Advanced Selective Oxidation Strategy for Commercial Linaclotide Production
Introduction to Next-Generation Linaclotide Manufacturing
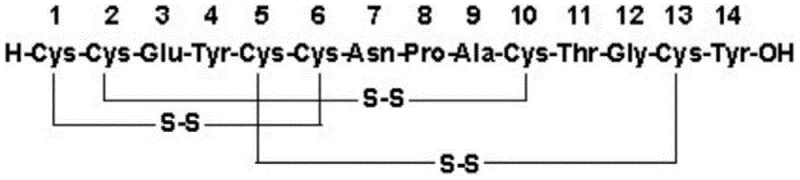
The pharmaceutical landscape for treating chronic idiopathic constipation and IBS-C has been revolutionized by Linaclotide, a 14-amino acid polypeptide agonist of the GC-C receptor. However, the commercial viability of this potent therapeutic has historically been constrained by the immense synthetic challenge posed by its structure, specifically the requirement to form three specific pairs of disulfide bonds among six cysteine residues. Patent CN106892968B introduces a groundbreaking synthesis method that addresses these bottlenecks through a strategy of complete selective formation of disulfide bonds. This technical breakthrough shifts the paradigm from stochastic oxidation to a controlled, stepwise assembly, ensuring that the correct connectivity is established with high fidelity. By integrating solid-phase peptide synthesis (SPPS) with sequential orthogonal deprotection and oxidation, this methodology offers a robust pathway for producing high-purity Linaclotide suitable for global regulatory standards.
For procurement and supply chain leaders, the implications of this patent are profound. Traditional methods often suffer from low yields due to the formation of scrambled isomers, which necessitates expensive and loss-prone purification steps. The approach detailed in CN106892968B mitigates these risks by enforcing the correct structural topology at each stage of the synthesis. This not only enhances the final product quality but also stabilizes the supply chain by reducing batch-to-batch variability. As a reliable linaclotide supplier, understanding these mechanistic nuances is critical for evaluating the long-term sustainability and cost-effectiveness of API sourcing strategies in the competitive gastrointestinal therapeutic market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to Linaclotide synthesis have predominantly relied on either fully random oxidation or semi-selective strategies that fail to completely eliminate isomeric impurities. In conventional Method 1, as cited in the background of the patent, a single protecting group such as Trt is used for all cysteine residues, followed by a one-step liquid-phase oxidation. While operationally simple, this lack of differentiation leads to a statistical distribution of disulfide pairings, resulting in a complex mixture of mismatched isomers that are structurally similar to the target molecule. Separating these isomers requires extensive preparative HPLC, which drastically reduces overall yield and increases solvent consumption. Furthermore, Method 2 attempts a semi-selective approach using Trt and Acm groups, but literature indicates that iodine oxidation steps can severely reduce crude peptide yield. Other prior art patents describe one-step oxidation systems using GHS/GSSH or cysteine/DMSO buffers, yet none successfully avoid the thermodynamic trap of disulfide scrambling, leading to inconsistent product quality that is highly dependent on external variables like temperature and buffer composition.
The Novel Approach
The innovative methodology disclosed in CN106892968B overcomes these limitations by employing a triple-orthogonal protection strategy involving Mmt, Dpm, and Methyl groups. This allows for the discrete and sequential formation of each disulfide bond, effectively guiding the peptide folding pathway towards the native structure. Unlike the chaotic environment of one-step oxidation, this process isolates the formation of the first bond on the solid phase, where steric constraints and local concentration effects can be leveraged to favor the correct pairing. Subsequent steps utilize specific chemical triggers—mild acid for Mmt removal and strong acid cleavage for Dpm removal—to expose only the intended cysteine pairs for oxidation. This logical progression eliminates the possibility of forming incorrect cross-links early in the synthesis, thereby preserving the integrity of the linear precursor. The result is a synthesis route that is not only chemically superior in terms of purity but also operationally more stable, reducing the risk of batch failures during commercial scale-up of complex peptide intermediates.
