Optimizing Prisamod Production: A Convergent Synthetic Strategy for Commercial Scale-Up
Optimizing Prisamod Production: A Convergent Synthetic Strategy for Commercial Scale-Up
The pharmaceutical landscape for immunosuppressants is evolving rapidly, with S1P1 receptor modulators like Prisamod emerging as potent candidates for treating rheumatoid arthritis. As detailed in patent CN111087357A, a significant technological breakthrough has been achieved in the manufacturing of this active pharmaceutical ingredient (API). The disclosed method shifts away from traditional, inefficient linear syntheses toward a highly optimized convergent strategy. This innovation addresses critical pain points in modern drug manufacturing, specifically focusing on yield enhancement, impurity control, and environmental sustainability. For R&D directors and procurement specialists, understanding this shift is vital for securing a reliable API intermediate supplier capable of meeting stringent quality and volume demands.
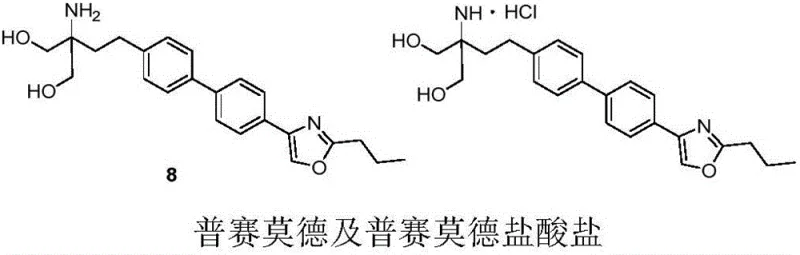
The core of this technological advancement lies in the re-engineering of the synthetic pathway. Historically, the preparation of Prisamod relied on a linear sequence starting from biphenyl, involving nine distinct steps including Friedel-Crafts acylation, multiple reductions, and cyclizations. This legacy approach suffered from a cumulative total yield of merely 9.9%, creating substantial bottlenecks in supply continuity. Furthermore, the reliance on column chromatography for purification at various stages introduced variability and excessive solvent consumption. The new methodology described in the patent fundamentally alters this paradigm by decoupling the synthesis into two parallel streams that converge at a late stage, thereby maximizing efficiency and minimizing resource depletion.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The conventional linear synthesis of Prisamod presents severe challenges for industrial scalability. The process initiates with biphenyl and traverses a cumbersome nine-step sequence involving harsh reagents such as strong Lewis acids and extensive use of organic solvents. A critical drawback is the low overall yield of 9.9%, which necessitates processing large volumes of starting materials to obtain small quantities of the final API, driving up raw material costs significantly. Moreover, the requirement for column chromatography purification not only slows down production throughput but also generates massive amounts of silica gel waste, complicating disposal and increasing the environmental footprint. The heavy reliance on anhydrous Lewis acids, with consumption rates reaching up to 19.2 kilograms per kilogram of product, exacerbates safety hazards and waste treatment costs, making the process economically unviable for large-scale commercialization without significant optimization.
The Novel Approach
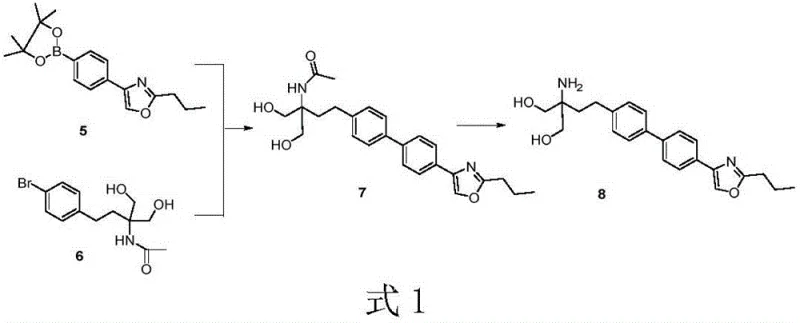
In stark contrast, the novel convergent synthesis route outlined in the patent achieves a remarkable total yield of 31.6%, representing a more than threefold improvement over the traditional method. This strategy involves the independent preparation of two key fragments: the boronate ester intermediate (Compound 5) and the bromide intermediate (Compound 6). These fragments are then united via a robust palladium-catalyzed Suzuki coupling reaction to form the biaryl core (Compound 7). This convergence allows for the optimization of each fragment synthesis separately, ensuring high purity before the final assembly. Crucially, the entire process eliminates the need for column chromatography, relying instead on crystallization and extraction techniques that are inherently more scalable and cost-effective. The reduction in Lewis acid usage to approximately 1-2 kilograms per kilogram of product further underscores the green chemistry credentials of this new approach.
