Advanced Synthesis of Triphenylamine-Thiophene Dyes for Scalable DSSC Manufacturing
Advanced Synthesis of Triphenylamine-Thiophene Dyes for Scalable DSSC Manufacturing
The global transition towards sustainable energy solutions has intensified the demand for high-performance, cost-effective materials for dye-sensitized solar cells (DSSC). As detailed in patent CN103554957A, a novel class of triphenylamine-thiophene organic dyes has emerged as a superior alternative to traditional Ruthenium-based polypyridine complexes. These metal-free organic sensitizers leverage a unique molecular architecture where an alkoxy-substituted triphenylamine donor is linked to a cyanoacrylic acid acceptor via a thiophene-derived conjugated bridge. This structural design not only mitigates the prohibitive costs associated with precious metal catalysts but also enhances the photovoltaic performance through suppressed electron recombination. The general structural formula of these advanced materials, featuring variable aryl groups (Ar) such as bithiophene or dithienopyrrole derivatives, is illustrated below, highlighting the modularity that allows for fine-tuning of electronic properties.
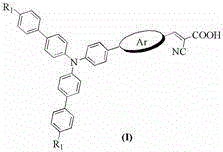
For procurement specialists and supply chain managers, the shift from inorganic coordination complexes to fully organic architectures represents a paradigm shift in sourcing strategy. The reliance on Ruthenium often introduces volatility in pricing and availability, whereas the precursors for these triphenylamine derivatives—such as substituted boronic esters and bromoaldehydes—are readily accessible within the fine chemical market. Furthermore, the synthetic pathway described avoids the need for specialized high-pressure equipment or cryogenic conditions, relying instead on standard thermal reflux processes. This inherent simplicity translates directly into lower capital expenditure for manufacturing facilities and a more resilient supply chain capable of sustaining the massive throughput required for gigawatt-scale solar deployment.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the benchmark for DSSC efficiency has been set by Ruthenium-based dyes, such as the N3 and N719 complexes, which have demonstrated power conversion efficiencies exceeding 10%. However, the commercial viability of these materials is severely hampered by the scarcity and high cost of Ruthenium, a platinum-group metal. The purification of these organometallic complexes is notoriously difficult, often requiring multiple recrystallization steps and chromatographic separations to remove trace metal impurities that can act as recombination centers, degrading device performance. Additionally, the synthetic routes for Ru-complexes often involve harsh conditions and low atom economy, generating significant hazardous waste. From a supply chain perspective, the concentration of Ruthenium mining in geopolitically unstable regions poses a continuous risk of supply disruption, making long-term planning for solar module manufacturers precarious and expensive.
The Novel Approach
The innovative approach presented in the patent data utilizes a purely organic framework centered around a triphenylamine donor and a thiophene pi-conjugated bridge. As shown in the specific structural variations below, the Ar group can be modulated between a bithiophene unit or a fused dithienopyrrole system, allowing chemists to precisely engineer the HOMO-LUMO energy levels for optimal light harvesting. This metal-free design inherently eliminates the heavy metal burden, simplifying the downstream purification process and ensuring a cleaner environmental footprint. The twisted stereostructure of the alkoxytriphenylamine moiety serves a dual purpose: it acts as an insulating barrier to prevent the approach of electrolyte ions to the semiconductor surface, thereby inhibiting charge recombination, while simultaneously ensuring good solubility in common organic solvents for easy processing. This results in a photosensitizer that combines high molar absorptivity with robust stability, offering a compelling value proposition for next-generation photovoltaics.
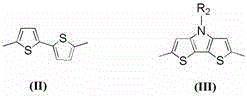
Mechanistic Insights into Suzuki-Knoevenagel Cascade Synthesis
The synthesis of these high-performance dyes relies on a robust two-step sequence beginning with a palladium-catalyzed Suzuki-Miyaura cross-coupling reaction. In this critical carbon-carbon bond-forming step, a hexyloxy-substituted triphenylamine boronic ester reacts with a thiophene-containing bromoaldehyde in the presence of a tetrakis(triphenylphosphine)palladium(0) catalyst and potassium carbonate base. The reaction proceeds in ethylene glycol dimethyl ether under reflux, facilitating the efficient coupling of the electron-rich donor to the conjugated bridge. The choice of solvent and base is crucial; the polar aprotic nature of the glyme solvent stabilizes the charged intermediates, while the mild inorganic base ensures the activation of the boron species without degrading the sensitive aldehyde functionality. This step constructs the core scaffold of the dye, establishing the extended pi-conjugation system necessary for effective electron delocalization and light absorption across the visible spectrum.
