Scalable Synthesis of 7-Troc-Docetaxel: A Strategic Breakthrough for Oncology R&D
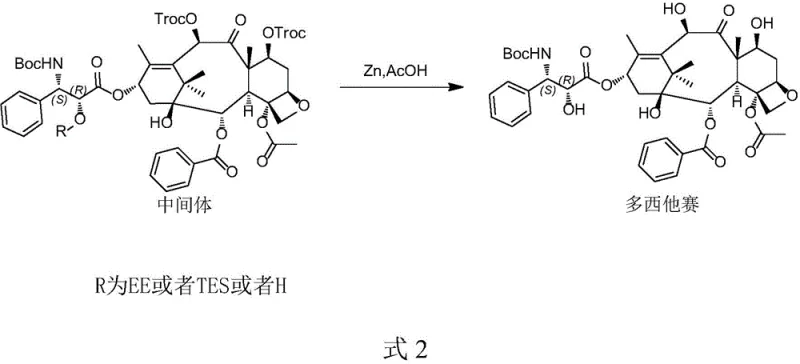
The pharmaceutical industry's relentless pursuit of high-purity oncology intermediates has led to significant advancements in taxane chemistry, specifically regarding the synthesis of critical impurities and reference standards. Patent CN115197176A introduces a robust, five-step preparation method for 7-trichloroethoxycarbonyl docetaxel (7-Troc-Docetaxel), a compound of immense value for impurity profiling and metabolic studies of the blockbuster drug Docetaxel. Unlike traditional approaches that struggle with regioselectivity, this invention leverages a sophisticated orthogonal protection strategy starting from 10-deacetylbaccatin III (10-DAB). By meticulously controlling the introduction and removal of Tes, Cbz, and Troc protecting groups, the process achieves directional synthesis with minimal by-product formation. For global procurement teams and R&D directors, this represents a pivotal shift towards more reliable pharmaceutical intermediate supplier capabilities, ensuring that critical research materials are available with the stringent purity specifications required for regulatory submissions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of taxane derivatives involving trichloroethoxycarbonyl (Troc) groups has been plagued by significant regiochemical challenges. Conventional literature often describes the direct use of TrocCl to protect hydroxyl groups on the baccatin core. However, as illustrated in the reaction scheme below, this approach lacks the necessary selectivity, frequently resulting in the simultaneous protection of both the C-7 and C-10 hydroxyl positions. This non-selective reactivity generates a complex mixture of structural analogs, including the di-Troc protected species, which possess physicochemical properties nearly identical to the target mono-protected impurity. Separating these closely related by-products requires extensive and costly chromatographic purification, drastically reducing overall throughput. Furthermore, older routes often rely on expensive four-membered or five-membered ring side chain acids early in the synthesis, inflating raw material costs and complicating the supply chain for what is essentially a research-grade intermediate.
The Novel Approach
The methodology disclosed in CN115197176A fundamentally reengineers the synthetic logic by decoupling the protection events. Instead of a brute-force protection strategy, the novel approach employs a 'mask-and-unmask' technique. It begins by selectively installing a triethylsilyl (Tes) group at the C-10 position, effectively blocking it from subsequent reactions. This allows for the exclusive installation of a benzyloxycarbonyl (Cbz) group at the C-7 position. Subsequent manipulation allows for the selective removal of the C-10 Tes group and its replacement with the desired Troc group. This stepwise construction ensures that at every stage, the reaction product is a single, distinct chemical entity rather than a statistical mixture. By deferring the introduction of the complex docetaxel side chain until the protecting group architecture is fully established, the process minimizes the risk of degrading expensive chiral acids, thereby offering substantial cost reduction in anticancer drug manufacturing contexts where material efficiency is paramount.
