Advanced Asymmetric Synthesis of Pyrano-Pyrazole Acrylate Derivatives for Oncology Drug Development
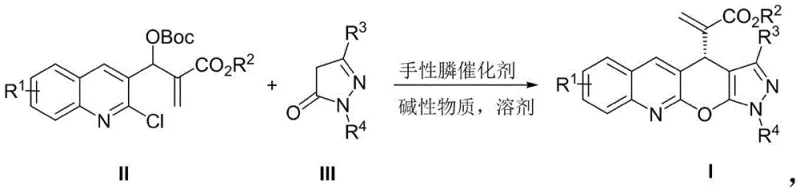
The pharmaceutical industry is constantly seeking robust and efficient pathways to construct complex heterocyclic scaffolds that serve as critical cores for next-generation therapeutics. Patent CN112939999A discloses a groundbreaking methodology for the preparation of pyrano-pyrazole acrylate derivatives, specifically 1,4-dihydropyrazolo[4',3':5,6]pyrano[2,3-b]quinoline-4-acrylate derivatives. This technology represents a significant leap forward in the field of asymmetric organocatalysis, offering a streamlined route to compounds with potent antitumor activities. The invention addresses the long-standing challenge of constructing these fused ring systems with high stereocontrol, utilizing a chiral phosphine-catalyzed asymmetric [3+3] cycloaddition strategy. For R&D directors and procurement specialists alike, this patent outlines a process that not only delivers high chemical purity but also relies on accessible starting materials, positioning it as a viable candidate for reliable pharmaceutical intermediate supplier networks aiming to support oncology drug pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of fused pyranopyrazole frameworks has been fraught with difficulties, often requiring multi-step sequences that suffer from poor atom economy and low overall yields. Conventional approaches frequently rely on harsh reaction conditions or stoichiometric amounts of chiral auxiliaries, which generate substantial waste and complicate the purification process. Furthermore, achieving high enantiomeric excess in the formation of the quaternary carbon centers within these rigid structures has historically been a bottleneck, often necessitating costly resolution steps that discard half of the produced material. These inefficiencies translate directly into higher manufacturing costs and extended lead times, creating significant friction in the supply chain for high-purity pharmaceutical intermediates. The lack of regioselectivity in older methods also leads to complex impurity profiles, demanding rigorous and expensive analytical controls to ensure patient safety.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages the unique reactivity of Morita-Baylis-Hillman (MBH) carbonates containing quinoline heterocycles. By employing a chiral small molecule catalyst, specifically a chiral phosphine, the method facilitates a direct and highly selective [3+3] cycloaddition with pyrazolone derivatives. This strategy bypasses the need for pre-functionalized intermediates and allows for the simultaneous formation of multiple bonds and stereocenters in a single operational step. The reaction proceeds under mild conditions, typically between -20°C and 80°C, and demonstrates exceptional tolerance for various functional groups on both the quinoline and pyrazolone rings. This versatility means that a wide library of analogs can be generated rapidly for structure-activity relationship (SAR) studies without redesigning the entire synthetic route, thereby accelerating the drug discovery timeline significantly.
Mechanistic Insights into Chiral Phosphine-Catalyzed Cycloaddition
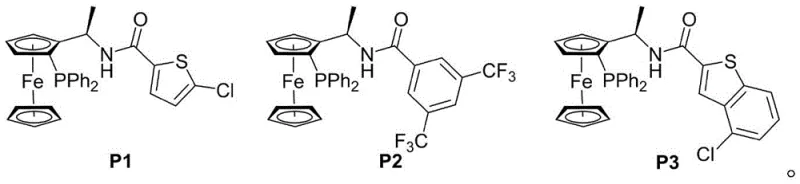
The core of this technological breakthrough lies in the activation of the MBH carbonate by the chiral phosphine catalyst. The catalyst initiates the reaction by attacking the electrophilic center of the MBH carbonate, generating a reactive zwitterionic dipolar intermediate. This intermediate is stabilized by the chiral environment provided by the catalyst backbone, which dictates the facial selectivity of the subsequent nucleophilic attack by the pyrazolone derivative. The precise spatial arrangement of the catalyst, such as the ferrocene-based structures P1, P2, and P3 shown in the patent data, ensures that the cyclization occurs with high fidelity, locking in the desired stereochemistry. This mechanism avoids the use of transition metals, eliminating the risk of heavy metal contamination in the final API, a critical consideration for regulatory compliance.
