Scalable Manufacturing of Antibacterial Tetraphenylpyrazole Intermediates via Novel Cyclization
Scalable Manufacturing of Antibacterial Tetraphenylpyrazole Intermediates via Novel Cyclization
The pharmaceutical industry is constantly seeking robust, scalable, and cost-effective routes to access novel heterocyclic scaffolds that possess significant biological activity. A recent technological breakthrough documented in patent CN110885313B introduces a highly efficient method for synthesizing antibacterial active tetraphenylpyrazole compounds. These compounds, characterized by the core structure of Formula I, represent a promising class of intermediates for the development of next-generation antimicrobial agents. The innovation lies in a unique cyclization strategy that bypasses traditional harsh conditions, utilizing a base-mediated reaction between a benzisothiazole oxide derivative and a nitrilimine precursor. This approach not only streamlines the synthetic pathway but also ensures high purity profiles essential for downstream drug development. As a leading manufacturer, we recognize the immense value of this chemistry in addressing the growing demand for effective antibiotics.
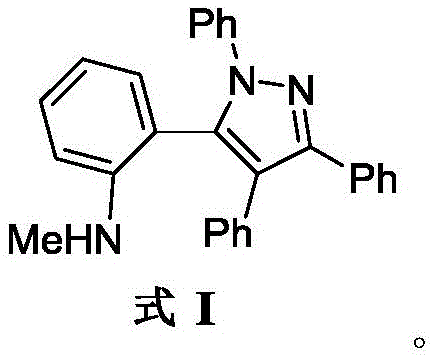
For R&D directors and process chemists, the structural integrity and accessibility of the tetraphenylpyrazole core are paramount. The compound features a densely substituted pyrazole ring fused with multiple phenyl groups and an aniline moiety, creating a sterically crowded yet electronically rich environment conducive to biological interaction. The ability to construct this complex architecture in a single pot from readily available starting materials marks a significant departure from conventional multi-step syntheses. By leveraging this patented methodology, manufacturers can achieve substantial reductions in process mass intensity (PMI) and waste generation, aligning perfectly with modern green chemistry principles while maintaining the rigorous quality standards required for reliable pharmaceutical intermediate supplier partnerships.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for constructing tetrasubstituted pyrazoles often rely on the condensation of 1,3-dicarbonyl compounds with hydrazines or the cycloaddition of alkynes with diazo compounds. These classical routes frequently suffer from significant drawbacks, including the requirement for elevated temperatures, prolonged reaction times, and the use of stoichiometric amounts of hazardous reagents. Furthermore, controlling regioselectivity in the formation of fully substituted pyrazoles can be challenging, often leading to complex mixtures of isomers that are difficult to separate. The reliance on transition metal catalysts in some modern variations introduces another layer of complexity, necessitating expensive metal scavenging steps to meet stringent residual metal limits imposed by regulatory bodies for high-purity pharmaceutical intermediates. These inefficiencies translate directly into higher production costs and extended lead times, creating bottlenecks in the supply chain for critical drug substances.
The Novel Approach
In stark contrast, the methodology disclosed in CN110885313B offers a transformative solution by employing a tandem reaction sequence involving benzisothiazole 1,1-dioxide and hydrazonoyl chlorides. As illustrated in the reaction scheme below, the process proceeds smoothly in a protic solvent like methanol under the influence of a mild inorganic base. This novel approach eliminates the need for external heating, operating efficiently at room temperature, which drastically reduces energy consumption. The reaction exhibits exceptional atom economy, with sulfur dioxide serving as the only byproduct, which is easily removed from the reaction mixture. This clean reaction profile minimizes the formation of side products, thereby simplifying the downstream purification process and enhancing the overall yield. For procurement teams, this translates to cost reduction in API manufacturing through reduced solvent usage, lower energy bills, and higher throughput.
