Advanced Fmoc-Based Fragment Condensation for High-Purity Deserelin Acetate Manufacturing
Advanced Fmoc-Based Fragment Condensation for High-Purity Deserelin Acetate Manufacturing
The pharmaceutical industry constantly seeks robust synthetic routes for complex peptide therapeutics, and the methodology disclosed in patent CN110041407B represents a significant leap forward in the production of Deserelin acetate. This technical disclosure addresses the critical bottlenecks of conventional solid-phase peptide synthesis (SPPS), specifically targeting the issues of low crude product purity, complex purification workflows, and extended synthesis cycles that plague traditional linear approaches. By shifting from a direct long-chain assembly to a strategic fragment condensation strategy, this innovation enables the parallel synthesis of three distinct peptide segments, which are subsequently coupled with high efficiency. For R&D directors and process chemists, this approach offers a compelling solution to the persistent challenge of managing impurity profiles in decapeptide synthesis, ensuring that the final active pharmaceutical ingredient (API) meets stringent regulatory standards without necessitating excessive downstream processing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing Deserelin, such as the 5+4 fragment condensation technique referenced in prior art like Chinese patent 201510735214.8, suffer from inherent structural and kinetic disadvantages that hinder industrial scalability. In these conventional routes, the condensation of two large molecular fragments—a pentapeptide and a tetrapeptide—often results in steric hindrance that drastically lowers coupling efficiency and promotes the formation of difficult-to-separate impurities. The sheer size of these reacting fragments means that any incomplete reaction or racemization event creates byproducts that are structurally very similar to the target molecule, making purification via standard chromatography or crystallization extremely labor-intensive and costly. Furthermore, the linear nature of these older processes means that the entire synthesis timeline is sequential; a delay or yield loss in the early stages propagates through the entire batch, leading to unpredictable lead times and inconsistent batch-to-batch quality that is unacceptable for commercial API manufacturing.
The Novel Approach
The innovative strategy outlined in CN110041407B fundamentally reengineers the synthetic logic by decomposing the target decapeptide into three smaller, more manageable fragments that can be synthesized simultaneously and purified individually before final assembly. This modular approach allows for the rigorous purification of intermediates, such as the Fmoc-protected dipeptide and tripeptide segments, ensuring that only high-purity building blocks enter the final coupling stages. By utilizing H-Leu-2-chlorotrityl chloride resin as a stable anchor and employing optimized coupling reagents like HBTU and HOBT, the new method achieves a crude product yield of approximately 86% with a purity exceeding 87% prior to final purification. This represents a substantial improvement in process mass intensity and operational efficiency, as the ability to recrystallize intermediates removes impurities early in the workflow, thereby simplifying the final isolation of Deserelin acetate and reducing the overall solvent consumption and waste generation associated with the manufacturing process.
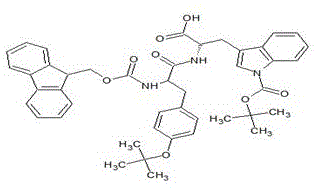
Mechanistic Insights into Fmoc Solid-Phase Fragment Condensation
The core of this synthetic breakthrough lies in the precise orchestration of Fmoc (9-fluorenylmethoxycarbonyl) chemistry, which offers orthogonal protection strategies that are essential for constructing complex peptide sequences without side reactions. The mechanism involves the activation of carboxylic acid termini on the peptide fragments using uronium salts like HBTU or TBTU in the presence of bases such as N-methylmorpholine (NMM), facilitating rapid amide bond formation with the free amine groups on the growing resin-bound chain. Crucially, the use of the 2-chlorotrityl chloride resin linker provides an acid-labile connection that allows for the mild cleavage of the fully protected peptide fragment under very mild acidic conditions (e.g., 1% TFA in DCM), preserving the acid-sensitive side-chain protecting groups like Boc and tBu until the final global deprotection step. This selectivity is vital for maintaining the integrity of sensitive residues such as Tryptophan and Histidine, preventing the formation of deletion sequences or modified byproducts that often arise from harsh cleavage conditions in traditional Wang or Rink amide resin protocols.
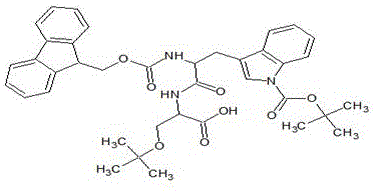
Impurity control is further enhanced by the specific selection of protecting groups and the implementation of intermediate recrystallization steps that act as kinetic filters for the reaction pathway. For instance, the synthesis of the H-Arg(pbf)-Pro-NHEt fragment involves the use of the Pbf (2,2,5,7,8-pentamethylchroman-6-sulfonyl) group for arginine protection, which is highly effective at suppressing guanidine side reactions and racemization during coupling. The patent data indicates that by hydrolyzing methyl esters and recrystallizing the resulting acid fragments, the process effectively removes unreacted starting materials and oligomeric impurities before they can be incorporated into the final peptide chain. This "purify-as-you-go" philosophy ensures that the final coupling between the resin-bound heptapeptide and the solution-phase dipeptide fragment proceeds with maximal efficiency, minimizing the formation of the notoriously difficult (n-1) deletion impurities that typically require preparative HPLC to remove in less optimized processes.
