Advanced Synthetic Strategy for Doramide Intermediates: Enhancing Purity and Scalability for Global Pharma Supply Chains
Introduction to the Novel Doramide Synthesis Platform
The global demand for high-quality androgen receptor antagonists, specifically Doramide (Darolutamide), has necessitated the development of more efficient and sustainable manufacturing processes. Patent CN111087324A introduces a groundbreaking synthetic methodology that fundamentally restructures the production of this critical prostate cancer therapeutic. Unlike traditional approaches that rely on complex cross-coupling reactions, this invention leverages a concise sequence starting from 4-acetyl-2-chlorobenzonitrile. The core innovation lies in the formation of a vinylogous amide intermediate via reaction with DMF-DMA, which serves as a versatile precursor for pyrazole ring construction. This strategic pivot not only streamlines the synthetic pathway but also addresses critical pain points regarding impurity control and process safety. By integrating this advanced chemistry, manufacturers can achieve a significant reduction in process mass intensity while maintaining stringent quality specifications required for oncology drugs.
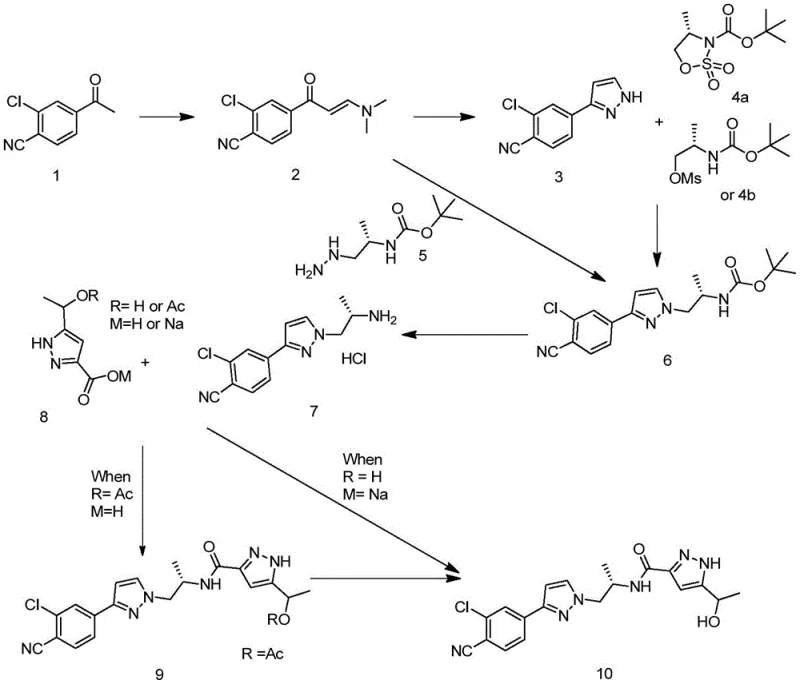
The comprehensive reaction scheme illustrates the convergence of two major structural fragments through a series of highly selective transformations. The left-hand side of the synthesis focuses on the construction of the chlorophenyl-pyrazole moiety, while the right-hand side details the assembly of the hydroxyethyl-pyrazole carboxamide segment. The elegance of this route is evident in its modularity; the key intermediate, Compound 6, can be accessed via two distinct pathways depending on the availability of chiral starting materials. This flexibility provides supply chain managers with multiple options for sourcing and production planning, ensuring continuity even if specific reagents face market volatility. Furthermore, the final deprotection and coupling steps are optimized for high yield and minimal byproduct formation, setting a new benchmark for the commercial production of high-purity Doramide intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those disclosed in PCT patent WO2011051540A, typically depend on palladium-catalyzed Suzuki couplings to link the aryl halide and pyrazole boronic acid fragments. While effective on a small scale, these transition metal-mediated reactions introduce significant challenges for large-scale manufacturing, including the high cost of noble metal catalysts and the rigorous requirement for residual metal removal to meet ICH Q3D guidelines. Additionally, alternative routes reported in WO2016120530A utilize indium trichloride for cyclization or enzymatic reductions, which can suffer from scalability issues and high reagent costs. A particularly concerning aspect of older syntheses is the reliance on ethyl diazoacetate, a hazardous and potentially explosive reagent that poses severe safety risks during kilogram-to-ton scale operations. The accumulation of phosphine oxides from Mitsunobu reactions further complicates downstream processing, often requiring extensive chromatography or recrystallization steps that erode overall yield and increase waste generation.
