required in molar excess to drive chemical reactions to completion. Instead, the use of biocatalysts allows for a more atom-economical process where the enzyme is recycled or used in minute quantities relative to the substrate. This reduction in reagent consumption directly lowers the variable cost per kilogram of the final product. Additionally, the higher yield achieved (up to 88% versus 30-40%) means that less starting material is wasted, effectively doubling the output from the same amount of linear peptide precursor. This efficiency gain is critical for maintaining competitive pricing in the global market for peptide APIs.
- Enhanced Supply Chain Reliability: By relying on commercially available enzymes and standard buffer salts, the manufacturing process becomes less dependent on niche chemical suppliers who may face production bottlenecks. Enzymes such as thermolysin and subtilisin are well-established industrial biocatalysts with robust supply chains, ensuring continuity of supply even during market disruptions. The simplified workflow also reduces the number of unit operations and intermediate handling steps, which minimizes the potential for operational delays or equipment downtime. This streamlined process flow enhances the agility of the supply chain, allowing for faster response times to fluctuating market demand and ensuring a steady flow of high-quality intermediates to downstream partners.
- Scalability and Environmental Compliance: The move towards aqueous-based enzymatic reactions aligns perfectly with increasingly stringent environmental regulations regarding solvent emissions and hazardous waste discharge. Scaling up a chemical cyclization process often requires massive volumes of organic solvents like DMF or DCM, posing significant safety and environmental challenges in large reactors. In contrast, the enzymatic method operates safely in water-based buffers, significantly reducing the facility's environmental footprint and simplifying the permitting process for capacity expansion. This green chemistry advantage not only future-proofs the manufacturing asset against regulatory changes but also appeals to end-clients who prioritize sustainability in their vendor selection criteria, thereby strengthening the long-term commercial partnership potential.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic synthesis route for PT141. These insights are derived directly from the experimental data and technical specifications outlined in patent CN109306366B, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this technology into their existing manufacturing portfolios or sourcing strategies. The answers reflect the specific conditions, such as pH levels and enzyme types, that define the success of this novel approach.
Q: What is the primary advantage of the enzymatic cyclization method for PT141?
A: The enzymatic method significantly improves yield compared to traditional chemical cyclization. While conventional methods using coupling reagents like PyBOP achieve yields around 30-40%, the enzymatic approach using Butlasse 1 can reach yields of 80-90% with purity exceeding 99%.
Q: Which enzymes are suitable for the cyclization of PT141 linear peptide?
A: The patent specifies several proteases effective for this transformation, including thermolysin, papain, A2 trypsin, subtilisin, and Butlasse 1. Among these, Butlasse 1 demonstrated the highest efficiency in the provided examples.
Q: What are the optimal reaction conditions for the enzymatic step?
A: The cyclization is optimally performed in a PBS buffer solution with a pH between 5 and 7, specifically pH 6.0. The reaction temperature is mild, preferably between 10-35°C, with 30°C being the specific embodiment for maximum yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable PT141 Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the enzymatic cyclization technology disclosed in CN109306366B and are fully equipped to leverage these advancements for our global clientele. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this high-yield process are realized at an industrial scale. Our state-of-the-art facilities are designed to handle both solid-phase peptide synthesis and downstream enzymatic transformations with precision, supported by rigorous QC labs that enforce stringent purity specifications for every batch. We understand that consistency and quality are paramount in the pharmaceutical supply chain, and our commitment to technical excellence ensures that every gram of PT141 we produce meets the highest international standards.
We invite you to collaborate with us to explore how this innovative synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable PT141 supplier dedicated to driving value through scientific innovation and operational excellence, ensuring your projects move forward without interruption.