Following the isolation of the aldehyde intermediate, the final functionalization is achieved via a Knoevenagel condensation reaction. The aldehyde precursor is condensed with cyanoacetic acid in a mixture of chloroform and acetonitrile, catalyzed by piperidine. This reaction installs the anchoring carboxylic acid group and the electron-withdrawing cyano group, completing the push-pull electronic structure essential for directional electron injection into the TiO2 conduction band. The mechanism involves the formation of an enamine intermediate which attacks the carbonyl carbon, followed by dehydration to form the alpha,beta-unsaturated nitrile. The complete synthetic route for the bithiophene variant is depicted below, illustrating the straightforward transformation from commercially available building blocks to the final high-value photosensitizer with minimal side reactions.
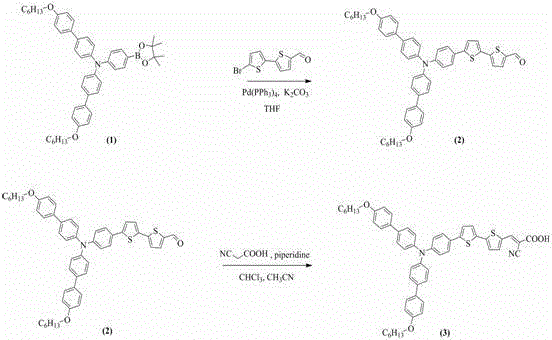
How to Synthesize Triphenylamine-Thiophene Dye Efficiently
The operational protocol for producing these dyes is designed for reproducibility and scalability, utilizing standard laboratory glassware and heating mantles that translate easily to pilot plant reactors. The process begins with the rigorous exclusion of oxygen via nitrogen purging to protect the palladium catalyst from oxidation, followed by the controlled addition of reagents to manage exotherms. After the initial coupling, the workup involves simple aqueous quenching and organic extraction, avoiding complex distillation setups. The final condensation step requires careful monitoring of reaction time to ensure complete conversion of the aldehyde, as residual starting material can be difficult to separate from the highly polar product. For a detailed breakdown of the specific stoichiometric ratios, temperature profiles, and purification parameters required to achieve the reported yields of up to 95% in the final step, please refer to the standardized synthesis guide below.
- Perform Suzuki coupling between hexyloxy-substituted triphenylamine borate and bithiophene bromoaldehyde using Pd(PPh3)4 catalyst in refluxing ethylene glycol dimethyl ether.
- Quench the reaction, extract with ethyl acetate, and purify the intermediate triphenylamine dithiophene formaldehyde via column chromatography.
- Conduct Knoevenagel condensation of the aldehyde intermediate with cyanoacetic acid and piperidine in chloroform/acetonitrile to yield the final dye.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this triphenylamine-thiophene synthesis route offers profound strategic advantages for organizations aiming to optimize their cost structures and secure their material supply lines. By transitioning away from noble metal-dependent chemistries, manufacturers can decouple their production costs from the volatile commodities market associated with Ruthenium and other platinum-group metals. The raw materials required, such as substituted phenylboronic acids and thiophene aldehydes, are produced on a multi-ton scale for various pharmaceutical and agrochemical applications, ensuring a deep and competitive supplier base. This abundance drives down the entry price for feedstocks and reduces the risk of single-source dependency. Furthermore, the elimination of heavy metals simplifies the regulatory compliance landscape, reducing the administrative burden and costs associated with environmental reporting and waste disposal in jurisdictions with strict toxicity controls.