How to Synthesize Deserelin Acetate Efficiently
The synthesis of Deserelin acetate via this patented route requires a disciplined adherence to the three-fragment strategy, beginning with the parallel preparation of the H-Arg(pbf)-Pro-NHEt, Fmoc-Tyr(tBu)-D-Trp(Boc)-OH, and Fmoc-Trp(Boc)-Ser(tBu)-OH segments. Each fragment is subjected to rigorous quality control, including recrystallization and purity verification, before being introduced to the solid-phase synthesis cycle on the chlorotrityl resin. The detailed operational parameters, including specific molar ratios, solvent volumes, and reaction times for each coupling and deprotection step, are critical for reproducing the high yields and purity reported in the patent examples. To ensure successful technology transfer and scale-up, it is imperative to follow the standardized protocol precisely, particularly regarding the swelling of the resin and the monitoring of reaction endpoints using the ninhydrin test to prevent incomplete couplings.
- Simultaneously synthesize three key peptide fragments: H-Arg(pbf)-Pro-NHEt, Fmoc-Tyr(tBu)-D-Trp(Boc)-OH, and Fmoc-Trp(Boc)-Ser(tBu)-OH using solution-phase condensation and recrystallization.
- Perform Fmoc solid-phase synthesis on H-Leu-2Cl-Trt-Cl resin, sequentially coupling the synthesized fragments and removing Fmoc groups to build the protected peptide chain.
- Cleave the fully protected peptide from the resin, couple with the Arg-Pro fragment, remove side-chain protecting groups, and purify via ether precipitation to obtain Deserelin acetate.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this fragment condensation methodology offers transformative benefits in terms of cost stability and raw material security. By decoupling the synthesis into independent fragments, manufacturers can mitigate the risk of total batch loss; if one fragment fails quality control, it can be discarded and re-synthesized without compromising the expensive resin-bound intermediates or the other successfully synthesized segments. This modularity significantly enhances supply chain resilience, allowing for the stocking of key purified intermediates which can be rapidly assembled to meet fluctuating market demand for Deserelin acetate. Furthermore, the ability to purify intermediates via crystallization rather than relying solely on expensive preparative chromatography translates to a drastic reduction in solvent usage and processing time, directly lowering the cost of goods sold (COGS) and improving the overall margin profile for the final API.
- Cost Reduction in Manufacturing: The elimination of complex, low-yield direct condensations and the replacement with high-efficiency fragment couplings significantly reduces the consumption of expensive coupling reagents and solvents. The process avoids the need for extensive preparative HPLC purification of the crude product by ensuring high purity at the intermediate stage, which substantially lowers the operational expenditure associated with downstream processing and waste disposal.
- Enhanced Supply Chain Reliability: The parallel synthesis capability allows for the concurrent production of multiple fragments, effectively shortening the critical path of the manufacturing timeline and enabling faster response to procurement orders. The use of commercially available and stable starting materials, combined with robust recrystallization purification steps, ensures a consistent supply of high-quality intermediates, reducing the likelihood of production delays caused by raw material variability or synthesis failures.
- Scalability and Environmental Compliance: The protocol is designed with industrial scalability in mind, utilizing standard solid-phase equipment and avoiding exotic catalysts that pose handling hazards or environmental risks. The reduction in piperidine usage and the optimization of cleavage conditions contribute to a greener manufacturing process with a lower E-factor, aligning with modern sustainability goals and simplifying regulatory compliance for environmental discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Fmoc-based synthesis route for Deserelin acetate. These insights are derived directly from the experimental data and process descriptions within patent CN110041407B, providing a clear understanding of the method's capabilities and limitations for potential licensees and manufacturing partners. Understanding these nuances is essential for R&D teams evaluating the feasibility of adopting this technology for their own production pipelines.
Q: How does the fragment condensation method improve purity compared to traditional 5+4 synthesis?
A: The novel method utilizes three smaller, highly pure fragments synthesized in parallel with rigorous recrystallization steps, significantly reducing the accumulation of deletion sequences and difficult-to-remove impurities common in longer direct condensations.
Q: What are the scalability advantages of this Fmoc-based route?
A: By breaking the synthesis into manageable fragments that can be produced independently and purified before assembly, the process mitigates the risks of batch failure and simplifies quality control, making multi-kilogram production more robust.
Q: Does this method reduce the use of hazardous reagents?
A: Yes, the optimized protocol specifically reduces the reliance on piperidine for deprotection and utilizes efficient coupling agents like HBTU/HOBt, leading to a safer operational environment and simplified waste treatment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Deserelin Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the competitive landscape of peptide therapeutics, and we possess the technical expertise to translate the innovations of CN110041407B into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Deserelin acetate meets the highest international pharmacopoeia standards, providing our partners with the confidence needed to advance their clinical and commercial programs.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By leveraging our optimized fragment condensation platform, we can offer a supply solution that balances cost-efficiency with uncompromising quality. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and a comprehensive quotation that demonstrates the tangible value of partnering with a leader in advanced peptide manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