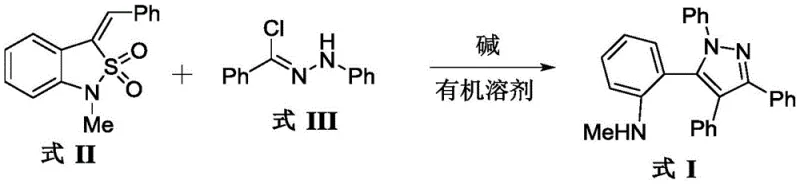
Mechanistic Insights into Base-Mediated Cyclization and SO2 Extrusion
Understanding the mechanistic underpinnings of this transformation is crucial for optimizing the process on a commercial scale. The reaction initiates with the deprotonation of the hydrazonoyl chloride (Formula III) by the base to generate a reactive nitrilimine intermediate in situ. This 1,3-dipole then undergoes a [3+2] cycloaddition with the exocyclic double bond of the benzisothiazole oxide (Formula II). The resulting cyclic adduct is unstable and spontaneously undergoes a cheletropic extrusion of sulfur dioxide (SO2). This extrusion step is the thermodynamic driving force of the reaction, pushing the equilibrium towards the formation of the pyrazoline intermediate. Subsequently, a facile aromatization occurs, likely facilitated by the basic conditions, to yield the final tetraphenylpyrazole product. This elegant cascade avoids the accumulation of high-energy intermediates, ensuring a safe and controllable process suitable for large-scale operations.
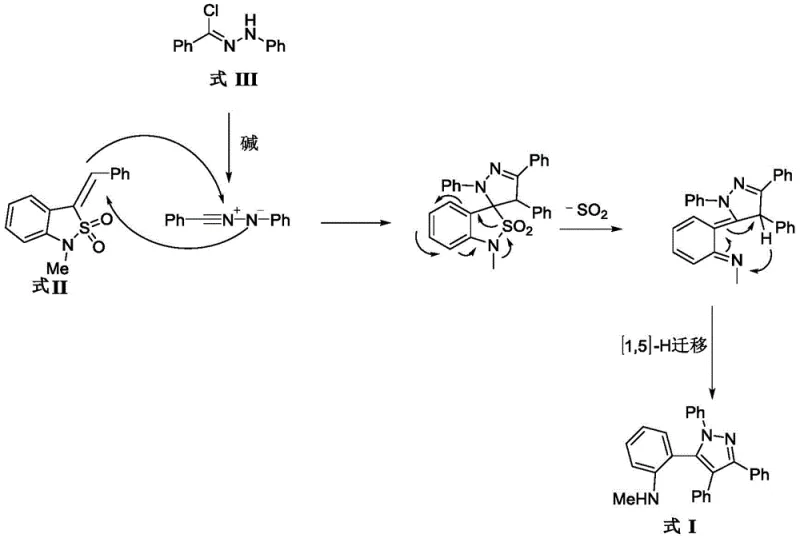
From an impurity control perspective, the mechanism offers distinct advantages. The specificity of the [3+2] cycloaddition minimizes the formation of oligomeric byproducts that are common in radical-based pyrazole syntheses. Furthermore, the rapid extrusion of SO2 prevents the reverse reaction, locking the product into the desired structural framework. The subsequent [1,5]-H migration, as depicted in the mechanistic proposal, ensures the correct positioning of the aniline substituent on the phenyl ring, a critical feature for the compound's antibacterial activity. By strictly controlling the molar ratio of reactants (preferably 1:1.0-1.2) and the amount of base (2.0-2.5 equivalents), manufacturers can suppress minor side reactions, ensuring a consistent and high-quality output. This level of mechanistic clarity allows for precise process parameter optimization, guaranteeing batch-to-batch reproducibility essential for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Tetraphenylpyrazole Efficiently
The practical implementation of this synthesis is remarkably straightforward, making it accessible for both laboratory-scale discovery and industrial production. The protocol involves dissolving the benzisothiazole oxide and the hydrazonoyl chloride in methanol, followed by the addition of a base such as sodium hydroxide or cesium carbonate. The reaction mixture is stirred at ambient temperature for a short duration, typically ranging from 0.5 to 1.0 hours, before being quenched and worked up. The simplicity of the workup procedure, involving standard extraction and chromatography, further underscores the operational ease of this method. For detailed operational parameters and safety considerations, please refer to the standardized guide below.
- Dissolve the benzisothiazole oxide precursor (Formula II) and the hydrazonoyl chloride (Formula III) in a protic solvent such as methanol at a molar ratio of approximately 1: 1.2.
