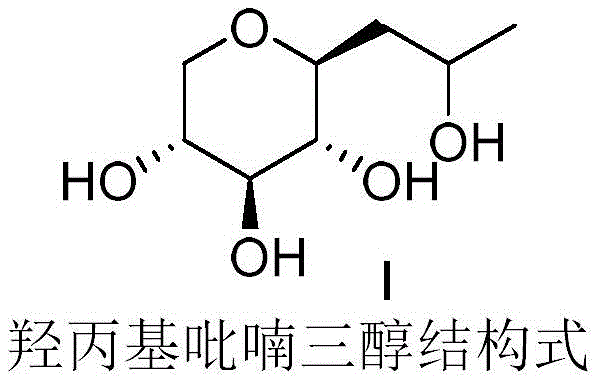
Scaling High-Purity Hydroxypropyl Pyranotriol Production via Continuous Flow Technology
Scaling High-Purity Hydroxypropyl Pyranotriol Production via Continuous Flow Technology
The landscape of cosmetic active ingredient manufacturing is undergoing a significant transformation driven by the need for higher purity and sustainable processing. Patent CN114605366A introduces a groundbreaking continuous flow synthesis method for Hydroxypropyl Pyranotriol, also known commercially as Pro-Xylane. This technology addresses critical bottlenecks in traditional batch production, specifically targeting the issues of impurity formation and process safety. By leveraging microreactor technology, this innovation enables the seamless conversion of D-xylose into the target bioactive molecule with exceptional efficiency. The structural integrity and stereochemistry of the final product are meticulously preserved, ensuring optimal biological activity for anti-aging applications. This report analyzes the technical merits and commercial implications of adopting this continuous flow methodology for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional batch synthesis routes for Hydroxypropyl Pyranotriol have long been plagued by inefficiencies that hinder large-scale commercial viability. Historical methods often rely on prolonged reaction times, sometimes extending up to twelve hours or more, which inevitably leads to thermal degradation of the sensitive carbohydrate starting materials. This extended exposure to heat and alkaline conditions promotes the Maillard reaction and other degradation pathways, resulting in the formation of dark, caramel-like impurities that are notoriously difficult to remove. Furthermore, many prior art processes depend on expensive and potentially toxic heavy metal catalysts, such as Ruthenium on Carbon or rare earth complexes, which introduce stringent regulatory hurdles regarding residual metal limits in cosmetic formulations. The necessity for complex post-treatment procedures, including column chromatography and multiple extraction steps, drastically increases both the operational expenditure and the environmental footprint of the manufacturing process.
The Novel Approach
In stark contrast, the continuous flow methodology outlined in the patent represents a paradigm shift towards intensification and precision. By utilizing a series of interconnected microreactors and mixers, the reaction time is compressed from hours to mere seconds or minutes, effectively suppressing the kinetic pathways that lead to unwanted byproducts. The process integrates the substitution and reduction steps into a single, uninterrupted stream, eliminating the need for isolating unstable intermediates. This telescoped approach not only enhances the overall space-time yield but also ensures a consistent product quality profile with minimal isomer formation. The use of inexpensive and readily available reagents, such as sodium carbonate and sodium borohydride, further simplifies the supply chain logistics. The result is a transparent, high-purity oily product that requires minimal downstream purification, making it ideally suited for direct application in high-end cosmetic formulations.
Mechanistic Insights into Alkaline Condensation and In-Situ Reduction
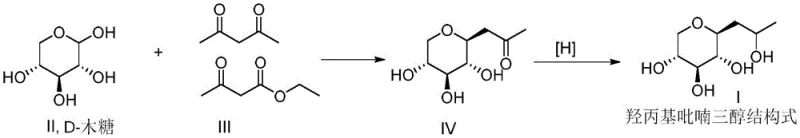
The core of this innovative synthesis lies in the precise orchestration of two distinct chemical transformations within a continuous flow environment. The first stage involves the alkaline-catalyzed condensation of D-xylose with an acylation reagent, such as acetylacetone or ethyl acetoacetate. In the microchannel reactor, the reactants are rapidly mixed and heated to temperatures between 120°C and 150°C for a strictly controlled residence time of 30 to 300 seconds. This high-temperature, short-time regime facilitates the rapid formation of the C-glycoside intermediate while minimizing the thermal decomposition that typically occurs in batch vessels. The efficient heat transfer characteristics of the microreactor ensure that the reaction mixture does not experience localized hot spots, which are a primary cause of caramelization in conventional systems. This precise thermal management is critical for maintaining the stereochemical integrity of the sugar moiety.
Following the condensation step, the reaction stream immediately enters a second mixing zone where a reducing agent, typically sodium borohydride or potassium borohydride, is introduced. This in-situ reduction converts the carbonyl group of the intermediate into the corresponding hydroxyl group, finalizing the structure of Hydroxypropyl Pyranotriol. The reduction is conducted at milder temperatures, generally between 20°C and 80°C, to prevent over-reduction or side reactions. The continuous nature of the flow ensures that the reducing agent is consumed efficiently, and the subsequent quenching with acid is performed instantly to stop the reaction. This seamless transition from substitution to reduction without intermediate workup prevents the exposure of the reactive intermediate to air or moisture, thereby maximizing the overall yield. The final quenching and separation steps are also integrated into the flow system, allowing for continuous liquid-liquid extraction and solvent removal.
