Scalable Synthesis of Norepinephrine Bitartrate: A Technical Breakthrough for Global API Manufacturing
Scalable Synthesis of Norepinephrine Bitartrate: A Technical Breakthrough for Global API Manufacturing
The global demand for critical care medications, particularly vasopressors like norepinephrine bitartrate, necessitates robust and reliable supply chains capable of delivering high-purity active pharmaceutical ingredients (APIs) without interruption. Patent CN114394907A introduces a transformative preparation method that addresses longstanding inefficiencies in the synthesis of this life-saving drug. By utilizing 3,4-dibenzyloxyacetophenone as a stable starting material, the invention circumvents the oxidation issues plaguing earlier methodologies while ensuring high yields and exceptional product quality. This technical advancement represents a significant leap forward for any reliable API intermediate supplier seeking to optimize their manufacturing portfolio. The protocol eliminates the need for hazardous reagents and streamlines the reaction sequence, thereby offering substantial cost reduction in pharmaceutical manufacturing while maintaining rigorous quality standards required by international regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of norepinephrine and its salts has been fraught with significant technical and safety challenges that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Traditional routes often rely on starting materials such as chloroacetyl catechol or unprotected catechol derivatives, which are inherently unstable and prone to rapid oxidation during processing. This instability not only compromises the purity of the final product but also necessitates stringent and costly inert atmosphere controls throughout the synthesis. Furthermore, legacy methods frequently employ highly toxic or potentially explosive reagents, creating severe occupational health and safety risks for manufacturing personnel. The cumulative effect of these drawbacks is a lengthy, cumbersome process with low overall yields, making it economically unviable for large-scale industrial production and creating vulnerabilities in the supply chain for essential medicines.
The Novel Approach
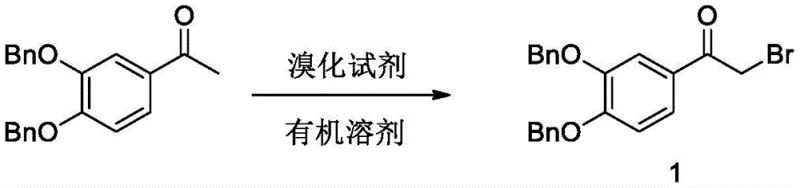
The methodology outlined in patent CN114394907A offers a sophisticated solution by employing a benzyl-protected strategy that fundamentally alters the reaction landscape. By initiating the synthesis with 3,4-dibenzyloxyacetophenone, the process effectively shields the sensitive phenolic hydroxyl groups from premature oxidation, ensuring the integrity of the molecular scaffold throughout the early reaction stages. This protective approach allows for the use of milder, more manageable reagents such as copper bromide for bromination, replacing dangerous elemental bromine or chlorinating agents. The subsequent steps are designed for operational simplicity, utilizing common organic solvents and avoiding extreme conditions. This streamlined workflow not only enhances the safety profile of the manufacturing plant but also drastically simplifies post-reaction workup procedures, resulting in a cleaner crude product that requires less intensive purification efforts before the final crystallization.
Mechanistic Insights into Catalytic Hydrogenation and Chiral Resolution
A critical component of this novel synthesis is the strategic application of catalytic hydrogenation, which serves a dual purpose in the transformation of intermediate 2 to intermediate 3. In this pivotal step, the reaction mixture containing the amino-ketone precursor is subjected to hydrogen gas in the presence of a palladium on carbon (Pd/C) catalyst. Mechanistically, this single operation achieves two distinct chemical transformations simultaneously: the reduction of the carbonyl group to a secondary alcohol and the hydrogenolysis of the benzyl ether protecting groups. This convergence of reactions is highly efficient, as it removes the need for separate deprotection steps that would otherwise require additional reagents, solvents, and time. The use of Pd/C is particularly advantageous due to its high activity and selectivity, allowing the reaction to proceed smoothly at moderate temperatures between 30°C and 60°C and pressures ranging from 0.3 MPa to 1.0 MPa, which are easily achievable in standard industrial hydrogenation reactors.
Following the formation of the racemic amine, the process employs a classical yet highly effective chiral resolution strategy to isolate the biologically active R-enantiomer. The use of D-tartaric acid as a resolving agent facilitates the formation of diastereomeric salts, which exhibit different solubility properties in alcoholic solvents. This difference allows for the selective crystallization of the desired isomer, effectively purifying the compound while establishing the correct stereochemistry required for pharmacological activity. The subsequent conversion to the final bitartrate salt using L-tartaric acid ensures the formation of the stable, pharmaceutically acceptable form of the drug. This meticulous control over stereochemistry is paramount for meeting the stringent purity specifications demanded by modern pharmacopeias, ensuring that the final API is free from harmful enantiomeric impurities that could lead to adverse patient outcomes.