Furthermore, the reaction mechanism inherently suppresses the formation of side products through kinetic control. The rapid cyclization of the activated intermediate prevents competing decomposition pathways that are common with MBH carbonates. The choice of base, such as sodium carbonate, plays a crucial role in deprotonating the pyrazolone to enhance its nucleophilicity without promoting hydrolysis of the sensitive carbonate leaving group. This delicate balance results in products with excellent enantiomeric excess values, often exceeding 90% ee, as demonstrated in the specific examples provided. For quality assurance teams, this high level of stereocontrol simplifies the impurity profile, reducing the burden on downstream purification processes and ensuring consistent batch-to-batch quality essential for clinical supply.
How to Synthesize 1,4-Dihydropyrazolo[4',3':5,6]pyrano[2,3-b]quinoline Derivatives Efficiently
The practical implementation of this synthesis is straightforward and amenable to standard laboratory and pilot plant equipment. The general procedure involves mixing the quinoline-containing MBH carbonate and the pyrazolone derivative in a solvent like o-dichlorobenzene or toluene. Upon addition of the chiral phosphine catalyst and a mild inorganic base, the reaction progresses smoothly over a period of 36 to 240 hours depending on the specific substrate reactivity. The workup is equally simple, involving solvent removal and standard column chromatography, yielding the target acrylate derivatives as pale yellow solids with high purity. This operational simplicity is a key factor in reducing the cost reduction in pharmaceutical intermediate manufacturing, as it minimizes the need for specialized reactors or extreme temperature controls.
- Mix the quinoline heterocycle-containing MBH carbonate (Formula II) and the pyrazolone derivative (Formula III) in a suitable organic solvent such as o-dichlorobenzene or toluene.
- Add a chiral phosphine catalyst (such as P1, P2, or P3) and an alkaline substance like sodium carbonate to the reaction mixture under inert atmosphere.
- Stir the reaction at temperatures ranging from -20°C to 80°C for 36 to 240 hours, then recover the solvent and purify the product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers compelling advantages for supply chain stability and cost management. The reliance on organocatalysis rather than precious metal catalysis removes the volatility associated with rhodium or palladium pricing and availability. Additionally, the starting materials, including substituted quinolines and pyrazolones, are commodity chemicals that can be sourced from multiple vendors, mitigating the risk of single-source supply disruptions. The robustness of the reaction conditions allows for flexible scheduling in multipurpose manufacturing facilities, enhancing overall asset utilization. These factors combine to create a resilient supply chain capable of meeting the rigorous demands of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the high atom economy of the [3+3] cycloaddition significantly lower the raw material costs per kilogram of product. The high yields and enantioselectivity reduce the need for recycling or discarding off-spec material, further driving down the effective cost of goods sold. Moreover, the simplified purification process reduces solvent consumption and waste disposal fees, contributing to substantial cost savings in pharmaceutical intermediate manufacturing without compromising on quality standards.
- Enhanced Supply Chain Reliability: By utilizing readily available building blocks and avoiding specialized reagents, the procurement process becomes more agile and less prone to delays. The stability of the chiral phosphine catalysts ensures long shelf-life and consistent performance, reducing the frequency of catalyst replenishment orders. This reliability is crucial for maintaining continuous production schedules and meeting tight delivery windows for clinical trial materials and commercial launches.
- Scalability and Environmental Compliance: The reaction conditions are mild and do not require high pressure or cryogenic temperatures, making the scale-up from gram to ton scale technically feasible with standard engineering controls. The absence of heavy metals simplifies the environmental permitting process and reduces the load on wastewater treatment systems. This alignment with green chemistry principles not only lowers regulatory hurdles but also enhances the corporate sustainability profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these pyrano-pyrazole derivatives. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for potential partners.
Q: What are the key advantages of using chiral phosphine catalysts for this synthesis?
A: The use of chiral phosphine catalysts enables high enantioselectivity and regioselectivity in the construction of the complex fused heterocyclic framework, avoiding the need for expensive transition metals and simplifying downstream purification.
Q: Can this synthetic route be scaled for commercial production of API intermediates?
A: Yes, the method utilizes readily available raw materials and operates under relatively mild conditions with simple workup procedures, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Q: What is the biological potential of these pyrano-pyrazole derivatives?
A: The synthesized derivatives exhibit significant in vitro proliferation inhibition against human colon cancer cells (HT-29), non-small cell lung cancer cells (A549), and triple-negative breast cancer cells (MDA-MB-231).
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrano-Pyrazole Acrylate Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this asymmetric synthesis technology in advancing oncology therapeutics. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from benchtop discovery to full-scale manufacturing. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality and efficiency makes us the ideal partner for bringing complex molecules like these pyrano-pyrazole derivatives to market.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis to your specific needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to evaluate specific COA data and route feasibility assessments for new analogs, we are ready to provide the support you need. Contact us today to explore a partnership that combines cutting-edge chemistry with reliable commercial execution.
