Advanced Solid-Phase Fragment Synthesis of Carbetocin for Commercial Scalability
Introduction to Next-Generation Carbetocin Manufacturing
The pharmaceutical landscape for uterotonic agents is undergoing a significant transformation driven by the need for higher purity and more sustainable manufacturing processes. Patent CN106854235B introduces a groundbreaking solid-phase fragment condensation strategy for the synthesis of Carbetocin, a long-acting oxytocin analogue critical for preventing postpartum hemorrhage. Unlike traditional methods that rely heavily on liquid-phase cyclization, this innovation leverages the principles of solid-phase peptide synthesis (SPPS) to create a cyclic heptapeptide fragment before final assembly. This approach not only addresses the chronic issues of racemization and low yields but also aligns perfectly with the modern demand for green chemistry in pharmaceutical intermediates production. By anchoring the peptide chain to a solid support, manufacturers can achieve a level of process control and impurity management that was previously unattainable with solution-phase techniques.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Carbetocin has been plagued by the inefficiencies inherent in liquid-phase cyclization. To form the critical intramolecular disulfide or thioether bond without inducing intermolecular polymerization, conventional protocols require extreme dilution conditions. This necessitates the use of vast quantities of organic solvents, leading to exorbitant costs related to solvent procurement, recovery, and hazardous waste disposal. Furthermore, liquid-phase reactions often suffer from slow kinetics and poor selectivity, resulting in complex impurity profiles that include dimers, oligomers, and epimerized byproducts. These impurities significantly burden downstream purification processes, often requiring multiple rounds of preparative HPLC which drastically reduces the overall process yield and extends production lead times, making it difficult to secure a reliable carbetocin intermediate supplier capable of meeting large-scale demand.
The Novel Approach
The methodology outlined in CN106854235B revolutionizes this workflow by shifting the cyclization step onto the solid phase. By utilizing a Wang resin carrier, the peptide chain is spatially isolated, effectively creating a "pseudo-dilution" environment that favors intramolecular cyclization even at higher concentrations. This eliminates the need for massive solvent volumes, reducing reaction waste liquid to less than one-tenth of that generated by liquid-phase counterparts. The process involves sequentially coupling protected amino acids to form a linear hexapeptide, capping with 4-chlorobutyric acid, and then performing an on-resin cyclization using DBU. This streamlined approach not only shortens the reaction time to merely 2-3 hours but also significantly simplifies the workup procedure, as the resin can simply be washed to remove excess reagents and byproducts, leaving the desired cyclic intermediate bound and ready for the next coupling stage.
Mechanistic Insights into Solid-Phase Fragment Condensation
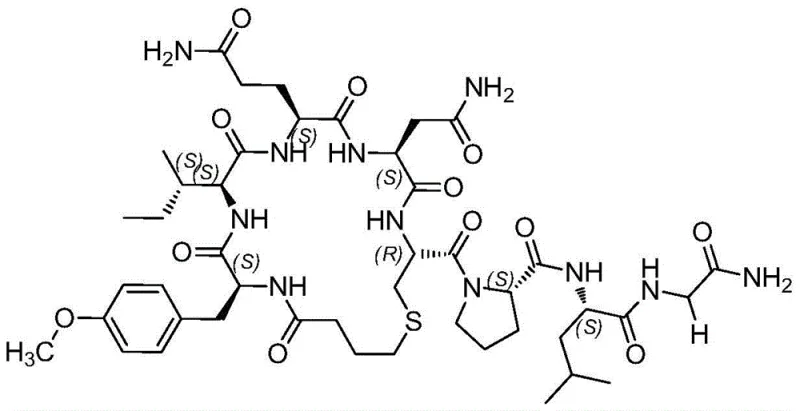
The core technical brilliance of this patent lies in the strategic selection of the fragmentation site and the specific protection group strategy employed to maintain stereochemical integrity. The synthesis splits the nonapeptide into a cyclic heptapeptide fragment and a C-terminal dipeptide (Leu-Gly). Crucially, the fragmentation point is chosen between Proline and Leucine. Alternative strategies that initiate the fragment with Cysteine (Cys-Pro start) are prone to severe racemization of the Cysteine residue during the initial coupling to the resin, generating difficult-to-remove enantiomeric impurities. Conversely, splitting at the Leu-Gly bond leaves a single Glycine as a fragment, which introduces significant steric hindrance during the final fragment condensation, leading to incomplete couplings and deletion sequences. The Pro-Leu split elegantly bypasses both pitfalls, ensuring high optical purity and efficient ligation.