How to Synthesize Norepinephrine Bitartrate Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize yield and minimize impurity formation. The process begins with the careful bromination of the protected ketone, followed by amination via the urotropine method, which avoids the handling of gaseous ammonia. Detailed operational guidelines regarding solvent ratios, temperature gradients, and stirring speeds are critical for reproducibility. For R&D teams looking to implement this technology, understanding the nuances of the hydrogenation step is vital, as catalyst loading and hydrogen pressure directly influence the rate of debenzylation. The detailed standardized synthesis steps see the guide below for a comprehensive breakdown of the experimental procedure.
- Brominate 3,4-dibenzyloxyacetophenone using copper bromide in dichloromethane to form the alpha-bromo ketone intermediate.
- React the bromo-intermediate with urotropine followed by acid hydrolysis to introduce the amine functionality.
- Perform catalytic hydrogenation using Pd/C to simultaneously reduce the carbonyl group and remove benzyl protecting groups.
- Resolve the racemic amine using D-tartaric acid to isolate the desired R-enantiomer.
- Form the final salt by reacting the resolved amine with L-tartaric acid in a methanol-water system.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers compelling advantages that directly address the pain points of procurement managers and supply chain directors. The shift away from expensive and difficult-to-source starting materials to readily available commodity chemicals significantly lowers the raw material cost base. Furthermore, the elimination of toxic and explosive reagents reduces the regulatory burden and insurance costs associated with hazardous chemical storage and handling. This translates into a more resilient supply chain that is less susceptible to disruptions caused by environmental regulations or safety incidents. The simplified workflow also means shorter cycle times, allowing manufacturers to respond more agilely to market demand fluctuations.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the consolidation of reaction steps and the use of inexpensive catalysts. By combining the reduction and deprotection steps into a single hydrogenation operation, the process eliminates the need for separate reagents and solvents, thereby reducing utility consumption and waste disposal costs. Additionally, the high yield reported in the patent examples indicates minimal material loss, which maximizes the output per batch and improves the overall cost efficiency of the production line. These factors collectively contribute to a lower cost of goods sold (COGS), providing a competitive edge in the pricing of the final API.
- Enhanced Supply Chain Reliability: The reliance on stable, protected intermediates mitigates the risk of raw material degradation during storage and transport. Unlike unprotected catechols that require specialized cold chain logistics to prevent oxidation, 3,4-dibenzyloxyacetophenone is robust and shelf-stable. This stability ensures a consistent supply of quality starting materials, reducing the likelihood of production delays caused by substandard inputs. Moreover, the use of common solvents like methanol and ethanol ensures that solvent supply remains secure even during global shortages of specialty chemicals, further bolstering supply chain continuity.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of heavy metal contaminants in the final product simplify the path to regulatory approval and environmental compliance. The process generates less hazardous waste compared to traditional methods, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing facility. This eco-friendly profile is increasingly important for pharmaceutical companies aiming to meet sustainability goals. The scalability of the hydrogenation and crystallization steps ensures that the process can be seamlessly transferred from pilot scale to multi-ton commercial production without significant re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these aspects is crucial for stakeholders evaluating the feasibility of adopting this technology for their own manufacturing operations.
Q: How does this new synthesis route improve safety compared to conventional methods?
A: Unlike traditional routes that utilize highly toxic or potentially explosive reagents like chloroacetyl chloride on unprotected catechols, this patented method employs mild bromination with copper bromide and avoids hazardous intermediates, significantly enhancing operational safety and reducing EHS compliance burdens.
Q: What are the yield advantages of the catalytic hydrogenation step?
A: The process utilizes a efficient one-pot catalytic hydrogenation strategy using Pd/C that achieves dual functionality: reducing the ketone to an alcohol and removing benzyl protecting groups simultaneously. This consolidation of steps minimizes material loss and simplifies downstream processing, leading to superior overall yields compared to multi-step deprotection sequences.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction conditions are specifically optimized for scalability, operating at moderate temperatures (20-60°C) and manageable hydrogen pressures (0.3-1.0 MPa). The use of cheap, readily available starting materials and the elimination of complex purification steps make it highly conducive to commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Norepinephrine Bitartrate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner for the production of life-saving cardiovascular medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of global pharmaceutical markets. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of norepinephrine bitartrate meets the highest international standards. Our state-of-the-art facilities are equipped to handle the specific catalytic hydrogenation and chiral resolution processes required by this advanced synthesis route, delivering consistent quality and reliability.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis can benefit your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages specific to your volume needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions about securing a sustainable and cost-effective supply of this essential API intermediate.
