Advanced 9-Chloroacridine Manufacturing: Enhancing Safety and Yield for Global Supply Chains
Introduction to Next-Generation 9-Chloroacridine Synthesis
The landscape of fine chemical manufacturing is constantly evolving, driven by the dual imperatives of safety and efficiency. A pivotal advancement in this domain is detailed in patent CN115710226A, which introduces a robust preparation method for 9-chloroacridine, a critical intermediate widely utilized in the synthesis of fluorescent probes and bio-active compounds. This technology addresses longstanding challenges in heterocyclic chemistry, specifically the hazards associated with phosphorus oxychloride handling. By shifting from traditional aqueous quenching to a vacuum distillation workup, the process not only mitigates the risk of violent exothermic reactions but also significantly enhances the overall yield and purity profile of the final product. For R&D directors and procurement specialists, understanding this mechanistic shift is essential for evaluating reliable pharmaceutical intermediate suppliers who prioritize both operational safety and cost-effectiveness in their manufacturing protocols.
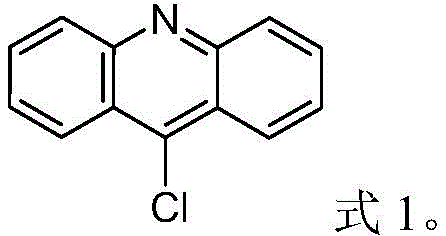
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 9-chloroacridine has been plagued by significant safety and efficiency bottlenecks. Traditional protocols, such as those referenced in prior art like CN107721925A, typically involve reacting N-phenyl anthranilic acid with a massive excess of phosphorus oxychloride, often in molar ratios ranging from 1:5 to 1:22.8. The critical failure point in these legacy methods is the post-reaction workup, which necessitates pouring the reaction mixture into large volumes of ice water. This step triggers a violent hydrolysis of the unreacted phosphorus oxychloride, releasing substantial amounts of heat and corrosive hydrogen chloride gas. Furthermore, the harsh aqueous environment promotes the hydrolysis of the target 9-chloroacridine itself, leading to the formation of acridone byproducts and a consequent reduction in yield, often capping efficiency at around 75% while generating excessive wastewater.
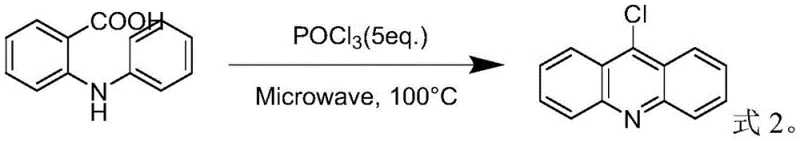
The Novel Approach
In stark contrast, the methodology disclosed in CN115710226A represents a paradigm shift towards green chemistry principles. Instead of immediate quenching, the novel process employs a vacuum distillation step immediately following the cyclization reaction. This allows for the simultaneous recovery of the solvent (toluene or xylene) and the unreacted phosphorus oxychloride before any water is introduced. By removing the acid source prior to neutralization, the process eliminates the violent exotherm and drastically reduces the volume of alkaline waste required for pH adjustment. This approach not only safeguards the integrity of the chloro-substituent against hydrolysis but also enables the direct recycling of valuable reagents, creating a closed-loop system that aligns perfectly with modern cost reduction in pharmaceutical intermediate manufacturing strategies.
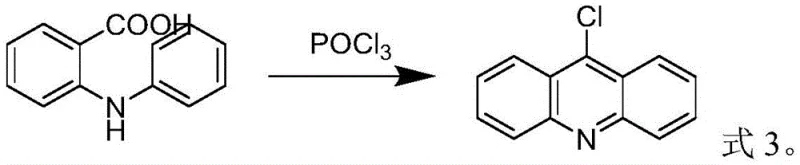
Mechanistic Insights into POCl3-Mediated Cyclization
The core of this synthesis lies in the Vilsmeier-Haack type cyclization mechanism, where phosphorus oxychloride acts as both a dehydrating agent and a chlorinating source. In the optimized protocol, N-phenyl anthranilic acid undergoes intramolecular condensation in a non-polar solvent like toluene at temperatures between 90°C and 110°C. The precise control of the molar ratio, maintained between 1:1.5 and 1:3, ensures sufficient activation of the carboxylic acid group without the gross excess seen in older methods. The reaction proceeds through the formation of an imidoyl chloride intermediate, which subsequently cyclizes to form the acridine ring system. The stability of this intermediate is crucial; by maintaining anhydrous conditions during the reaction phase and utilizing vacuum distillation for workup, the system prevents premature degradation, ensuring that the chlorine atom remains intact at the 9-position, which is vital for downstream nucleophilic substitution reactions.
Impurity control is another critical aspect where this mechanism excels. In conventional aqueous quenching, the localized high concentration of HCl and heat can catalyze the hydrolysis of the 9-chloro group to a ketone (acridone). The new method circumvents this by physically separating the acidic phosphorus species via distillation at 80-100°C under reduced pressure (-0.1 to -0.08 MPa). The subsequent neutralization is performed in a diluted organic phase using mild bases like ammonia water at controlled temperatures (0-45°C). This gentle pH adjustment (to 7-9) ensures that any residual acid is neutralized without exposing the sensitive chloro-acridine product to harsh hydrolytic conditions, resulting in purity levels consistently exceeding 99% and minimizing the formation of difficult-to-remove polar impurities.