Mechanistic Insights into Pd-Catalyzed Suzuki Coupling
The heart of this synthetic innovation is the Suzuki-Miyaura cross-coupling reaction between the aryl bromide (Compound 6) and the aryl boronic ester (Compound 5). This transformation constructs the critical biphenyl linkage found in the Prisamod structure. The patent highlights a meticulous optimization of the catalytic system, identifying dibromo-bis(tri-tert-butylphosphine)dipalladium(I) (Pd-Dimer) as the superior catalyst. Unlike traditional phosphine ligands that may degrade under harsh conditions, the bulky tri-tert-butylphosphine ligands stabilize the palladium center, facilitating efficient oxidative addition and reductive elimination cycles even in protic solvents. The reaction proceeds effectively with catalyst loadings as low as 0.1 to 0.6 mol%, demonstrating high turnover numbers that are essential for minimizing residual metal impurities in the final pharmaceutical product.
Furthermore, the choice of solvent system plays a pivotal role in the reaction's success and environmental profile. The process utilizes water or ethanol-water mixtures as the primary reaction medium, a significant departure from the toxic organic solvents typically required for such couplings. This aqueous compatibility not only reduces volatile organic compound (VOC) emissions but also simplifies the workup procedure, as the product can often be isolated via simple filtration or extraction. The presence of inorganic bases such as potassium carbonate or potassium fluoride is essential to activate the boron species, forming the reactive boronate complex that undergoes transmetallation. This mechanistic precision ensures that side reactions, such as homocoupling or deboronation, are minimized, resulting in a high-purity intermediate that requires minimal downstream purification.
How to Synthesize Prisamod Efficiently
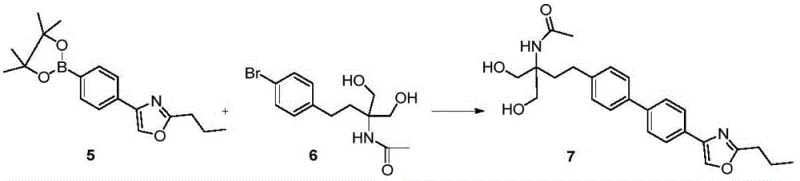
The synthesis of Prisamod via this convergent route is designed for operational simplicity and robustness, making it ideal for transfer from laboratory to pilot plant. The process begins with the parallel synthesis of the two key building blocks. Intermediate 5 is derived from bromobenzene through a sequence of acylation, esterification, oxazole ring formation, and finally, Miyaura borylation. Simultaneously, Intermediate 6 is prepared either from p-bromophenethyl alcohol or benzene, involving alkylation with diethyl acetamidomalonate followed by reduction and bromination. Once these high-purity fragments are secured, they are subjected to the optimized Suzuki coupling conditions. The detailed standardized synthesis steps, including specific temperature profiles, stoichiometric ratios, and isolation protocols, are critical for reproducibility and are outlined in the technical guide below.
- Synthesize key intermediate 5 (boronate ester) from bromobenzene via acylation, esterification, cyclization, and borylation.
- Prepare intermediate 6 (bromide) from benzene or p-bromophenethyl alcohol through alkylation and reduction steps.
