Strategic Analysis of Chiral Oxazoline Zinc Complex Synthesis and Commercial Scalability for Global Pharmaceutical Supply Chains
The landscape of asymmetric catalysis is continuously evolving, driven by the relentless demand for high-purity chiral intermediates in the pharmaceutical sector. A pivotal development in this domain is documented in patent CN102212078A, which details the synthesis and application of a novel chiral oxazoline-zinc complex. This specific metal-organic coordination compound, chemically defined as 2-[(4S)-4,5-dihydro-4-(1',1'-dimethylethyl)-3-oxazolinyl]aniline zinc chloride, represents a significant advancement in ligand design for transition metal catalysis. By leveraging the inherent chirality of D-leucinol, this complex establishes a robust stereogenic environment capable of inducing high levels of enantioselectivity in critical carbon-carbon bond-forming reactions. For R&D directors and procurement strategists alike, understanding the nuances of this technology is essential for optimizing synthetic routes and securing a reliable supply of advanced catalytic materials.
Furthermore, the integration of zinc as the central metal atom offers a compelling economic and environmental alternative to precious metal catalysts such as palladium or rhodium. The patent elucidates a straightforward yet highly effective preparation method involving the reflux of m-cyanoaniline and D-leucinol in the presence of anhydrous zinc chloride. This approach not only simplifies the manufacturing protocol but also aligns with modern green chemistry principles by reducing reliance on scarce resources. As we delve deeper into the technical specifics, it becomes evident that this innovation holds substantial promise for enhancing the efficiency of producing high-value pharmaceutical intermediates and fine chemicals on a commercial scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of chiral catalysts for asymmetric induction has often relied upon complex multi-step ligand preparations or the use of prohibitively expensive noble metals. Conventional methodologies frequently suffer from issues related to catalyst stability, where the active species may degrade under rigorous reaction conditions, leading to inconsistent batch-to-batch reproducibility. Moreover, many existing protocols require stringent anhydrous conditions that are difficult to maintain in large-scale reactors, thereby increasing operational costs and safety risks associated with specialized equipment. The reliance on rare earth metals or precious group metals also introduces significant supply chain vulnerabilities, as geopolitical factors can drastically impact availability and pricing. Additionally, the removal of trace metal residues from the final API intermediate often necessitates additional purification steps, further complicating the downstream processing and extending the overall production timeline.
The Novel Approach
In stark contrast, the methodology presented in the patent data offers a streamlined pathway that addresses many of these historical bottlenecks. By utilizing a direct condensation reaction between m-cyanoaniline and D-leucinol mediated by zinc chloride, the process achieves the formation of the active oxazoline-zinc complex in a single pot under reflux conditions. This novel approach eliminates the need for pre-functionalized ligands, thereby reducing the number of synthetic steps and minimizing waste generation. The use of chlorobenzene as a solvent provides a high-boiling medium that facilitates the necessary thermal energy for cyclization without requiring excessive pressure. 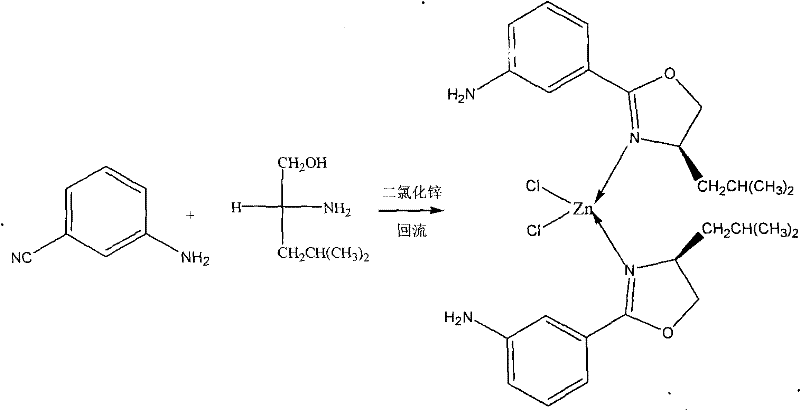 The resulting complex exhibits remarkable stability and catalytic activity, as evidenced by its performance in Henry reactions, making it a superior candidate for industrial adoption compared to less robust traditional systems.
The resulting complex exhibits remarkable stability and catalytic activity, as evidenced by its performance in Henry reactions, making it a superior candidate for industrial adoption compared to less robust traditional systems.
Mechanistic Insights into Zn-N Coordination and Cyclization
The efficacy of this chiral oxazoline-zinc complex stems from its unique coordination geometry, which creates a well-defined chiral pocket around the zinc center. The nitrogen atoms from both the aniline moiety and the oxazoline ring act as donor sites, forming stable coordinate covalent bonds with the zinc ion. This bidentate binding mode locks the ligand into a rigid conformation, effectively transmitting the chiral information from the leucine-derived side chain to the substrate during the catalytic cycle. The steric bulk provided by the tert-butyl group on the oxazoline ring plays a crucial role in discriminating between prochiral faces of the incoming electrophile, thereby ensuring high enantioselectivity. Understanding this mechanistic framework is vital for process chemists aiming to optimize reaction parameters such as temperature and concentration to maximize turnover numbers.
