Advanced Synthesis of Topramezone Intermediates: A Safer, Scalable Route for Global Agrochemical Production
The global demand for high-efficiency herbicides like topramezone continues to drive innovation in the synthesis of key heterocyclic intermediates. Patent CN113372288A introduces a transformative synthetic methodology for producing 3-halogenated-5,5-dimethylisoxazole, a critical building block in modern agrochemistry. This technology represents a paradigm shift from hazardous gas-phase reactions to a controlled, liquid-phase protocol starting from readily available glyoxylic acid. For R&D directors and procurement strategists, this patent offers a compelling alternative to legacy processes that have long been plagued by safety concerns and complex purification requirements. By leveraging a robust [3+2] cyclization strategy followed by a Huang Minlon reduction, this route ensures high purity levels exceeding 95% while simplifying the operational workflow. The implications for supply chain stability are profound, as the reliance on dangerous gaseous feedstocks is completely eliminated in favor of stable, storable liquid reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 5,5-dimethylisoxazole derivatives has relied heavily on methodologies that introduce significant operational risks and logistical bottlenecks. One prominent prior art method, disclosed in WO2006068092A1, utilizes the cyclization of dibromoformaldehyde oxime with isobutene gas. While chemically effective, the use of isobutene presents severe safety challenges, particularly when scaling up to multi-ton production capacities where gas leakage and explosion risks are non-trivial concerns. Furthermore, this legacy process typically employs n-butyl acetate as a solvent, which possesses a high boiling point and is prone to hydrolysis under alkaline conditions, making solvent recovery difficult and energy-intensive. Another existing approach, described in WO2008000469, depends on a complex catalytic system involving organic acids and bases such as N-methylaniline and trifluoroacetic acid. These catalysts are notoriously difficult to separate from the reaction mixture, leading to contamination of the recycled solvent and necessitating costly disposal or regeneration protocols. Additionally, the direct use of chlorine gas for chlorination in these older routes adds another layer of hazardous material handling that modern safety standards strive to minimize.
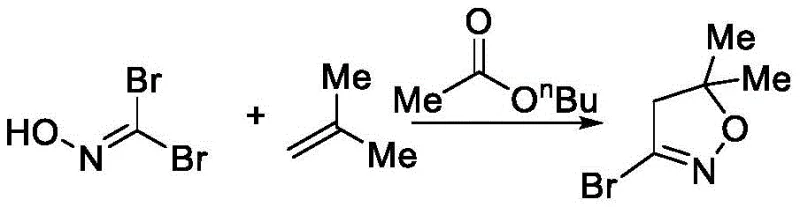
The Novel Approach
In stark contrast to these hazardous legacy methods, the novel synthesis route detailed in CN113372288A utilizes glyoxylic acid as a foundational starting material, initiating a sequence that is inherently safer and more controllable. The process begins with the formation of a glyoxylic oxime, which is subsequently halogenated to create a reactive dihalo-intermediate. This intermediate then undergoes a highly efficient [3+2] cycloaddition with unsaturated aldehydes or alkenes to construct the isoxazole core. A key advantage of this approach is the avoidance of gaseous hydrocarbon feedstocks; instead, the reaction proceeds in common organic solvents like dichloromethane or methanol, which are easier to handle and recycle. The final transformation utilizes the Huang Minlon reduction to convert the formyl group into the requisite methyl group, completing the synthesis of the 3-halogenated-5,5-dimethylisoxazole scaffold. This strategic redesign of the synthetic pathway not only mitigates safety risks associated with high-pressure gases but also streamlines the downstream processing by reducing the formation of tarry byproducts and facilitating easier solvent removal.