Mechanistic Insights into Selective Disulfide Bond Formation
The core of this technological advancement lies in the precise manipulation of cysteine side-chain protecting groups to achieve orthogonality. The synthesis begins with the assembly of the linear peptide on a Wang or 2-chloro resin, where Cys residues at positions 1 and 6 are protected with 4-methoxytrityl (Mmt), positions 2 and 10 with diphenylmethyl (Dpm), and positions 5 and 13 with Methyl (Me). The first critical mechanistic step involves the selective removal of the acid-labile Mmt groups using a dilute TFA/DCM solution (1-5% TFA). This mild condition leaves the Dpm and Methyl groups intact, exposing only the first pair of thiols. Subsequent oxidation with N-Chlorosuccinimide (NCS) on the solid phase forms the first disulfide ring with high efficiency. This solid-phase oxidation is advantageous as it prevents intermolecular aggregation, a common issue in solution-phase reactions of hydrophobic peptides. The resin-bound intermediate is then subjected to a cleavage cocktail containing high concentrations of TFA, thioanisole, and water. This harsh environment serves a dual purpose: it cleaves the peptide from the resin and simultaneously removes the Dpm protecting groups, exposing the second pair of cysteines while the first ring remains stable.
The final and perhaps most elegant mechanistic feature is the formation of the third disulfide bond. The remaining cysteine residues at positions 5 and 13 are protected by Methyl groups, which are typically resistant to standard acidic cleavage. Instead of using hazardous heavy metal catalysts or harsh chemical demethylating agents, the patent employs an enzymatic strategy using Horseradish Peroxidase (HRP). In this step, the enzyme facilitates the simultaneous oxidative demethylation and coupling of the thiols. This biocatalytic approach operates under mild physiological pH conditions (e.g., phosphate buffer pH 6), which preserves the stereochemical integrity of the sensitive amino acid residues. The use of HRP not only simplifies the workflow by combining deprotection and oxidation into a single unit operation but also aligns with green chemistry principles by avoiding toxic reagents. This mechanistic sophistication ensures that the final product achieves purity levels exceeding 99.5%, as demonstrated in the patent examples, making it ideal for high-purity API intermediate manufacturing where impurity profiles are strictly regulated.
How to Synthesize Linaclotide Efficiently
The synthesis of Linaclotide via this selective pathway requires precise control over reaction conditions and reagent stoichiometry to maximize the benefits of the orthogonal protection strategy. The process integrates solid-phase peptide synthesis (SPPS) techniques with solution-phase enzymatic transformations, demanding a seamless transition between heterogeneous and homogeneous reaction environments. Operators must carefully monitor the deprotection endpoints, particularly the colorimetric change associated with Mmt removal, to ensure complete exposure of the reactive thiols before oxidation. The following guide outlines the critical operational phases derived from the patent data, providing a framework for implementing this high-yield route in a GMP-compliant facility. For detailed standard operating procedures and specific parameter optimization, please refer to the technical guide below.
- Synthesize the linear peptide precursor resin using Fmoc-SPPS with orthogonal protecting groups: Cys(1,6)-Mmt, Cys(2,10)-Dpm, and Cys(5,13)-Me.
- Perform selective deprotection of Mmt groups on-resin followed by solid-phase oxidation with NCS to form the first disulfide bond.
- Cleave the peptide from resin using TFA cocktail to remove Dpm groups, followed by liquid-phase oxidation to form the second disulfide bond.