- Add an inorganic base, preferably sodium hydroxide or cesium carbonate (2.0-2.5 equivalents), to the reaction mixture at room temperature to initiate the cyclization.
- Stir the reaction for 0.5 to 1.0 hours, followed by standard aqueous workup involving ethyl acetate extraction, acid/base washing, and silica gel chromatography to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this novel synthetic route offers compelling economic and logistical benefits. The shift from multi-step, high-energy processes to a single-pot, room-temperature reaction fundamentally alters the cost structure of producing these valuable intermediates. By eliminating the need for specialized high-pressure equipment or cryogenic cooling systems, capital expenditure (CAPEX) requirements are significantly lowered. Moreover, the use of commodity chemicals like methanol and sodium hydroxide ensures a stable and resilient supply of raw materials, mitigating the risks associated with sourcing exotic or regulated reagents. This robustness is critical for maintaining continuity of supply in a volatile global market.
- Cost Reduction in Manufacturing: The primary driver for cost efficiency in this process is the dramatic simplification of the reaction workflow. By avoiding the use of expensive transition metal catalysts, the process eliminates the costly and time-consuming step of metal scavenging, which is often a bottleneck in API production. Additionally, the high yields reported (up to 97%) mean that less raw material is wasted, directly improving the material cost per kilogram of the final product. The reduced reaction time also increases reactor turnover rates, allowing for higher production volumes without additional infrastructure investment.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as benzisothiazole derivatives and hydrazonoyl chlorides ensures that the supply chain is not vulnerable to single-source bottlenecks. These precursors are commercially available from multiple vendors globally, providing procurement teams with the flexibility to negotiate better pricing and secure backup supplies. The mild reaction conditions also reduce the risk of process deviations or safety incidents that could lead to unplanned shutdowns, thereby ensuring a steady and predictable delivery schedule for downstream customers.
- Scalability and Environmental Compliance: From an environmental standpoint, the generation of sulfur dioxide as the sole byproduct is manageable and can be effectively scrubbed using standard industrial gas treatment systems. The absence of heavy metals and halogenated solvents (if methanol is used) simplifies waste disposal and reduces the environmental footprint of the manufacturing site. This alignment with green chemistry principles not only aids in regulatory compliance but also enhances the corporate sustainability profile, a factor increasingly important for major pharmaceutical buyers evaluating their supplier base.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these tetraphenylpyrazole compounds. The answers are derived directly from the experimental data and mechanistic insights provided in the patent literature, offering a transparent view of the technology's capabilities. Understanding these details helps stakeholders make informed decisions about integrating this chemistry into their development pipelines.
Q: What are the key advantages of this synthesis route over traditional pyrazole formation?
A: This method utilizes a unique [1,5]-H migration mechanism driven by SO2 extrusion, allowing the reaction to proceed rapidly at room temperature without expensive transition metal catalysts, significantly simplifying the purification process and reducing heavy metal impurities.
Q: Can this process be scaled for commercial production of API intermediates?
A: Yes, the process demonstrates excellent scalability potential due to the use of common solvents like methanol, mild reaction conditions (room temperature), and high yields (up to 97%), making it highly suitable for kilogram-to-ton scale manufacturing.
Q: What is the antibacterial spectrum of the resulting tetraphenylpyrazole compounds?
A: The synthesized compounds exhibit potent activity against Gram-positive bacteria, including Bacillus subtilis and Staphylococcus aureus, with MIC values as low as 0.25 µg/mL, indicating strong potential for developing new antibacterial therapeutics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetraphenylpyrazole Supplier
At NINGBO INNO PHARMCHEM, we combine deep technical expertise with state-of-the-art manufacturing capabilities to deliver high-quality chemical intermediates. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the pre-clinical or commercial phase. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of tetraphenylpyrazole intermediate meets the highest industry standards. Our commitment to quality and consistency makes us a trusted partner for global pharmaceutical companies seeking to accelerate their drug development programs.
We invite you to explore the potential of this innovative synthesis route for your specific applications. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your project needs, demonstrating how switching to this efficient method can optimize your budget. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive quotations. Let us collaborate to bring your next-generation antibacterial therapies to market faster and more economically.