Mechanistic Insights into Orthogonal Protecting Group Strategy
The core innovation of this synthesis lies in the precise exploitation of the differing reactivities and stabilities of silyl ethers, carbamates, and carbonates. The initial silylation of 10-DAB using triethylchlorosilane (TesCl) and 2-methylimidazole in DMF at 0-5°C is kinetically controlled to favor the C-10 hydroxyl due to steric and electronic factors inherent to the baccatin III skeleton. The resulting Intermediate I serves as a crucial pivot point. In the subsequent step, the introduction of benzyl chloroformate (Cbz-Cl) with DMAP catalysis targets the remaining free C-7 hydroxyl. The Cbz group is chosen specifically for its stability under the acidic conditions required for later silyl deprotection, yet it remains orthogonal to the Troc group. The critical transformation occurs in Step S3, where hydrogen fluoride-pyridine complex is utilized to cleave the C-10 silyl ether without disturbing the C-7 Cbz carbonate. This regenerated C-10 hydroxyl is then immediately captured by TrocCl at low temperatures (0-10°C). This sequence guarantees that the Troc moiety ends up exclusively at the C-10 position (which corresponds to the 7-position numbering in the final docetaxel context after side chain attachment logic), avoiding the formation of the unwanted 7,10-di-Troc impurity.
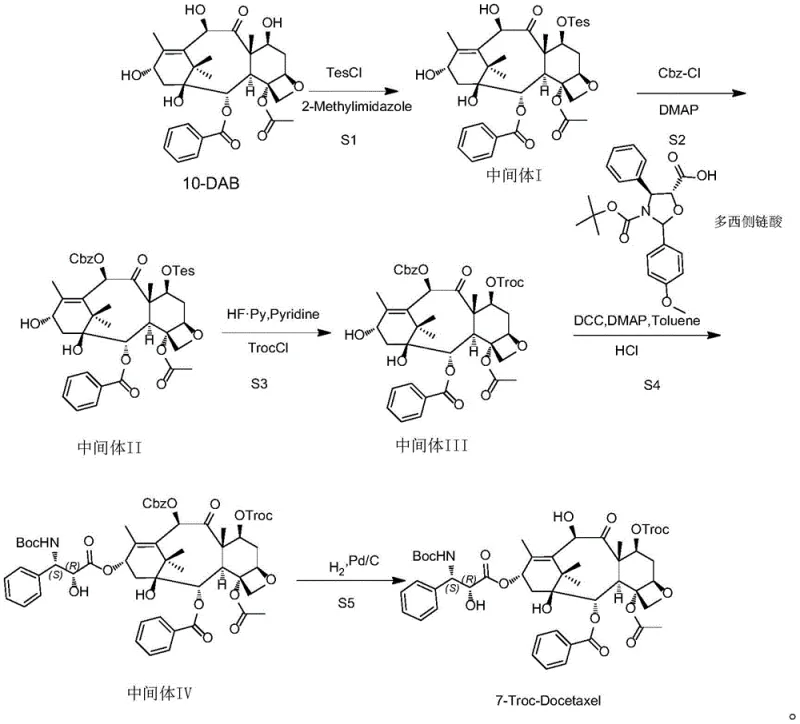
Impurity control is further enhanced by the final hydrogenolysis step. The use of 10% Pd/C under hydrogen atmosphere selectively removes the C-7 Cbz protecting group while leaving the Troc carbonate and the ester side chain intact. This chemoselectivity is vital; harsher deprotection methods could hydrolyze the sensitive oxetane ring or the ester linkages. The patent data indicates that by strictly maintaining reaction temperatures between 15-25°C during the coupling and deprotection phases, the formation of degradation products is suppressed. The result is a final product with demonstrated purity exceeding 95%, achieved through a logical progression of intermediates that are each easily purified by crystallization, minimizing the need for repetitive chromatography until the final polish.
How to Synthesize 7-Trichloroethoxycarbonyl Docetaxel Efficiently
The synthesis protocol outlined in the patent provides a clear, reproducible pathway for laboratory and pilot-scale production. The process begins with the dissolution of 10-DAB in DMF, followed by the controlled addition of TesCl to generate Intermediate I. This intermediate is then carried forward into THF for Cbz protection. A key operational detail is the temperature control during the Troc installation (Step S3), where the reaction mixture must be cooled to 0-10°C to prevent over-reaction or migration of the protecting groups. The coupling of the docetaxel side chain acid in Step S4 utilizes standard Steglich esterification conditions (DCC/DMAP) in toluene, a solvent choice that facilitates the removal of dicyclohexylurea (DCU) byproduct via filtration. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are provided in the technical guide below.
- Selective silylation of 10-DAB at the C-10 position using TesCl and 2-methylimidazole to form Intermediate I.
- Protection of the C-7 hydroxyl group with Cbz-Cl using DMAP catalysis to generate Intermediate II.
- Deprotection of the C-10 silyl group followed by selective Troc protection at C-10 to yield Intermediate III.
