Optimizing DL-Naproxen Production: A Technical Analysis of the Novel Ketal-First Industrial Route
Optimizing DL-Naproxen Production: A Technical Analysis of the Novel Ketal-First Industrial Route
The global demand for non-steroidal anti-inflammatory drugs (NSAIDs) continues to drive the need for efficient, safe, and cost-effective manufacturing processes for key intermediates like DL-Naproxen. Patent CN101234963A introduces a significant technological breakthrough in the industrial synthesis of DL-Naproxen base, addressing critical limitations found in legacy production methods. This technical insight report analyzes the proprietary ketal-first synthetic route, which fundamentally reorders the reaction sequence to prioritize stability and yield. By shifting the ketalization step to the beginning of the synthesis, prior to bromination, the process mitigates the decomposition issues plaguing traditional propionyl metallization technologies. For R&D directors and procurement leaders, understanding this mechanistic shift is vital for evaluating supply chain resilience and long-term cost structures in the competitive NSAID market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of DL-Naproxen has relied heavily on two primary pathways: the acetonaphthone technology and the standard propionyl metallization process. The acetonaphthone route, while established, presents severe safety and environmental challenges due to its reliance on methyl chloroacetate and boron trifluoride, both of which are highly toxic and corrosive. Furthermore, the recovery of boron trifluoride and diethyl ether solutions requires stringent security measures and complex equipment, creating bottlenecks in continuous production. On the other hand, the conventional propionyl metallization process suffers from intrinsic chemical instability; during the halogenation phase, the bromo-derivative intermediates are prone to rapid decomposition, often causing the reaction feed liquid to turn black. This degradation necessitates repeated extraction and decolorization steps with toluene, drastically reducing overall yield and complicating the purification workflow.
The Novel Approach
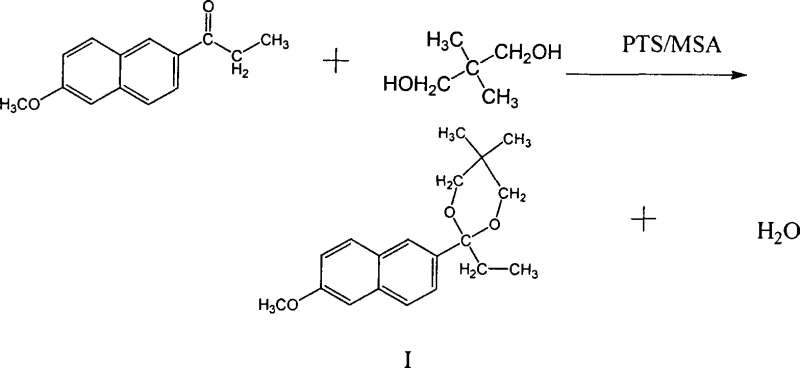
The innovative synthesis technique described in the patent data fundamentally alters the reaction order to overcome these stability hurdles. Instead of attempting to brominate an unstable ketone directly, the process first converts 2-propionyl-6-methoxynaphthalene into a stable ketal intermediate (Compound I) using neopentyl glycol. This protective group strategy shields the carbonyl functionality, allowing subsequent alpha-bromination to proceed with high selectivity and minimal side reactions. The ketalization step itself is remarkably efficient, achieving near-quantitative yields of approximately 100% under optimized conditions with catalysts like p-toluenesulfonic acid (PTS) or methanesulfonic acid (MSA). By establishing a robust molecular scaffold early in the synthesis, the subsequent rearrangement and hydrolysis steps become far more manageable, eliminating the need for the hazardous boron trifluoride catalysts used in older acetylation-based routes.
Mechanistic Insights into Ketal-Stabilized Alpha-Bromination and Rearrangement
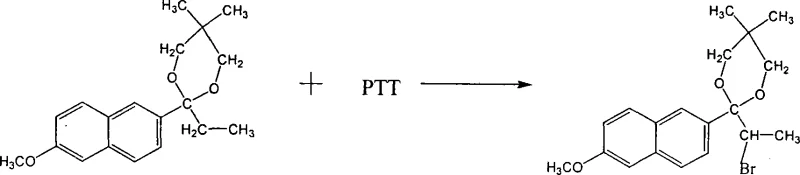
The core chemical innovation lies in the precise control of the alpha-bromination and subsequent rearrangement mechanisms. Once the stable ketal intermediate is formed, it undergoes alpha-bromination using phenyltrimethylammonium tribromide (PTT) in solvents such as dichloroethane or toluene. This specific brominating agent offers superior control compared to elemental bromine, minimizing poly-bromination and ring-substitution byproducts. Following bromination, the molecule undergoes a zinc-catalyzed rearrangement where the ketal ring opens, and the carbon skeleton reorganizes to form the naproxen precursor. This rearrangement is typically conducted at elevated temperatures between 50°C and 120°C using zinc oxide or zinc salts, which act as mild Lewis acids to facilitate the migration without degrading the sensitive naphthalene core.
