Revolutionizing Naproxen Intermediate Production via Scalable Electrooxidative Technology
Revolutionizing Naproxen Intermediate Production via Scalable Electrooxidative Technology
The pharmaceutical industry is currently witnessing a paradigm shift towards sustainable manufacturing processes, driven by the urgent need to reduce environmental footprints and operational costs. A pivotal development in this arena is documented in Chinese patent CN113308704B, which discloses a novel electrooxidative preparation method for 6-methoxy-2-naphthylglyoxylate, a critical intermediate in the synthesis of the non-steroidal anti-inflammatory drug (NSAID) Naproxen. This technology represents a significant departure from traditional stoichiometric oxidation methods, leveraging electricity as a clean reagent to drive chemical transformations. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, understanding the implications of this electrochemical approach is vital. The patent details a robust protocol that operates under mild conditions, utilizing an undivided electrolytic cell to achieve high conversion rates without the need for hazardous chemical oxidants. This innovation not only addresses the growing regulatory pressure for greener chemistry but also offers tangible benefits in terms of process safety and scalability, positioning it as a superior alternative for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 6-methoxy-2-naphthylglyoxylate has relied on methodologies that pose significant challenges for industrial implementation. One prominent route involves the formation of a Grignard reagent from 2-bromo-6-methoxynaphthalene, followed by reaction with ethyl chlorooxalate. As illustrated in the reaction scheme below, this pathway is fraught with operational hazards.
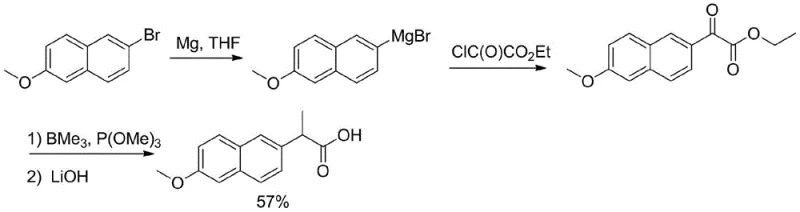
The preparation of the requisite Grignard reagent demands strictly anhydrous and oxygen-free conditions, necessitating expensive inert gas purging and specialized equipment. Furthermore, the subsequent reaction with oxalate derivatives often requires cryogenic temperatures, typically around -78°C, to control selectivity and prevent side reactions. Maintaining such ultra-low temperatures on a multi-ton scale is energy-intensive and logistically complex. Another reported method involves palladium-catalyzed coupling with nitroacetates, which introduces additional complications. This route relies on expensive transition metal catalysts and fluorine-containing reagents for the final transformation, often requiring glovebox techniques for laboratory execution. These factors collectively contribute to high production costs, significant waste generation, and extended lead times, creating bottlenecks for supply chain heads aiming for cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
In stark contrast, the electrooxidative method presented in patent CN113308704B offers a streamlined and environmentally benign solution. The core innovation lies in the direct anodic oxidation of 6-methoxy-2-naphthylacetate esters. Instead of relying on stoichiometric amounts of toxic oxidants or cryogenic cooling, this process utilizes electrons to drive the reaction. The setup is remarkably simple: an undivided electrolytic cell equipped with a carbon felt anode and a platinum mesh cathode. The reaction proceeds in a mixture of organic solvent (such as acetonitrile) and water, mediated by N-hydroxyphthalimide (NHPI) derivatives. By applying a constant current at moderate temperatures ranging from 15°C to 65°C, the substrate is efficiently converted to the desired alpha-keto ester. This approach eliminates the need for hazardous reagents and extreme thermal conditions, thereby simplifying the engineering requirements for plant design. The ability to tune selectivity and conversion simply by adjusting current density provides process chemists with a powerful handle for optimization, ensuring consistent quality of the high-purity pharmaceutical intermediate essential for downstream API synthesis.
Mechanistic Insights into Electrooxidative Alpha-Functionalization
The success of this electrochemical transformation hinges on the efficient mediation of electron transfer at the electrode surface. The mechanism involves the anodic oxidation of the N-hydroxyphthalimide (NHPI) mediator to generate the phthalimide-N-oxyl (PINO) radical. This stable radical species acts as a hydrogen atom transfer (HAT) agent, selectively abstracting a hydrogen atom from the benzylic position of the 6-methoxy-2-naphthylacetate substrate. The resulting carbon-centered radical is then further oxidized at the anode or by another equivalent of the PINO radical to form a carbocation or directly couple with oxygen sources present in the electrolyte, ultimately yielding the alpha-keto ester functionality. This catalytic cycle allows the reaction to proceed at lower potentials than direct substrate oxidation would require, minimizing electrode fouling and side reactions. The use of a carbon felt anode provides a high surface area for this catalytic turnover, enhancing the overall efficiency of the cell. Understanding this mechanistic nuance is crucial for R&D teams, as it highlights the importance of mediator concentration and current density in maintaining the balance between reaction rate and selectivity.
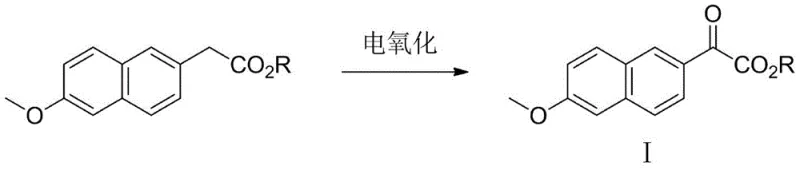
Furthermore, the impurity profile of the electrochemical process is inherently cleaner compared to traditional chemical oxidation. In conventional methods using strong oxidants like chromium or manganese salts, over-oxidation to carboxylic acids or degradation of the naphthalene ring is a common concern, leading to difficult purification steps. In the electrooxidative protocol, the mild potential window controlled by the NHPI mediator prevents such aggressive degradation. The primary byproducts are typically derived from the mediator itself or minor solvent interactions, which are easily removed during standard aqueous workup and crystallization. This inherent selectivity ensures that the resulting 6-methoxy-2-naphthylglyoxylate meets stringent purity specifications required for GMP manufacturing. For quality assurance teams, this means reduced risk of genotoxic impurities and a more robust control strategy, facilitating smoother regulatory filings for the final drug product.