How to Synthesize Hydroxypropyl Pyranotriol Efficiently
The implementation of this continuous flow protocol requires a specialized setup comprising feeding systems, micromixers, and tubular reactors designed to handle corrosive and reactive streams. The process begins with the preparation of three distinct feed solutions: a D-xylose solution, an acylation reagent solution, and an alkaline solution. These streams are pumped into a first mixer and then through a heated microchannel reactor to effect the condensation. Subsequently, a reducing agent suspension is merged with the effluent in a second mixer before passing through a baffled reactor for the reduction phase.
- Mix D-xylose, acylation reagent (acetylacetone or ethyl acetoacetate), and alkaline solution in a microreactor for substitution reaction.
- Directly introduce a reducing agent (e.g., sodium borohydride) into the stream for immediate reduction without intermediate isolation.
- Quench the reaction mixture, perform continuous oil-water separation, and desolventize to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this continuous flow technology offers compelling economic and operational benefits that extend beyond simple yield improvements. The elimination of column chromatography, a standard but costly purification step in batch synthesis, results in a drastic reduction in consumable costs and waste generation. By removing the need for large volumes of silica gel and elution solvents, the process significantly lowers the variable cost per kilogram of the active ingredient. Furthermore, the continuous nature of the reaction allows for a smaller physical footprint compared to equivalent batch plants, reducing capital expenditure requirements for new production lines. The ability to operate the plant continuously rather than in discrete batches enhances asset utilization rates and provides a more predictable output schedule for meeting market demand.
- Cost Reduction in Manufacturing: The streamlined process eliminates expensive heavy metal catalysts and complex purification steps, leading to substantial savings in raw material and waste disposal costs. The use of commodity chemicals like sodium carbonate and borohydride instead of specialized organometallic catalysts reduces dependency on volatile precious metal markets. Additionally, the high atom economy and reduced solvent consumption inherent in flow chemistry contribute to a leaner manufacturing cost structure. These factors collectively enable a more competitive pricing strategy for the final cosmetic active without compromising on quality standards.
- Enhanced Supply Chain Reliability: Continuous manufacturing systems are inherently more robust against supply disruptions because they maintain a steady state of production rather than relying on large, infrequent batches. The simplified workflow reduces the number of unit operations and potential points of failure, ensuring a consistent and reliable supply of high-purity material. The shorter lead times associated with continuous processing allow for more agile responses to fluctuations in customer demand. This reliability is crucial for long-term contracts with major cosmetic brands that require guaranteed availability of key active ingredients.
- Scalability and Environmental Compliance: The modular nature of flow reactors allows for straightforward scale-up by increasing the number of reactor units or extending the run time, avoiding the engineering challenges of scaling up batch vessel volumes. The process generates significantly less hazardous waste due to the absence of heavy metals and reduced solvent usage, aligning with increasingly stringent global environmental regulations. The improved safety profile, characterized by small reactor hold-up volumes and better thermal control, minimizes the risk of runaway reactions. This compliance with green chemistry principles enhances the sustainability credentials of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the continuous flow synthesis of Hydroxypropyl Pyranotriol. These insights are derived directly from the experimental data and process descriptions found in the patent literature. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing manufacturing portfolios.
Q: How does continuous flow improve the purity of Hydroxypropyl Pyranotriol compared to batch methods?
A: Continuous flow allows for precise control of reaction temperature and residence time, significantly reducing the formation of caramel-like impurities and isomers common in long-duration batch processes.
Q: Does this synthesis method involve heavy metal catalysts?
A: No, the patented continuous flow method utilizes alkaline conditions and borohydride reducing agents, eliminating the risk of toxic heavy metal residues associated with Ru/C or rare earth catalysts.
Q: What are the scalability advantages of this continuous process?
A: The process operates with short residence times (seconds to minutes) and eliminates complex purification steps like column chromatography, making it highly suitable for large-scale industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydroxypropyl Pyranotriol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of continuous flow chemistry in the production of high-value cosmetic actives like Hydroxypropyl Pyranotriol. Our team of expert process chemists is dedicated to translating laboratory-scale innovations into robust, commercial-scale manufacturing processes. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of material that meets stringent purity specifications. Our state-of-the-art facilities are equipped with rigorous QC labs capable of performing detailed impurity profiling to guarantee the safety and efficacy of every batch produced.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By collaborating with us, you can leverage our expertise to achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to request specific COA data and route feasibility assessments to verify the superior quality and economic advantages of our continuous flow-derived Hydroxypropyl Pyranotriol. Let us help you secure a sustainable and cost-effective source for this critical anti-aging ingredient.
