Advanced Synthesis of Erlotinib-13C6 for Metabolic Research and Commercial Scale-up
Advanced Synthesis of Erlotinib-13C6 for Metabolic Research and Commercial Scale-up
The pharmaceutical industry's relentless pursuit of precision medicine has elevated the demand for stable isotope-labeled compounds, particularly for Absorption, Distribution, Metabolism, and Excretion (ADME) studies. Patent CN111187224A introduces a groundbreaking synthetic methodology for Erlotinib-13C6, a critical stable isotope-labeled analog of the EGFR inhibitor Erlotinib. This innovation addresses a significant gap in the market, as prior art lacked a disclosed method for synthesizing the key intermediate, m-aminophenylacetylene-13C6. By leveraging Benzene-13C6 as the foundational building block, this novel route offers a streamlined, cost-effective pathway to produce high-purity labeled standards essential for clinical toxicological and pharmacological research. The strategic design of this synthesis not only enhances isotopic integrity but also aligns with modern green chemistry principles by optimizing reaction conditions and reagent selection.
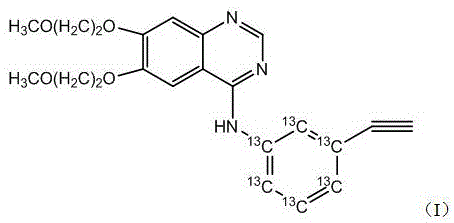
For R&D directors and procurement specialists, the implications of this patent extend beyond mere academic interest; it represents a viable commercial strategy for securing a reliable supply of complex labeled intermediates. The ability to synthesize Erlotinib-13C6 from basic aromatic precursors mitigates the risks associated with sourcing scarce, high-cost labeled anilines. This report delves into the technical nuances of the six-step synthesis, analyzing its mechanistic robustness, scalability, and potential for substantial cost reduction in pharmaceutical intermediate manufacturing. As we dissect the reaction pathway, it becomes evident that this methodology provides a blueprint for the efficient production of next-generation diagnostic and research tools.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of Erlotinib and its analogs relies heavily on the availability of specific aniline derivatives, such as m-aminophenylacetylene. In the context of stable isotope labeling, sourcing these specific precursors with high isotopic enrichment presents a formidable challenge. Conventional strategies often necessitate the purchase of expensive, custom-synthesized labeled anilines, which drives up the cost of goods sold (COGS) and introduces significant supply chain vulnerabilities. Furthermore, existing synthetic routes may involve harsh conditions or multiple protection-deprotection sequences that can lead to isotopic scrambling or loss of yield. The lack of a standardized, efficient protocol for converting simple labeled aromatics into the required ethynyl-aniline motif has historically bottlenecked the production of Erlotinib-13C6, limiting its accessibility for widespread clinical trial support and metabolic profiling studies.
The Novel Approach
The methodology disclosed in CN111187224A revolutionizes this landscape by adopting a "bottom-up" construction strategy starting from Benzene-13C6. This approach bypasses the need for complex labeled starting materials, utilizing a logical sequence of electrophilic aromatic substitution and cross-coupling reactions to install the necessary functional groups. The route features a highly efficient nitration followed by a selective iodination, setting the stage for a pivotal Sonogashira coupling. This strategic assembly allows for the precise placement of the 13C label within the phenyl ring while maintaining the structural integrity required for biological activity. By integrating a mild reduction step using sodium hydrosulfite and a final nucleophilic substitution, the process achieves the target molecule with high fidelity. This novel approach not only simplifies the synthetic tree but also opens the door for significant cost reduction in pharmaceutical intermediate manufacturing by utilizing more abundant and affordable raw materials.