- Perform palladium-catalyzed Suzuki coupling of intermediates 5 and 6 in aqueous media to form compound 7, followed by hydrolysis to Prisamod.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers transformative benefits that extend beyond mere chemical yield. The shift from a linear to a convergent architecture fundamentally de-risks the supply chain by allowing for the stocking of stable intermediates (Compounds 5 and 6) rather than relying on a single long linear chain where a failure at step one halts the entire process. This modularity enhances supply continuity and allows for flexible production scheduling. Additionally, the elimination of column chromatography removes a major bottleneck in manufacturing throughput, enabling faster batch turnover times and reducing the dependency on specialized silica gel supplies, which can be subject to market volatility. The ability to use water as a primary solvent also drastically cuts down on solvent recovery costs and hazardous waste disposal fees, contributing to a leaner and more sustainable cost structure.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the dramatic increase in overall yield from 9.9% to 31.6%. This tripling of efficiency means that for the same amount of final product, the consumption of raw materials is reduced by approximately two-thirds. Furthermore, the drastic reduction in Lewis acid usage—from 19.2 kg/kg to roughly 1-2 kg/kg—lowers reagent costs and minimizes the expense associated with neutralizing and disposing of acidic waste streams. The removal of column chromatography also eliminates the recurring cost of purchasing large quantities of silica gel and the associated solvent volumes required for elution, leading to substantial operational expenditure (OpEx) savings.
- Enhanced Supply Chain Reliability: The convergent nature of the synthesis provides inherent redundancy and flexibility. Since the two main branches of the synthesis (Intermediate 5 and Intermediate 6) can be produced independently, any disruption in one supply line does not necessarily halt the entire operation if inventory buffers are managed correctly. The use of commodity chemicals like bromobenzene and benzene as starting materials ensures that the supply chain is not dependent on exotic or single-source precursors. Moreover, the robustness of the aqueous Suzuki coupling reduces the risk of batch failures due to moisture sensitivity, a common issue in traditional organometallic chemistry, thereby ensuring consistent delivery schedules to downstream formulation teams.
- Scalability and Environmental Compliance: Scaling chemical processes often magnifies safety and environmental issues, but this route is designed with green chemistry principles in mind. The use of water as a solvent significantly reduces the fire hazard and VOC emissions associated with large-scale organic synthesis. The simplified purification protocol, which relies on crystallization rather than chromatography, is inherently easier to scale from grams to tons without losing efficiency. This alignment with environmental regulations reduces the regulatory burden and potential fines associated with waste management, making the facility more attractive to investors and partners who prioritize ESG (Environmental, Social, and Governance) criteria in their supply chain audits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Prisamod intermediates. These answers are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a transparent view of the technology's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing infrastructure.
Q: What is the primary advantage of the new Prisamod synthesis route?
A: The new convergent route increases total yield from 9.9% to 31.6% and eliminates the need for column chromatography, significantly reducing production costs and environmental waste.
Q: Which catalyst system is preferred for the key coupling step?
A: Dibromo-bis(tri-tert-butylphosphine)dipalladium(I) (Pd-Dimer) is the preferred catalyst, operating efficiently at low loadings (0.1-0.6 mol%) in water or ethanol-water mixtures.
Q: How does this method improve environmental compliance?
A: By reducing Lewis acid consumption from 19.2 kg/kg product to roughly 1-2 kg/kg and removing silica gel purification, the process drastically lowers hazardous waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prisamod Supplier
The transition to this advanced convergent synthesis route represents a significant opportunity for pharmaceutical companies to secure a more stable and cost-effective supply of Prisamod intermediates. NINGBO INNO PHARMCHEM stands at the forefront of this technological adoption, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of palladium-catalyzed reactions and aqueous processing, ensuring that every batch meets stringent purity specifications. With our rigorous QC labs and commitment to process safety, we provide a partnership model that mitigates risk and accelerates your time-to-market for this promising immunosuppressant therapy.
We invite you to engage with our technical procurement team to discuss how this optimized route can benefit your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on the most efficient and reliable chemical foundation available.