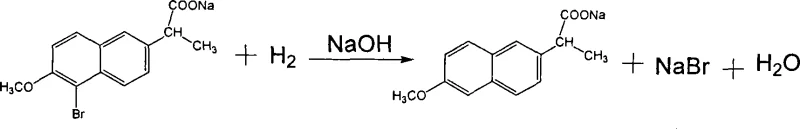
A critical aspect of this mechanism is the management of the 5-bromo byproduct (Compound III), which inevitably forms during the halogenation stage. Unlike traditional methods where this impurity might persist or require difficult chromatographic removal, this process leverages a catalytic hydrogenation step to convert the unwanted bromo-impurity directly into the desired product structure. Using active nickel or palladium on carbon under mild pressure (0.1–0.4 MPa), the carbon-bromine bond is cleaved and replaced with hydrogen. This "repair" mechanism not only enhances the final chemical purity to over 99.7% but also maximizes atom economy by salvaging material that would otherwise be lost as waste, demonstrating a sophisticated approach to impurity profiling and control.
How to Synthesize DL-Naproxen Efficiently
The synthesis of DL-Naproxen via this optimized route involves a telescoped sequence that minimizes intermediate isolation. The process begins with the condensation of the propionyl naphthol methyl ether with a glycol, followed by in-situ or sequential bromination and rearrangement. The operational simplicity allows for the reaction mass to proceed through hydrolysis and debromination without the need for drying unstable intermediates, which is a common failure point in legacy processes. For detailed standard operating procedures regarding stoichiometry, temperature ramping, and workup protocols, please refer to the standardized synthesis guide below.
- Perform ketalization of 2-propionyl-6-methoxynaphthalene with neopentyl glycol using PTS or MSA catalyst to form Ketal I.
- Conduct alpha-bromination of Ketal I using PTT (phenyltrimethylammonium tribromide) to obtain Bromo Ketal II.
- Execute rearrangement with zinc oxide, followed by alkaline hydrolysis, catalytic hydrogenation for debromination, and final acidification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this ketal-first synthesis route offers transformative advantages for supply chain stability and cost management. By eliminating the dependence on highly regulated and hazardous reagents like boron trifluoride, manufacturers can significantly reduce compliance costs and insurance premiums associated with handling dangerous chemicals. The process utilizes common, recyclable solvents such as toluene and dichloroethane, which are readily available in the global chemical market, thereby insulating the supply chain from the volatility of specialty reagent pricing. Furthermore, the high yield of the initial ketalization step ensures that expensive starting materials are not wasted in low-conversion reactions, directly improving the cost-of-goods-sold (COGS) profile for the final API intermediate.
- Cost Reduction in Manufacturing: The elimination of toxic raw materials like methyl chloroacetate removes the need for expensive containment systems and specialized waste disposal services. Additionally, the ability to recycle unreacted starting materials in the ketalization step drives raw material utilization efficiency to near-theoretical maximums. The simplified workflow reduces energy consumption by removing complex drying and solvent exchange steps required in the acetonaphthone technology, leading to substantial operational expenditure savings.
- Enhanced Supply Chain Reliability: The robustness of the ketal intermediate against decomposition means that production campaigns are less likely to be interrupted by batch failures or off-spec material. The use of stable catalysts like zinc oxide and PTT ensures consistent reaction kinetics, allowing for predictable cycle times. This reliability is crucial for maintaining continuous supply to downstream API manufacturers, reducing the risk of stockouts caused by unpredictable synthesis bottlenecks.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial tonnage production in mind. The reduction in hazardous waste generation, particularly the avoidance of boron-containing effluents, simplifies environmental permitting and wastewater treatment. The telescoped nature of the reaction sequence reduces the physical footprint required for production, allowing existing facilities to increase capacity without major capital infrastructure investments.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the comparative data and experimental embodiments provided in the patent literature, focusing on the practical implications for industrial adoption.
Q: What are the safety advantages of this ketal-first route compared to traditional acetonaphthone technology?
A: Traditional acetonaphthone technology relies on highly toxic raw materials like methyl chloroacetate and hazardous catalysts like boron trifluoride. The novel ketal-first route utilizes lower toxicity reagents such as neopentyl glycol and tosic acid, significantly reducing occupational health risks and simplifying waste treatment protocols.
Q: How does this process control impurities specifically related to bromination byproducts?
A: The process incorporates a dedicated catalytic hydrogenation step using active nickel or palladium carbon after hydrolysis. This effectively converts unstable 5-bromo byproducts into the desired product structure, ensuring high chemical purity without the need for complex chromatographic separation.
Q: Is this synthesis method suitable for large-scale commercial manufacturing?
A: Yes, the method is designed for industrial scalability. It features high-yield ketalization steps approaching quantitative conversion, allows for solvent recycling (toluene, dichloroethane), and eliminates difficult drying steps for unstable intermediates, making it robust for tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable DL-Naproxen Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthesis routes requires a partner with deep technical expertise and proven scale-up capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this ketal-first route are fully realized in a GMP-compliant environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of DL-Naproxen intermediate meets the exacting standards required for global pharmaceutical registration, providing our partners with confidence in both quality and consistency.
We invite procurement leaders and R&D teams to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized manufacturing platforms, we can help you navigate the complexities of NSAID intermediate sourcing. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced synthesis capabilities can drive value for your supply chain.
