Advanced Nickel-Catalyzed Synthesis of 2-Cyclohexyl Benzamide for Commercial Scale-Up
Advanced Nickel-Catalyzed Synthesis of 2-Cyclohexyl Benzamide for Commercial Scale-Up
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the urgent need for more sustainable, cost-effective, and scalable synthetic routes. A pivotal advancement in this domain is detailed in Chinese Patent CN110724064B, which discloses a novel method for synthesizing 2-cyclohexyl substituted benzamide derivatives under nickel catalysis. This technology represents a significant departure from traditional cross-coupling strategies that often rely on pre-functionalized substrates and expensive noble metal catalysts. By leveraging direct C-H activation strategies, this patent offers a streamlined pathway to access valuable benzamide scaffolds that are ubiquitous in medicinal chemistry and agrochemical applications. For R&D directors and procurement managers alike, understanding the nuances of this nickel-catalyzed protocol is essential for optimizing supply chains and reducing the overall cost of goods sold (COGS) for complex drug candidates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ortho-alkylated benzamides has been fraught with synthetic inefficiencies and economic bottlenecks. Traditional approaches often necessitate the installation of specific directing groups to achieve regioselectivity, which adds multiple synthetic steps and generates substantial waste. For instance, prior art methods, such as those described by W.F. Song et al., utilize quinoline as a directing group to coordinate with the metal center for ortho-C-H bond activation. 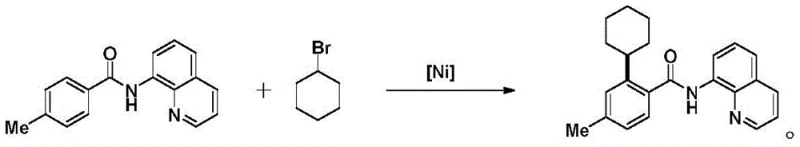 Similarly, other established protocols employ pyridine-based directing groups in conjunction with cobalt catalysts to facilitate cross-dehydrogenative coupling.
Similarly, other established protocols employ pyridine-based directing groups in conjunction with cobalt catalysts to facilitate cross-dehydrogenative coupling. 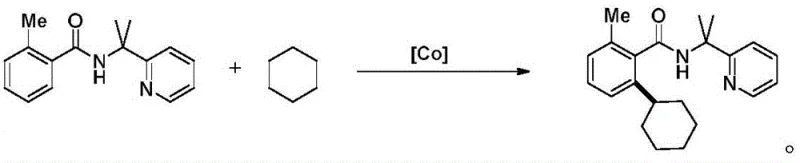 These legacy methods suffer from significant drawbacks: the starting materials must be pre-activated with these directing groups, which are often toxic or difficult to remove later in the synthesis. Furthermore, the reliance on specific transition metals like cobalt or the use of toxic halides as reactants introduces safety hazards and complicates the purification process, leading to lower overall throughput and higher environmental compliance costs for manufacturers.
These legacy methods suffer from significant drawbacks: the starting materials must be pre-activated with these directing groups, which are often toxic or difficult to remove later in the synthesis. Furthermore, the reliance on specific transition metals like cobalt or the use of toxic halides as reactants introduces safety hazards and complicates the purification process, leading to lower overall throughput and higher environmental compliance costs for manufacturers.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the method disclosed in patent CN110724064B introduces a remarkably elegant solution by utilizing benzamide directly as the substrate without any pre-installed directing groups. The core innovation lies in the use of a廉价 (inexpensive) nickel catalyst system that activates the ortho-C-H bond of the benzamide ring directly against cyclohexane. 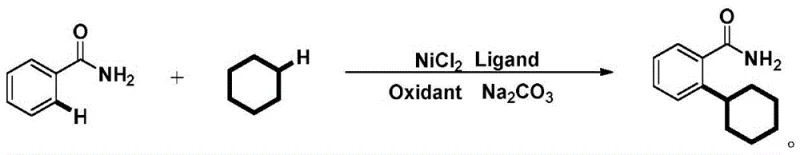 This approach not only simplifies the molecular architecture of the starting materials but also utilizes cyclohexane dually as both the solvent and the alkylating reagent. This dual functionality drastically reduces the volume of organic solvents required and eliminates the need for separate alkyl halide reagents. The reaction proceeds under oxidative conditions using common oxidants like tert-butyl peroxide, allowing for a direct, atom-economical transformation that bypasses the need for halogenated intermediates entirely, thereby offering a cleaner and more robust synthetic route for industrial application.