Mechanistic Insights into Glyoxylic Acid-Based Cyclization
The core chemical innovation lies in the utilization of a dihalogenated oxime derivative as a 1,3-dipole equivalent in the cycloaddition step. Mechanistically, the reaction between the halogenated oxime (Formula 1) and the electron-deficient alkene or aldehyde (Formula 2) proceeds through a concerted or stepwise dipolar cycloaddition to form the isoxazoline ring system. The presence of the halogen atoms on the oxime carbon enhances the electrophilicity of the dipole, facilitating the attack on the nucleophilic double bond of the alkene substrate. This electronic activation allows the reaction to proceed under milder conditions compared to unactivated nitrile oxides, typically requiring temperatures between -20°C and 50°C. The subsequent aromatization or stabilization of the ring is managed through the careful selection of base and solvent systems, ensuring that the delicate isoxazole structure is preserved without undergoing unwanted ring-opening side reactions. The use of inorganic bases such as potassium carbonate or sodium hydroxide further simplifies the workup, as these salts can be easily removed via aqueous washing, leaving behind a clean organic phase rich in the desired cyclic intermediate.
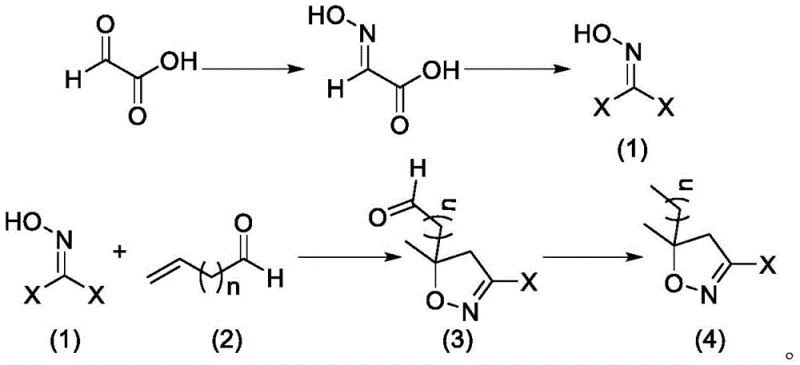
Impurity control is rigorously maintained throughout the four-step sequence, particularly during the final Huang Minlon reduction. In this step, the hydrazine hydrate reacts with the carbonyl functionality of the isoxazole intermediate to form a hydrazone, which is then decomposed under basic conditions at elevated temperatures (100-250°C) to release nitrogen gas and yield the methyl group. A critical aspect of this mechanism is the prevention of over-reduction or degradation of the isoxazole ring, which can be sensitive to strong nucleophiles. The patent specifies the use of high-boiling acetal solvents like diethylene glycol, which provide the necessary thermal environment for the reduction while stabilizing the reaction medium. By optimizing the molar ratio of hydrazine to substrate (typically 1:0.5 to 1:50) and strictly controlling the pH during the neutralization phase, the process minimizes the formation of hydrazine-derived impurities. This precise control results in a final product with a purity profile that consistently exceeds 95%, significantly reducing the burden on downstream crystallization or distillation units.
How to Synthesize 3-Halogenated-5,5-Dimethylisoxazole Efficiently
The synthesis of this valuable agrochemical intermediate is achieved through a logical four-step sequence that prioritizes operational simplicity and yield optimization. The process begins with the conversion of glyoxylic acid to its oxime derivative, followed by halogenation to activate the molecule for cyclization. The crucial ring-forming step involves reacting this activated species with an unsaturated partner, after which the Huang Minlon reduction finalizes the structure. Detailed standard operating procedures for temperature control, reagent addition rates, and workup protocols are essential for replicating the high yields reported in the patent examples. For a comprehensive guide on the specific stoichiometry and equipment requirements for each stage, please refer to the standardized synthesis steps outlined below.
- Perform oxime forming reaction on glyoxylic acid to obtain glyoxylic oxime formate.
- Conduct halogenation reaction on the acetaldoxime formic acid to generate the dihalogenated intermediate (Formula 1).
- Execute a [3+2] cyclization reaction between the halogenated intermediate and an unsaturated aldehyde/alkene to form the isoxazole ring.