How to Synthesize 9-Chloroacridine Efficiently
Implementing this advanced synthesis route requires precise adherence to the thermal and pressure parameters outlined in the patent data to ensure reproducibility and safety. The process begins with the dissolution of the starting material in an aromatic solvent, followed by the controlled addition of the chlorinating agent. The key differentiator is the post-reaction handling, where vacuum distillation replaces the traditional dump-and-quench method. This step is critical for recovering the solvent-reagent azeotrope for reuse. For a detailed breakdown of the specific operational parameters, including addition rates, temperature ramps, and distillation cut-points, please refer to the standardized synthesis guide below which encapsulates the critical process controls necessary for commercial scale-up of complex pharmaceutical intermediates.
- Dissolve N-phenyl anthranilic acid in toluene or xylene (Solvent S1) and add phosphorus oxychloride (1: 1.5 to 1:3 molar ratio) at 90-110°C for internal cyclization.
- Upon reaction completion, perform vacuum distillation at 80-100°C (-0.1 to -0.08 MPa) to separate and recover Solvent S1 and excess phosphorus oxychloride.
- Dissolve the solid crude residue in Solvent S2 (e.g., toluene), neutralize residual acid with aqueous ammonia or amine at 0-45°C to pH 7-9, and separate the organic layer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this vacuum-distillation-based protocol offers tangible strategic benefits beyond mere chemical yield. The primary advantage lies in the drastic simplification of the waste management infrastructure. By eliminating the need for massive quantities of ice water and the subsequent neutralization of huge volumes of acidic wastewater, the facility's environmental footprint is significantly reduced. This translates directly into lower disposal costs and reduced regulatory burden, facilitating smoother operations in regions with stringent environmental compliance standards. Furthermore, the ability to recycle the solvent and excess reagent creates a more resilient supply chain, reducing dependency on fluctuating raw material markets and insulating production costs from volatility.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the efficient utilization of phosphorus oxychloride. In traditional methods, the excess reagent is destroyed during quenching, representing a total loss of material cost. Here, the vacuum distillation step allows for the recovery and direct reuse of the phosphorus oxychloride-toluene mixture in subsequent batches. This circular economy approach within the reactor significantly lowers the variable cost per kilogram of product. Additionally, the reduction in base consumption for neutralization further trims operational expenses, delivering substantial cost savings without compromising on the quality of the high-purity OLED material or pharmaceutical intermediate produced.
- Enhanced Supply Chain Reliability: Safety is a cornerstone of supply chain continuity. The removal of the violent ice-water quenching step eliminates a major process hazard—the rapid generation of HCl gas and heat—which often necessitates specialized, corrosion-resistant equipment and extended downtime for maintenance. By adopting this milder workup procedure, manufacturers can utilize standard glass-lined or stainless steel reactors with greater confidence, reducing the risk of unplanned shutdowns due to equipment failure or safety incidents. This reliability ensures consistent delivery schedules for clients relying on these critical fluorescent probe intermediates for their own R&D pipelines.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies heat transfer and mixing challenges, particularly in exothermic quenches. This novel method is inherently more scalable because the heat load is managed through controlled distillation rather than instantaneous dilution. The reduction in "three wastes" (wastewater, waste gas, and solid waste) aligns with global sustainability goals, making the manufacturing process more attractive for long-term partnerships. The simplified effluent profile means that wastewater treatment facilities are less burdened, allowing for higher throughput capacities and ensuring that production can be ramped up to meet market demand without hitting environmental discharge limits.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this synthesis method. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering a transparent view into the process capabilities. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is vacuum distillation preferred over ice-water quenching in 9-chloroacridine synthesis?
A: Vacuum distillation removes excess phosphorus oxychloride before neutralization, preventing the violent exothermic hydrolysis and HCl gas release associated with traditional ice-water quenching, thereby improving safety and yield.
Q: How does this method improve the purity of 9-chloroacridine?
A: By avoiding the hydrolysis of the chloro-group which occurs during aggressive aqueous quenching, and by recycling solvents effectively, the process achieves purities exceeding 99% with minimal acridone impurities.
Q: Can the solvents and reagents be recycled in this process?
A: Yes, the close boiling points of toluene and phosphorus oxychloride allow them to be co-distilled and reused directly in subsequent batches without separation, significantly lowering raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9-Chloroacridine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer, more efficient synthetic routes is critical for maintaining competitiveness in the global fine chemicals market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the vacuum-distillation method for 9-chloroacridine are not just theoretical but practically viable on an industrial scale. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced chromatographic techniques to verify the absence of hydrolytic impurities like acridone, guaranteeing that every batch meets the exacting standards required for biological and electronic applications.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this optimized route can reduce your total cost of ownership. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to process innovation can drive value for your organization.