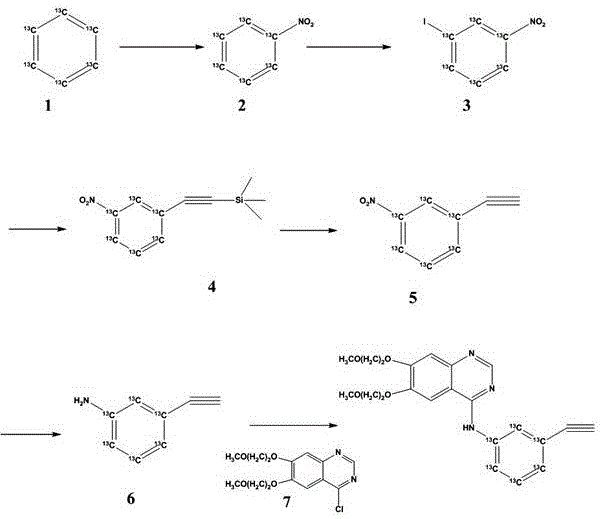
Mechanistic Insights into the Six-Step Synthetic Pathway
The core of this synthesis lies in its elegant manipulation of aromatic reactivity to construct the m-aminophenylacetylene scaffold. The process initiates with the nitration of Benzene-13C6 using ammonium nitrate and trifluoroacetic anhydride in chloroform. This electrophilic aromatic substitution proceeds under mild conditions to yield Nitrobenzene-13C6 (Compound 2), establishing the nitrogen handle required for the final drug scaffold. Subsequently, the introduction of the iodine atom via an oxidation-reduction system involving sodium periodate and potassium iodide in concentrated sulfuric acid is a critical transformation. This step generates 1-Iodo-3-nitrobenzene-13C6 (Compound 3), activating the ring for palladium-catalyzed cross-coupling. The choice of iodination over bromination or chlorination is deliberate, as the carbon-iodine bond offers superior reactivity in the subsequent Sonogashira reaction, ensuring high conversion rates even with the steric and electronic constraints imposed by the nitro group.
The Sonogashira coupling of Compound 3 with trimethylsilylacetylene represents the cornerstone of the carbon-carbon bond formation in this sequence. Catalyzed by a system comprising triphenylphosphine and cuprous iodide in a THF/triethylamine solvent matrix, this reaction installs the alkyne functionality with remarkable efficiency. Following this, the trimethylsilyl protecting group is cleanly removed using tetrabutylammonium fluoride (TBAF) to reveal the terminal alkyne in Compound 5. The reduction of the nitro group to the amine is achieved using sodium hydrosulfite in the presence of potassium carbonate. This choice of reducing agent is particularly advantageous for R&D teams focused on impurity control, as it avoids the use of catalytic hydrogenation which could potentially reduce the sensitive alkyne triple bond. Finally, the nucleophilic attack of the resulting 3-Ethynylaniline-13C6 (Compound 6) on the 4-chloro position of the quinazoline derivative (Compound 7) completes the synthesis, delivering Erlotinib-13C6 with the label precisely positioned for metabolic tracking.
How to Synthesize Erlotinib-13C6 Efficiently
Executing this synthesis requires careful attention to reaction stoichiometry and purification protocols to maximize yield and isotopic purity. The process is designed to be operationally simple, utilizing standard laboratory glassware and common organic solvents. Key parameters such as temperature control during the exothermic nitration and iodination steps, as well as the rigorous exclusion of moisture during the Sonogashira coupling, are critical for success. The following guide outlines the standardized operational framework derived from the patent examples, providing a clear roadmap for process chemists to replicate and scale this valuable transformation.
- Nitration of Benzene-13C6 using ammonium nitrate and trifluoroacetic anhydride to form Nitrobenzene-13C6.
- Iodination using sodium periodate and potassium iodide in sulfuric acid to yield 1-Iodo-3-nitrobenzene-13C6.
- Sonogashira coupling with trimethylsilylacetylene followed by TBAF deprotection to generate 3-Ethynylnitrobenzene-13C6.
- Reduction of the nitro group using sodium hydrosulfite to obtain 3-Ethynylaniline-13C6.