How to Synthesize 6-Methoxy-2-naphthylglyoxylate Efficiently
Implementing this electrochemical protocol requires careful attention to cell configuration and electrolyte composition to maximize yield and throughput. The patent outlines a generalized procedure that can be adapted for various scales, from laboratory benchtop to pilot plant operations. The process begins with the dissolution of the starting material, 6-methoxy-2-naphthylacetate, in a suitable organic solvent such as acetonitrile, often with a small percentage of water to facilitate proton management. Key additives include the NHPI mediator and a supporting electrolyte like tetrabutylammonium perchlorate to ensure sufficient conductivity. The detailed standardized synthesis steps, including specific molar ratios, current densities, and workup procedures, are provided in the guide below.
- Prepare the electrolyte by dissolving 6-methoxy-2-naphthylacetate, N-hydroxyphthalimide (NHPI), and tetrabutylammonium perchlorate in acetonitrile with a small amount of water.
- Install a carbon felt anode and a platinum mesh cathode in an undivided electrolytic cell equipped with magnetic stirring.
- Perform galvanostatic electrolysis at a constant current density between 5mA/cm² and 20mA/cm² at temperatures ranging from 15°C to 65°C until conversion is complete.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electrooxidative technology translates into significant strategic advantages beyond mere technical feasibility. The shift from batch chemical oxidation to continuous or semi-continuous electrochemical processing fundamentally alters the cost structure of manufacturing. By replacing expensive and hazardous chemical oxidants with electricity, the variable cost of goods sold (COGS) is substantially reduced. Moreover, the elimination of cryogenic requirements removes a major energy burden, allowing facilities to operate with standard cooling infrastructure rather than specialized low-temperature chillers. This simplification of utility requirements directly contributes to cost reduction in pharmaceutical intermediate manufacturing, making the supply of Naproxen intermediates more resilient against energy price volatility.
- Cost Reduction in Manufacturing: The electrochemical route eliminates the need for stoichiometric amounts of expensive oxidizing agents and transition metal catalysts, which are often subject to market price fluctuations. Additionally, the removal of cryogenic cooling (-78°C) drastically lowers energy consumption, as the reaction proceeds efficiently at near-ambient temperatures. The simplified workup procedure, which avoids complex quenching of reactive metals or removal of heavy metal residues, further reduces waste disposal costs and solvent usage, leading to substantial overall savings in the production budget.
- Enhanced Supply Chain Reliability: Traditional methods relying on Grignard reagents are sensitive to moisture and oxygen, making them prone to batch failures and inconsistent yields, which can disrupt supply schedules. The electrooxidative method is more robust, operating in the presence of water and air without significant degradation of performance. This reliability ensures consistent delivery timelines for downstream API manufacturers. Furthermore, the starting material, 6-methoxy-2-naphthylacetate, is readily available and stable, reducing the risk of raw material shortages that often plague complex organometallic precursors.
- Scalability and Environmental Compliance: Electrochemical reactors are inherently scalable through the addition of electrode plates or the use of flow cells, allowing for seamless transition from pilot to commercial production without extensive re-optimization. From an environmental perspective, the process generates minimal hazardous waste, as "electrons" serve as the clean reagent. This aligns perfectly with global sustainability goals and strict environmental regulations, reducing the regulatory burden and permitting risks associated with handling toxic oxidants and heavy metals, thereby securing long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrooxidative synthesis of Naproxen intermediates. These answers are derived directly from the experimental data and claims presented in patent CN113308704B, providing clarity on the feasibility and benefits of this advanced manufacturing technique. Understanding these details is essential for stakeholders evaluating the integration of this technology into their existing supply chains.
Q: What are the primary advantages of the electrooxidative method over traditional Grignard routes?
A: The electrooxidative method described in patent CN113308704B operates at mild temperatures (15°C-65°C) compared to the cryogenic conditions (-78°C) required for Grignard reagents. It eliminates the need for strictly anhydrous and oxygen-free environments, significantly reducing operational complexity and energy costs associated with cooling.
Q: Is this electrochemical process suitable for large-scale industrial manufacturing?
A: Yes, the process utilizes an undivided cell with robust electrode materials like carbon felt and platinum mesh. The absence of toxic chemical oxidants and the use of electrons as clean reagents simplify waste treatment and safety protocols, making it highly amenable to commercial scale-up.
Q: What is the role of N-hydroxyphthalimide (NHPI) in this reaction?
A: NHPI acts as a redox mediator or organocatalyst. It facilitates the electron transfer at the anode, generating a phthalimide-N-oxyl (PINO) radical that selectively abstracts a hydrogen atom from the benzylic position of the substrate, enabling efficient oxidation to the ketone ester without over-oxidation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Methoxy-2-naphthylglyoxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modern pharmaceutical manufacturing. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like the one described in CN113308704B can be successfully translated into industrial reality. Our state-of-the-art facilities are equipped to handle electrochemical processes with precision, adhering to stringent purity specifications and rigorous QC labs to guarantee the highest quality intermediates for your API synthesis. We are committed to delivering solutions that not only meet technical requirements but also drive efficiency and sustainability in your supply chain.
We invite you to collaborate with us to explore how this electrooxidative route can optimize your Naproxen production. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can support your goal of securing a reliable, cost-effective, and green supply of high-value pharmaceutical intermediates.