Moreover, the structural integrity of the complex is paramount for its long-term storage and handling characteristics. The patent data highlights the successful isolation of the complex as a white crystalline solid, confirmed by single-crystal X-ray diffraction analysis. ![Crystal structure of the 2-[(4S)-4,5-dihydro-4-(1',1'-dimethylethyl)-3-oxazolinyl]aniline zinc chloride complex](/insights/img/chiral-oxazoline-zinc-complex-pharma-supplier-20260305161321-01.png) This crystallographic evidence confirms the precise spatial arrangement of the ligands around the zinc atom, validating the proposed coordination model. From a quality control perspective, the distinct spectroscopic signatures, including specific IR absorption bands at 3353 cm⁻¹ and 1625 cm⁻¹, serve as reliable markers for verifying product identity and purity. Such rigorous structural characterization ensures that the material supplied meets the stringent specifications required for GMP-compliant pharmaceutical manufacturing, mitigating the risk of impurity-related failures in downstream synthesis.
This crystallographic evidence confirms the precise spatial arrangement of the ligands around the zinc atom, validating the proposed coordination model. From a quality control perspective, the distinct spectroscopic signatures, including specific IR absorption bands at 3353 cm⁻¹ and 1625 cm⁻¹, serve as reliable markers for verifying product identity and purity. Such rigorous structural characterization ensures that the material supplied meets the stringent specifications required for GMP-compliant pharmaceutical manufacturing, mitigating the risk of impurity-related failures in downstream synthesis.
How to Synthesize 2-[(4S)-4,5-dihydro-4-(1',1'-dimethylethyl)-3-oxazolinyl]aniline Zinc Chloride Efficiently
The synthesis of this high-performance chiral catalyst is designed to be operationally simple while maintaining high standards of chemical purity. The process begins with the careful selection of anhydrous reagents to prevent hydrolysis of the zinc chloride catalyst, followed by a controlled reflux period that allows for the complete conversion of starting materials into the desired coordination complex. Detailed procedural knowledge is essential for replicating the 45% yield reported in the patent embodiments, particularly regarding the workup and purification stages. For a comprehensive breakdown of the standardized operating procedures, please refer to the technical guide below.
- Combine m-cyanoaniline and D-leucinol with anhydrous ZnCl2 (30-40 mol%) in chlorobenzene under anhydrous and oxygen-free conditions.
- Reflux the mixture for 24 hours to ensure complete coordination and cyclization of the oxazoline ring.
- Remove solvent, dissolve residue in water, extract with chloroform, and purify via column chromatography to obtain the white crystalline complex.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this zinc-based catalytic technology presents a multitude of strategic benefits that extend beyond mere technical performance. The shift towards base metal catalysis significantly de-risks the supply chain by removing dependence on volatile precious metal markets. Furthermore, the simplified synthetic route reduces the overall manufacturing footprint, allowing for more flexible production scheduling and faster response times to market demands. By integrating this technology, organizations can achieve substantial cost reductions while simultaneously enhancing the sustainability profile of their chemical portfolios.
- Cost Reduction in Manufacturing: The replacement of expensive noble metal catalysts with abundant zinc chloride results in a drastic reduction in raw material costs. Since zinc is a widely available commodity metal, the price fluctuations are minimal compared to rhodium or palladium, leading to more predictable budgeting and lower cost of goods sold. Additionally, the elimination of complex ligand synthesis steps reduces labor and utility consumption, further driving down the operational expenditure associated with catalyst production. This economic efficiency makes the technology highly attractive for cost-sensitive applications in generic drug manufacturing and bulk fine chemical synthesis.
- Enhanced Supply Chain Reliability: The primary building blocks for this complex, specifically m-cyanoaniline and D-leucinol, are commercially available in large quantities from established global suppliers. This abundance ensures a continuous flow of raw materials, minimizing the risk of production stoppages due to原料 shortages. The robustness of the synthesis protocol also means that the catalyst can be produced in multiple geographic locations without significant loss of quality, facilitating a diversified and resilient supply network. This reliability is critical for maintaining uninterrupted API production schedules and meeting tight delivery deadlines for downstream customers.
- Scalability and Environmental Compliance: The use of chlorobenzene as a solvent, while requiring appropriate handling, is a well-understood industrial process that scales linearly from laboratory to plant scale. The reaction does not generate hazardous byproducts that require specialized disposal methods, simplifying waste management and reducing environmental compliance costs. The ability to purify the final product using standard column chromatography techniques ensures that the process can be adapted to continuous flow manufacturing or large batch operations with relative ease. This scalability supports the transition from pilot plant trials to full commercial production, enabling rapid market entry for new drug candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral oxazoline-zinc complex in industrial settings. These insights are derived directly from the patent specifications and practical experience in scaling similar coordination chemistry processes. Understanding these details helps stakeholders make informed decisions regarding technology transfer and process validation.
Q: What are the key advantages of using this zinc complex over traditional catalysts?
A: This complex offers a stable chiral environment derived from D-leucinol, providing specific enantioselectivity in reactions like the Henry reaction, while utilizing abundant zinc rather than expensive noble metals.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the process utilizes standard reflux conditions in chlorobenzene and common purification techniques like column chromatography, which are readily adaptable to larger scale manufacturing setups.
Q: What is the expected yield and purity profile for this complex?
A: The patent data indicates a synthesis yield of approximately 45% for the complex itself, with high structural integrity confirmed by elemental analysis and single-crystal diffraction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Oxazoline-Zinc Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced catalytic technologies play in accelerating drug discovery and development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless and efficient. We are committed to delivering materials with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Our dedication to quality assurance guarantees that the chiral oxazoline-zinc complex you receive meets the highest industry standards for performance and consistency.
We invite you to collaborate with us to explore how this innovative catalyst can optimize your specific synthetic challenges. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your current manufacturing processes. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value and efficiency in your supply chain.