This approach not only simplifies the molecular architecture of the starting materials but also utilizes cyclohexane dually as both the solvent and the alkylating reagent. This dual functionality drastically reduces the volume of organic solvents required and eliminates the need for separate alkyl halide reagents. The reaction proceeds under oxidative conditions using common oxidants like tert-butyl peroxide, allowing for a direct, atom-economical transformation that bypasses the need for halogenated intermediates entirely, thereby offering a cleaner and more robust synthetic route for industrial application.
Mechanistic Insights into Nickel-Catalyzed C-H Alkylation
The mechanistic underpinning of this transformation involves a sophisticated interplay between the nickel center, the ligand environment, and the oxidant to facilitate selective C-H bond cleavage. The cycle likely initiates with the coordination of the nickel(II) species, such as nickel chloride, with the carbonyl oxygen of the benzamide, acting as a weak directing moiety to position the metal proximal to the ortho-position. Upon oxidation by agents like tert-butyl peroxide or potassium persulfate, a high-valent nickel species is generated, which is capable of abstracting a hydrogen atom from the ortho-position of the aromatic ring or activating the C-H bond of the cyclohexane solvent via a radical pathway. The presence of bulky phosphine ligands, such as tricyclohexylphosphine, plays a critical role in stabilizing the active catalytic species and preventing the formation of inactive nickel black precipitates. This ligand acceleration effect ensures that the catalytic turnover number remains high throughout the 18-hour reaction window at elevated temperatures of 120-135°C.
From an impurity control perspective, this mechanism offers distinct advantages over radical halogenation pathways. Because the reaction avoids the use of alkyl halides, there is no risk of generating halogenated byproducts or dealing with the corrosive hydrogen halide waste streams typical of Friedel-Crafts type alkylations. The selectivity for the ortho-position is governed by the chelation assistance of the amide group, which effectively outcompetes statistical radical attack on other positions of the benzene ring. Furthermore, the use of sodium carbonate as a mild base helps to neutralize any acidic byproducts formed during the oxidation cycle, maintaining a stable pH environment that prevents the hydrolysis of the sensitive amide bond. This results in a crude product profile that is significantly cleaner, reducing the burden on downstream purification units and minimizing the loss of valuable material during column chromatography or recrystallization steps.
How to Synthesize 2-Cyclohexyl Benzamide Efficiently
Implementing this synthesis on a pilot or production scale requires strict adherence to the optimized parameters outlined in the patent examples to ensure reproducibility and safety. The process begins with the precise charging of the reactor with benzamide, followed by the sequential addition of the nickel source, the specific ligand (preferably tricyclohexylphosphine for highest yields), the oxidant, and the base. The choice of oxidant is critical; while tert-butyl peroxide provides the highest yields (up to 71%), alternatives like tert-butyl hydroperoxide or potassium persulfate can be employed depending on safety protocols and availability. The reaction mixture is then heated to a temperature range of 120-135°C, typically maintained for 18 hours to ensure complete conversion of the starting material.
- Charge the reactor with benzamide, nickel chloride catalyst, specific phosphine or nitrogen ligands, oxidant, and sodium carbonate base.
- Add cyclohexane as both the solvent and the alkylating reagent, then heat the mixture to 120-135°C for approximately 18 hours.