- Apply Huang Minlon reduction conditions to convert the formyl group to a methyl group, yielding the final 3-halogenated-5,5-dimethylisoxazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this glyoxylic acid-based route offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the raw material portfolio; by replacing hazardous gases like isobutene and chlorine with stable liquids and solids, the facility's safety compliance costs are significantly reduced. This shift eliminates the need for specialized high-pressure storage tanks and complex gas scrubbing systems, thereby lowering the capital expenditure required for plant retrofitting or new construction. Furthermore, the ability to operate without complex organic catalysts means that solvent recycling streams remain cleaner, extending the lifecycle of expensive organic solvents and reducing the volume of hazardous waste requiring third-party disposal. These factors combine to create a manufacturing process that is not only safer but also economically more resilient against fluctuations in raw material pricing and regulatory pressures.
- Cost Reduction in Manufacturing: The elimination of expensive and difficult-to-recover organic acid-base catalysts directly translates to lower variable production costs. In traditional methods, the contamination of solvents by catalysts like N-methylaniline often renders the solvent unfit for reuse without energy-intensive distillation, whereas the inorganic bases used in this new method allow for simple aqueous extraction and solvent recovery. Additionally, the avoidance of high-boiling solvents like n-butyl acetate reduces the energy consumption associated with solvent stripping and product drying. The overall process efficiency is further enhanced by the high purity of the crude product, which often allows for direct use in subsequent steps without intermediate purification, effectively collapsing multiple unit operations into a streamlined workflow.
- Enhanced Supply Chain Reliability: Sourcing glyoxylic acid and common halogenating agents is far more reliable than securing bulk quantities of specialized gaseous reagents which may be subject to transport restrictions or seasonal availability issues. The use of commodity chemicals ensures that the supply chain is robust and less susceptible to disruptions caused by logistics constraints on hazardous materials. Moreover, the stability of the intermediates allows for greater flexibility in production scheduling; batches can be held at certain stages without significant degradation, providing supply chain planners with the buffer needed to manage demand spikes. This reliability is crucial for maintaining continuous production lines for downstream herbicide formulations.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard liquid-phase reactor technology rather than specialized gas-liquid contactors. This makes technology transfer from pilot scale to commercial production straightforward and low-risk. From an environmental perspective, the reduction in 'three wastes' aligns with increasingly stringent global regulations on industrial emissions. The minimized generation of hazardous byproducts and the use of recyclable solvents position manufacturers using this technology as leaders in sustainable chemistry, potentially qualifying them for green manufacturing incentives and improving their corporate social responsibility profiles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these nuances is critical for making informed decisions about process adoption and vendor qualification.
Q: How does this new synthesis route improve safety compared to traditional isobutene methods?
A: Traditional methods often utilize isobutene gas, which poses significant explosion and handling risks in large-scale industrial settings. The patented glyoxylic acid route operates primarily in the liquid phase using stable solvents like dichloromethane and methanol, eliminating the need for high-pressure gaseous reagents and drastically reducing potential safety hazards.
Q: What are the purity specifications achievable with this manufacturing process?
A: The process is designed to minimize impurity generation through controlled reaction conditions and simplified workup procedures. Experimental data from the patent indicates that the final 3-halogenated-5,5-dimethylisoxazole compound can be obtained with a content exceeding 95%, meeting the stringent quality requirements for high-performance herbicide synthesis.
Q: Does this method reduce environmental waste compared to prior art?
A: Yes, the method significantly reduces the generation of 'three wastes' (waste water, waste gas, and waste residue). By avoiding complex organic acid-base catalyst systems that are difficult to recover and utilizing solvents that are easier to manage, the overall environmental footprint of the production process is substantially lowered.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Halogenated-5,5-Dimethylisoxazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer, more efficient synthetic routes is critical for the long-term viability of the agrochemical sector. Our team of process chemists has extensively analyzed the glyoxylic acid pathway described in CN113372288A and possesses the technical expertise to scale this methodology from laboratory benchtops to full commercial production. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high purity specifications and rigorous QC labs standards required for pesticide intermediates are consistently met. Our facilities are equipped to handle the specific solvent systems and thermal conditions required for the Huang Minlon reduction, guaranteeing a supply of 3-halogenated-5,5-dimethylisoxazole that meets the highest industry standards.
We invite you to collaborate with us to optimize your supply chain for topramezone and related herbicides. By leveraging our manufacturing capabilities, you can secure a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your production goals are met with precision, safety, and economic efficiency.