- Coupling with docetaxel side chain acid using DCC/DMAP activation to form Intermediate IV.
- Final hydrogenolysis using Pd/C to remove the C-7 Cbz group, yielding the target 7-Troc-Docetaxel.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the transition from non-selective to selective synthesis methodologies offers profound logistical and financial benefits. The primary advantage of this patented route is the elimination of complex separation trains. In conventional processing, isolating a specific mono-protected isomer from a di-protected mixture often consumes the majority of production time and solvent resources. By designing a route where each step yields a single dominant product, the new method drastically simplifies downstream processing. This streamlining translates directly into reduced solvent consumption and lower waste disposal costs, aligning with modern green chemistry initiatives. Furthermore, the reliance on commodity chemicals like TesCl, Cbz-Cl, and Pd/C, rather than specialized chiral beta-lactams for the core modification, insulates the production cost from the volatility of niche reagent markets.
- Cost Reduction in Manufacturing: The economic model of this synthesis is optimized by deferring the use of high-value chiral side chain acids until the final coupling step. In traditional routes where side chains are introduced earlier, any loss of material during protecting group manipulation results in the destruction of expensive chiral inventory. By building the protecting group architecture on the cheaper 10-DAB core first, the process ensures that the costly side chain is only exposed to a single coupling reaction. Additionally, the ability to purify intermediates via simple crystallization (using ethyl acetate and n-heptane) rather than flash chromatography significantly lowers the operational expenditure per kilogram, facilitating substantial cost savings in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more resilient supply chain. The process operates within mild temperature ranges (mostly 0-50°C), eliminating the need for cryogenic cooling or high-pressure reactors that can be bottlenecks in multi-purpose facilities. The reagents employed are stable, commercially available bulk chemicals with long shelf lives, reducing the risk of production delays due to raw material spoilage or scarcity. This reliability ensures consistent lead times for high-purity taxane intermediates, allowing R&D departments to plan their impurity profiling and stability studies with greater confidence.
- Scalability and Environmental Compliance: From a scale-up perspective, the avoidance of hazardous reagents and extreme conditions simplifies the technology transfer from lab to plant. The workup procedures involve standard aqueous washes and organic extractions, which are easily adaptable to large-scale separation equipment. Moreover, the high selectivity of the route reduces the generation of complex organic waste streams associated with separating structural isomers. This cleaner profile eases the burden on wastewater treatment facilities and supports compliance with increasingly stringent environmental regulations, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 7-Troc-Docetaxel. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, aiming to clarify the operational benefits of this new synthetic route for potential partners and technical stakeholders.
Q: Why is the conventional TrocCl protection method unsuitable for synthesizing 7-Troc-Docetaxel?
A: Conventional methods using TrocCl often result in non-selective protection of both C-7 and C-10 hydroxyl groups, creating difficult-to-separate structural analogs and significantly lowering the purity of the specific 7-Troc isomer required for impurity profiling.
Q: What are the key advantages of the 5-step selective protection route?
A: This novel route utilizes orthogonal protecting groups (Tes, Cbz, Troc) to directionally construct the molecule. It avoids expensive beta-lactam side chains until the final coupling, ensures single-product formation at each step, and operates under mild temperature conditions suitable for industrial scaling.
Q: Is this synthesis method scalable for commercial production?
A: Yes, the process avoids extreme reaction conditions, utilizing standard reagents like TesCl, Cbz-Cl, and Pd/C. The purification steps are streamlined, primarily requiring crystallization and a final column chromatography, making it highly viable for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Trichloroethoxycarbonyl Docetaxel Supplier
As the demand for well-characterized oncology impurities continues to grow, partnering with a manufacturer that possesses deep technical expertise in taxane chemistry is essential. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced synthetic strategies like the one described in CN115197176A to deliver superior quality materials. Our facility is equipped to handle the nuanced requirements of selective protection chemistry, ensuring that every batch meets rigorous quality standards. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, supported by stringent purity specifications and rigorous QC labs that utilize state-of-the-art HPLC and MS instrumentation to verify identity and purity.
We invite global pharmaceutical companies and research institutions to collaborate with us to secure their supply of critical taxane intermediates. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized processes can reduce your overall cost of goods. Please contact our technical procurement team today to request specific COA data and route feasibility assessments for your upcoming projects.