- Execute simultaneous demethylation and oxidative coupling of the third disulfide bond using Horseradish Peroxidase to obtain final Linaclotide.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of the synthesis method described in CN106892968B offers substantial strategic advantages for organizations managing the supply of gastrointestinal therapeutics. The primary value driver is the drastic simplification of the purification train. By preventing the formation of disulfide mismatched isomers at the source, the process eliminates the need for extensive recycling of off-spec material and reduces the load on preparative chromatography columns. This directly translates to cost reduction in peptide manufacturing, as solvent usage, column life, and processing time are significantly minimized. Furthermore, the reliance on robust chemical reagents like NCS and commercially available enzymes reduces dependency on exotic or volatile catalysts, enhancing supply chain reliability. The process stability ensures that lead times for high-purity peptide intermediates can be consistently met, mitigating the risk of stockouts that often plague complex peptide supply chains.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven by the elimination of expensive heavy metal catalysts and the reduction of purification losses. Traditional methods often require multiple rounds of HPLC to separate isomers, which is a major cost center in peptide production. By achieving high crude purity through selective bond formation, the overall yield of the process is maximized, effectively lowering the cost of goods sold (COGS). Additionally, the use of methyl-protected cysteine for the final step utilizes cheaper raw materials compared to more complex protecting groups, further optimizing the bill of materials. The enzymatic final step also reduces waste disposal costs associated with heavy metal remediation, contributing to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: Supply continuity is critical for API manufacturers, and this method enhances reliability by reducing process sensitivity. Random oxidation methods are notoriously sensitive to trace metals, oxygen levels, and pH fluctuations, leading to batch-to-batch variability. In contrast, the stepwise selective approach isolates variables, making the process more robust and predictable. The reagents used, such as TFA, DCM, and NCS, are commodity chemicals with stable global supply chains, reducing the risk of raw material shortages. This stability allows for more accurate production planning and inventory management, ensuring that downstream drug product manufacturers receive consistent quality material without unexpected delays.
- Scalability and Environmental Compliance: Scaling peptide synthesis from grams to kilograms often exposes hidden inefficiencies, but this protocol is designed with scalability in mind. The solid-phase steps utilize standard SPPS equipment, while the liquid-phase oxidations are performed in conventional reactors without the need for specialized high-pressure or cryogenic setups. The replacement of harsh chemical oxidants with Horseradish Peroxidase in the final step significantly improves the environmental profile of the synthesis. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the environmental footprint of the manufacturing site. The process generates less hazardous waste, simplifying compliance with increasingly stringent environmental regulations and supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this selective oxidation technology. These insights are derived directly from the experimental data and claims within Patent CN106892968B, providing clarity on the feasibility and advantages of this route for potential partners and stakeholders. Understanding these details is essential for making informed decisions about technology transfer and long-term supply agreements.
Q: How does the selective oxidation method improve Linaclotide purity compared to traditional one-step oxidation?
A: Traditional one-step oxidation often results in a mixture of mismatched disulfide isomers due to random pairing of the six cysteine residues. The method in CN106892968B utilizes orthogonal protecting groups (Mmt, Dpm, Me) to form disulfide bonds sequentially and selectively. This directed approach drastically reduces the formation of isomeric impurities, leading to significantly higher crude purity and simplified downstream purification.
Q: What is the advantage of using Horseradish Peroxidase in the final synthesis step?
A: The use of Horseradish Peroxidase allows for the simultaneous removal of the methyl protecting group and the oxidative coupling of the third disulfide bond in a single operation. This enzymatic approach operates under mild conditions, avoiding harsh chemical reagents that could degrade the sensitive peptide backbone, thereby enhancing overall yield and environmental compliance.
Q: Is this synthesis method suitable for large-scale commercial manufacturing?
A: Yes, the patent explicitly states the process is suitable for large-scale production. The use of stable solid-phase intermediates, combined with robust liquid-phase oxidation steps using commercially available reagents like NCS and enzymes, ensures process stability and reproducibility required for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Linaclotide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the synthesis of complex polypeptides like Linaclotide requires more than just chemical knowledge; it demands a partnership built on technical excellence and operational reliability. Our team has extensively analyzed the selective oxidation strategies outlined in recent patents and possesses the expertise to implement these advanced routes effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with state-of-the-art peptide synthesis reactors and rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the highest international standards for API intermediates.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our commitment to innovation and quality can drive value for your organization and secure your position in the competitive pharmaceutical market.