- Upon completion, extract the reaction mixture with ethyl acetate, wash, dry, concentrate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this nickel-catalyzed protocol presents a compelling value proposition centered on cost reduction and operational simplicity. The elimination of pre-functionalized starting materials means that the supply chain can rely on commodity chemicals like benzamide and cyclohexane, which are available in bulk quantities from numerous global suppliers, thereby mitigating the risk of single-source dependency. The removal of toxic halides and expensive directing groups from the bill of materials (BOM) directly translates to a lower raw material cost per kilogram of the final API intermediate. Additionally, the simplified workup procedure, which involves standard extraction and distillation rather than complex aqueous quenching of reactive organometallics, reduces the demand on utility infrastructure and shortens the batch cycle time, allowing for increased throughput in existing manufacturing facilities.
- Cost Reduction in Manufacturing: The substitution of precious or semi-precious metals like palladium or cobalt with abundant nickel significantly lowers the catalyst cost, which is often a major driver of expense in fine chemical synthesis. Moreover, the use of cyclohexane as both solvent and reagent eliminates the need to purchase and handle separate alkylating agents, further compressing the material costs. The high atom economy of this direct C-H functionalization means less waste is generated per unit of product, reducing the costs associated with waste disposal and environmental compliance. By streamlining the synthetic sequence and removing the steps required to install and remove directing groups, the overall processing time and labor costs are substantially reduced, enhancing the economic viability of the project.
- Enhanced Supply Chain Reliability: Relying on widely available commodity feedstocks such as benzamide and cyclohexane ensures a robust and resilient supply chain that is less susceptible to market volatility compared to specialized heterocyclic building blocks. The robustness of the nickel catalyst system, which tolerates a variety of ligands and oxidants, provides flexibility in sourcing; if one specific ligand faces supply constraints, alternatives like triphenylphosphine can be utilized with acceptable yields. This flexibility allows procurement teams to negotiate better terms with vendors and maintain continuous production schedules even during periods of raw material scarcity. The stability of the reagents also simplifies storage and logistics, as there is no need for specialized containment for moisture-sensitive or highly toxic intermediates.
- Scalability and Environmental Compliance: The process operates under relatively mild oxidative conditions without the generation of hazardous halogenated waste, aligning perfectly with modern green chemistry principles and stringent environmental regulations. The absence of toxic heavy metals in the catalyst system (compared to lead or mercury based processes) and the use of nickel, which is easier to recover and recycle, simplifies the environmental impact assessment for new facilities. The straightforward isolation procedure involving ethyl acetate extraction and vacuum distillation is easily adaptable to large-scale reactors, ensuring that the process can be scaled from kilograms to multi-ton production without encountering significant engineering hurdles. This scalability ensures that the technology can meet the growing demand for high-purity pharmaceutical intermediates without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nickel-catalyzed synthesis route. These insights are derived directly from the experimental data and technical specifications provided in patent CN110724064B, offering clarity on reaction optimization and potential challenges. Understanding these details is crucial for process chemists evaluating the feasibility of this route for their specific pipeline projects.
Q: What are the primary advantages of this nickel-catalyzed method over traditional cobalt or palladium methods?
A: This method eliminates the need for expensive directing groups like quinoline or pyridine, uses inexpensive nickel instead of cobalt or palladium, and utilizes cyclohexane as both solvent and reactant, significantly simplifying the process and reducing raw material costs.
Q: What represent the optimal reaction conditions for high yield?
A: Optimal results are achieved using nickel chloride with tricyclohexylphosphine as the ligand and tert-butyl peroxide as the oxidant at temperatures between 120°C and 135°C, yielding up to 71% of the target product.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process uses cheap, readily available raw materials, operates under relatively safe conditions without toxic halides, and involves a simple post-processing workflow involving extraction and distillation, making it highly scalable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Cyclohexyl Benzamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in reshaping the economics of pharmaceutical manufacturing. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this nickel-catalyzed protocol are successfully translated into robust industrial processes. We are committed to delivering high-purity 2-cyclohexyl benzamide derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality assurance ensures that every batch delivered to our global partners adheres to the highest industry standards, facilitating seamless integration into your downstream synthesis workflows.
We invite you to collaborate with us to leverage this cost-effective synthetic route for your upcoming projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, allowing you to make informed decisions that drive efficiency and profitability in your supply chain. Let us be your strategic partner in navigating the complexities of modern chemical manufacturing.