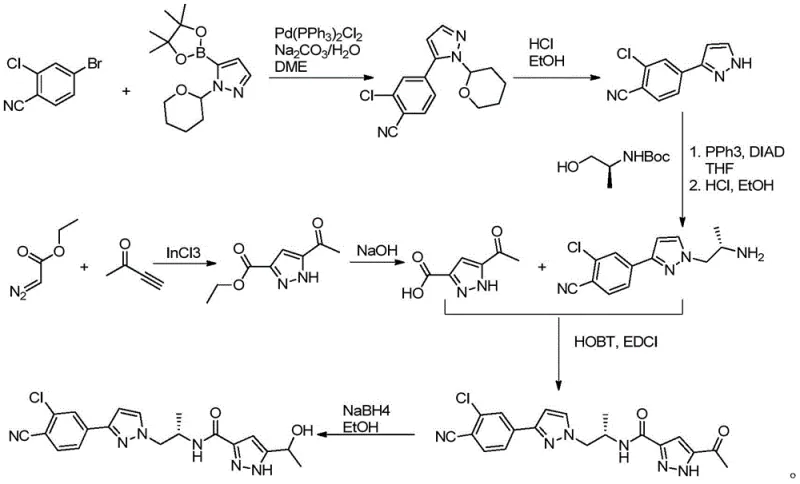
The Novel Approach
In stark contrast, the novel approach detailed in CN111087324A circumvents these obstacles by employing a metal-free condensation strategy. The initial transformation of the acetyl-nitrile starting material into an enamine using DMF-DMA activates the methyl ketone for subsequent nucleophilic attack by hydrazine. This eliminates the need for expensive palladium or indium catalysts entirely, thereby drastically lowering the raw material cost profile. The cyclization step proceeds under mild thermal conditions using common solvents like ethanol or ethyl acetate, which are far more environmentally benign than the chlorinated solvents often required for cross-coupling workups. Moreover, the avoidance of diazo compounds removes a major safety hazard from the plant floor, facilitating easier regulatory approval for commercial manufacturing sites. The result is a robust, linear synthesis that delivers the target intermediate with superior purity and a significantly simplified isolation protocol, making it ideally suited for cost reduction in pharmaceutical manufacturing.
Mechanistic Insights into Enamine-Mediated Pyrazole Formation
The mechanistic cornerstone of this improved synthesis is the formation and reactivity of the enamine intermediate, Compound 2. When 4-acetyl-2-chlorobenzonitrile reacts with DMF-DMA, the acidic alpha-protons of the ketone are abstracted, leading to the elimination of dimethylamine and the formation of a conjugated system. This vinylogous amide structure possesses enhanced electrophilicity at the beta-carbon, making it highly susceptible to nucleophilic attack by the terminal nitrogen of hydrazine hydrate. This electronic activation allows the cyclization to proceed rapidly and regioselectively to form the 1H-pyrazole ring without the need for harsh Lewis acids. The electron-withdrawing nitrile group on the phenyl ring further stabilizes the transition state, ensuring high conversion rates even at moderate temperatures. Understanding this electronic interplay is crucial for R&D directors aiming to optimize reaction parameters, as slight adjustments in pH or solvent polarity can fine-tune the ratio of regioisomers, although this specific route is designed to favor the desired 3-substituted pyrazole exclusively.
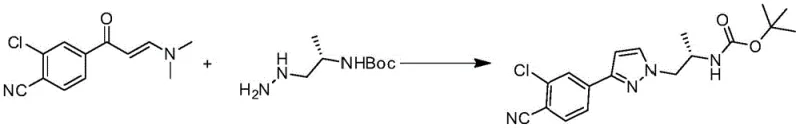
Furthermore, the patent describes an innovative one-pot variation where the enamine reacts directly with a chiral hydrazine derivative (Compound 5). This telescoped process combines the ring closure and the installation of the chiral alkyl side chain into a single operational unit. Mechanistically, this involves the initial formation of the pyrazole nucleus followed immediately by an intramolecular or intermolecular alkylation event, depending on the specific reagent design. This consolidation of steps not only saves time and solvent but also minimizes the exposure of reactive intermediates to potential degradation pathways. For process chemists, this represents a significant advancement in impurity control, as fewer isolation steps mean fewer opportunities for the introduction of particulate matter or solvent-borne contaminants. The ability to install chirality early in the synthesis ensures that downstream purification efforts are focused on removing diastereomers rather than racemic mixtures, thereby enhancing the overall optical purity of the final Doramide product.
How to Synthesize Doramide Efficiently
The execution of this synthesis requires precise control over reaction stoichiometry and temperature profiles to maximize the efficiency of the enamine formation and subsequent cyclization. The process begins with the heating of the acetyl-nitrile precursor with DMF-DMA, followed by the careful addition of hydrazine sources to trigger ring closure. Detailed standard operating procedures for each transformation, including specific workup protocols to remove dimethylamine byproducts and unreacted hydrazine, are critical for reproducibility. The following guide outlines the standardized synthesis steps derived from the patent examples, providing a clear roadmap for laboratory and pilot plant implementation.