- Cost Reduction in Manufacturing: The most significant economic driver is the complete removal of expensive transition metal catalysts from the final product structure, which eliminates the need for costly metal scavenging resins or complex chelation steps typically required to meet ppm-level metal specifications in electronic materials. The synthetic pathway utilizes commodity catalysts like palladium tetrakis which, while precious, are used in minute catalytic quantities and can potentially be recovered, unlike the stoichiometric amounts of Ruthenium needed in conventional dyes. Additionally, the high yields reported in the condensation step minimize raw material waste, directly improving the overall mass balance and reducing the cost per kilogram of the active pharmaceutical ingredient equivalent. The use of standard solvents like ethyl acetate and chloroform allows for efficient solvent recovery loops, further driving down operational expenditures.
- Enhanced Supply Chain Reliability: The modular nature of the Suzuki coupling allows for flexible sourcing of the aryl halide and boronic acid components; if one supplier faces disruption, the chemistry tolerates slight variations in substituent patterns without catastrophic failure of the device performance. This flexibility provides procurement managers with the leverage to negotiate better terms and maintain continuity of supply even during market fluctuations. The synthesis does not require specialized high-pressure hydrogenation or cryogenic cooling, meaning it can be manufactured in a wider range of existing multipurpose chemical plants without requiring dedicated capital investment in new infrastructure. This compatibility with existing assets accelerates the time-to-market for new product launches and ensures that production capacity can be ramped up rapidly to meet surging demand in the solar sector.
- Scalability and Environmental Compliance: The process generates primarily organic waste streams that are amenable to standard incineration or biological treatment, avoiding the generation of heavy metal sludge that requires hazardous waste landfills. The reaction conditions are mild, typically operating at atmospheric pressure and moderate temperatures, which significantly reduces energy consumption compared to high-energy processes. From a scale-up perspective, the exothermic profile of the reactions is manageable, allowing for safe operation in large batch reactors or continuous flow systems. This ease of scale-up ensures that the transition from gram-scale R&D batches to metric-ton commercial production is smooth and predictable, minimizing the technical risks that often delay the commercialization of new electronic chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this triphenylamine-thiophene dye technology in industrial settings. These answers are derived directly from the experimental data and structural analysis provided in the patent literature, offering a realistic view of the capabilities and limitations of the current state-of-the-art. Understanding these nuances is critical for R&D directors evaluating the feasibility of integrating these materials into existing DSSC assembly lines and for quality assurance teams establishing incoming inspection criteria.
Q: What are the key advantages of triphenylamine-thiophene dyes over Ruthenium-based complexes?
A: Triphenylamine-thiophene dyes eliminate the need for expensive noble metals like Ruthenium, significantly reducing raw material costs. Their distorted three-dimensional structure effectively inhibits electron recombination, leading to higher open-circuit voltage and improved photoelectric conversion efficiency exceeding 7%.
Q: What is the typical yield and purity profile for this synthesis route?
A: The patented process demonstrates robust yields, with the final Knoevenagel condensation step achieving up to 95% yield in optimized examples. The use of standard column chromatography ensures high purity suitable for sensitive optoelectronic applications.
Q: Is this synthesis scalable for industrial production of DSSC materials?
A: Yes, the process utilizes common solvents like ethylene glycol dimethyl ether and chloroform under standard reflux conditions, avoiding extreme pressures or temperatures. This makes the commercial scale-up of complex photosensitizers feasible and reliable for supply chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triphenylamine-Thiophene Dye Supplier
As the global demand for renewable energy technologies accelerates, the need for high-quality, consistently supplied photosensitizers has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this transition, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver these advanced organic dyes to the market. Our facility is equipped with stringent purity specifications and rigorous QC labs that utilize HPLC, NMR, and MS to ensure every batch meets the exacting standards required for high-efficiency solar cell fabrication. We understand that in the electronics and energy sectors, batch-to-batch consistency is not just a quality metric but a prerequisite for device reliability, and our ISO-certified processes guarantee the stability and performance you expect.
We invite potential partners to engage with our technical team to discuss how this metal-free dye technology can be integrated into your specific product roadmap. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching from Ru-based to organic sensitizers for your specific application volume. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments tailored to your project needs, ensuring a seamless transition to a more sustainable and cost-effective supply chain.