- Final nucleophilic substitution with 4-chloro-6,7-bis(2-methoxyethoxy)quinazoline to afford Erlotinib-13C6.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling economic and logistical benefits. By shifting the starting material from complex, low-volume labeled anilines to commodity-grade Benzene-13C6, the process fundamentally alters the cost structure of the final API intermediate. This strategic sourcing decision mitigates the risk of supply disruptions often associated with niche fine chemicals. Furthermore, the robustness of the reaction conditions—many of which proceed at room temperature or moderate heating—reduces energy consumption and equipment wear, contributing to a more sustainable and cost-efficient manufacturing profile. The elimination of specialized high-pressure hydrogenation equipment for the reduction step further lowers capital expenditure requirements for production facilities.
- Cost Reduction in Manufacturing: The most significant financial advantage stems from the raw material strategy. Benzene-13C6 is generally more accessible and cost-effective than custom-synthesized m-aminophenylacetylene-13C6. By performing the functionalization in-house through a linear six-step sequence, manufacturers can capture greater margin and reduce dependency on external suppliers for advanced intermediates. Additionally, the use of sodium hydrosulfite for reduction is economically favorable compared to precious metal catalysts, and the high yields reported in the patent examples (e.g., 95% for the Sonogashira step) indicate minimal material waste, directly translating to lower variable costs per kilogram of product.
- Enhanced Supply Chain Reliability: Diversifying the supply base is a key objective for any resilient supply chain. This method allows companies to qualify multiple vendors for the basic starting material, Benzene-13C6, rather than relying on a single source for the complex aniline precursor. The synthetic steps utilize widely available reagents like ammonium nitrate, potassium iodide, and triethylamine, which are staple chemicals in the global fine chemical market. This ubiquity ensures that production schedules are less likely to be impacted by regional shortages or logistics bottlenecks, guaranteeing a steady flow of material for clinical trial timelines.
- Scalability and Environmental Compliance: The process is inherently scalable, relying on unit operations such as liquid-liquid extraction, distillation, and crystallization that are standard in multi-purpose chemical plants. The solvent systems employed, including chloroform, THF, and isopropanol, have well-established recovery and recycling protocols, facilitating compliance with environmental regulations regarding volatile organic compound (VOC) emissions. The avoidance of heavy metal catalysts in the reduction step simplifies waste stream treatment, reducing the burden on effluent processing facilities and lowering the overall environmental footprint of the manufacturing campaign.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Erlotinib-13C6. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, offering clarity on process feasibility and quality assurance.
Q: What is the primary advantage of this Erlotinib-13C6 synthesis route?
A: The primary advantage is the use of commercially available Benzene-13C6 as the starting material, which significantly simplifies the supply chain compared to sourcing complex pre-labeled aniline derivatives. This strategy reduces raw material costs and improves overall process reliability.
Q: How does the process ensure high isotopic purity?
A: By initiating the synthesis with high-purity Benzene-13C6 and utilizing robust chemical transformations like nitration and Sonogashira coupling that preserve the carbon skeleton, the process minimizes isotopic dilution. Rigorous purification steps, including column chromatography, further ensure the final API intermediate meets stringent isotopic specifications.
Q: Is this method suitable for large-scale manufacturing?
A: Yes, the method employs standard unit operations such as liquid-liquid extraction, distillation, and reflux, which are easily scalable from kilogram to multi-ton production. The avoidance of exotic reagents and the use of common solvents like chloroform, THF, and isopropanol facilitate seamless technology transfer to commercial plants.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Erlotinib-13C6 Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality stable isotope-labeled compounds play in accelerating drug development and ensuring patient safety. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to clinical supply is seamless. We are committed to delivering Erlotinib-13C6 that meets stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality assurance guarantees that every batch provides the isotopic enrichment and chemical purity necessary for precise metabolic data interpretation.
We invite you to collaborate with us to optimize your supply chain for this vital research tool. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term R&D goals. Let us be your partner in advancing precision medicine through superior chemical synthesis.