- Condense 4-acetyl-2-chlorobenzonitrile with DMF-DMA to form the key enamine intermediate (Compound 2).
- Cyclize the enamine with hydrazine hydrate or a chiral hydrazine derivative to construct the pyrazole core (Compound 3 or 6).
- Deprotect the Boc group to form the amine salt (Compound 7), followed by amide coupling with the pyrazole acid fragment to yield Doramide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible strategic benefits that extend beyond simple chemistry. The primary advantage lies in the substantial cost savings achieved by eliminating noble metal catalysts and hazardous reagents from the bill of materials. By replacing palladium and indium with commodity chemicals like DMF-DMA and hydrazine, the direct material cost is significantly reduced, allowing for more competitive pricing in tender negotiations. Additionally, the simplified purification workflow reduces the consumption of silica gel and chromatography solvents, which are often hidden cost drivers in API manufacturing. This leaner process translates to a lower cost of goods sold (COGS), providing a buffer against raw material price fluctuations and enhancing the overall profitability of the supply chain.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts such as palladium and indium removes a major cost center from the production budget. Furthermore, the avoidance of specialized reagents like ethyl diazoacetate reduces the need for costly safety infrastructure and hazardous waste disposal services. The use of common organic solvents like ethanol and ethyl acetate, which are easily recovered and recycled, further drives down operational expenditures. These cumulative efficiencies result in a markedly lower production cost per kilogram, enabling significant margin improvement for generic manufacturers.
- Enhanced Supply Chain Reliability: The starting materials for this route, including 4-acetyl-2-chlorobenzonitrile and DMF-DMA, are widely available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions means that the process is less sensitive to minor variations in reagent quality, ensuring consistent output even when sourcing from different vendors. This resilience is critical for maintaining continuous supply to downstream formulation partners, minimizing the risk of stockouts that could impact patient access to medication. The simplified logistics of handling non-hazardous solids and liquids also streamline warehouse management and transportation compliance.
- Scalability and Environmental Compliance: The synthetic pathway is inherently designed for scale-up, utilizing reaction types that are well-understood in large-scale chemical engineering, such as reflux condensation and slurry filtration. The absence of heavy metals simplifies the environmental permitting process, as wastewater streams do not require complex metal scavenging treatments before discharge. This alignment with green chemistry principles not only reduces the environmental footprint but also future-proofs the manufacturing site against increasingly stringent regulatory standards. The high purity of the crude product minimizes the need for energy-intensive recrystallization cycles, contributing to a more sustainable and energy-efficient production lifecycle.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Doramide synthesis route. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing clarity on feasibility and performance. Understanding these nuances is essential for technical teams evaluating the transfer of this technology to commercial production facilities.
Q: Why is the new synthesis route for Doramide considered superior to prior art methods?
A: The novel route described in CN111087324A eliminates the need for expensive transition metal catalysts like Palladium and Indium, which are required in previous Suzuki-coupling or diazo-cyclization methods. This significantly reduces raw material costs and simplifies the removal of heavy metal impurities, ensuring higher purity standards for the final API.
Q: How does this process improve supply chain security for Doramide intermediates?
A: By utilizing readily available starting materials such as 4-acetyl-2-chlorobenzonitrile and avoiding hazardous reagents like ethyl diazoacetate, the process enhances operational safety and原料 availability. The robust nature of the condensation and cyclization steps allows for easier commercial scale-up, reducing the risk of supply disruptions.
Q: What are the purity advantages of avoiding Mitsunobu reactions in this synthesis?
A: Traditional routes often rely on Mitsunobu reactions which generate stoichiometric amounts of phosphine oxide byproducts that are difficult to remove. The new alkylation strategy using sulfonate esters or direct cyclization avoids these persistent impurities, leading to a cleaner crude profile and reduced burden on downstream purification processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Doramide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes for high-value oncology intermediates like Doramide. Our technical team has extensively analyzed the methodology presented in CN111087324A and possesses the expertise to implement these advanced enamine-based strategies effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our facility is equipped with rigorous QC labs capable of detecting trace impurities at ppm levels, guaranteeing that every batch meets the stringent purity specifications required by global regulatory agencies.
We invite pharmaceutical partners to collaborate with us to leverage this cost-effective and safe manufacturing platform. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to verify how this optimized synthesis can enhance your supply chain resilience and reduce overall project costs. Let us be your trusted partner in delivering high-quality Doramide intermediates to the market.