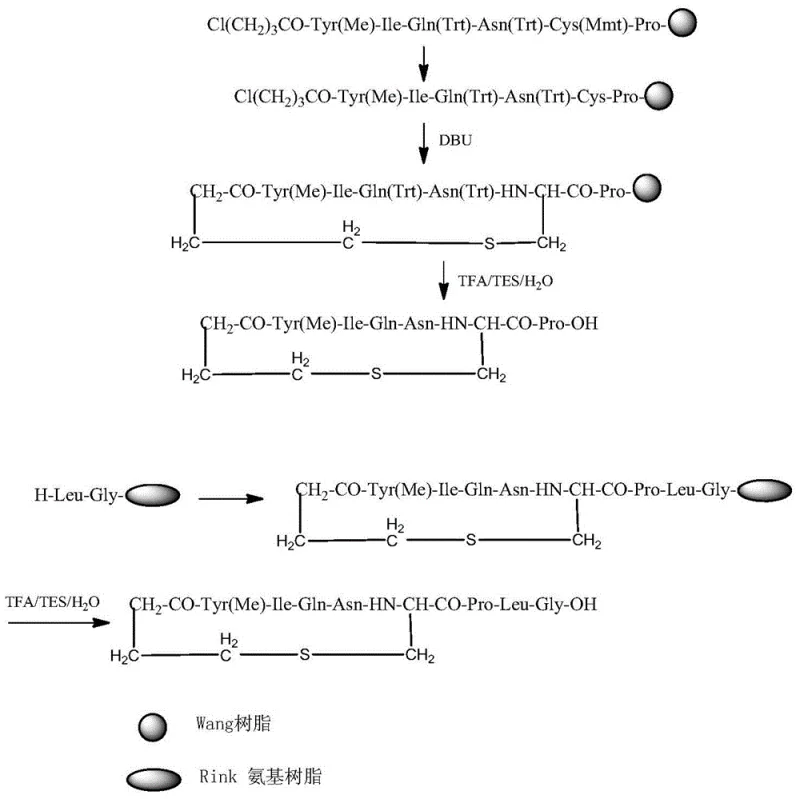
Furthermore, the cyclization mechanism itself is optimized for high fidelity. After the linear chain is assembled on the Wang resin, the Mmt (4-methoxytrityl) protecting group on the Cysteine sulfhydryl is selectively removed using a mild acidic cocktail (TFA/TIS/DCM), leaving other acid-labile groups like Trt intact. Subsequent treatment with a strong base like DBU (1,8-Diazabicyclo[5.4.0]undec-7-ene) deprotonates the free thiol, triggering a nucleophilic attack on the terminal chlorobutyl group attached to the Tyrosine N-terminus. This intramolecular SN2 reaction forms the stable thioether bridge while the peptide remains anchored to the solid support. This "on-resin" constraint prevents intermolecular cross-linking, a common side reaction in solution. The result is a cyclic heptapeptide fragment with crude purity exceeding 88%, which can be further recrystallized to >98% purity before final assembly, demonstrating superior cost reduction in peptide manufacturing through minimized purification burdens.
How to Synthesize Carbetocin Efficiently
The synthesis of Carbetocin via this fragment condensation method represents a paradigm shift in how complex cyclic peptides are constructed at scale. The process begins with the preparation of a fully protected cyclic heptapeptide fragment on Wang resin, utilizing orthogonal protection strategies (Fmoc for alpha-amino, Trt/Mmt for side chains) to ensure precise control over reactivity. Once the cyclic fragment is cleaved and purified, it is activated and coupled to a Leu-Gly dipeptide pre-loaded on Rink Amide resin. This convergent strategy allows for the independent optimization of the two major segments, ensuring that any impurities generated in the first half do not propagate through the entire synthesis. For detailed operational parameters, reagent ratios, and specific washing protocols required to replicate this high-yield process, please refer to the standardized synthesis guide below.
- Synthesize the linear hexapeptide fragment on Wang resin by sequentially coupling Pro, Cys(Mmt), Asn(Trt), Gln(Trt), Ile, and Tyr(Me), followed by 4-chlorobutyric acid capping.
- Perform on-resin cyclization by removing the Mmt group and treating with DBU to form the disulfide bridge, then cleave to obtain the cyclic heptapeptide fragment.
- Couple the cyclic heptapeptide fragment to a Leu-Gly dipeptide resin (Rink Amide), followed by final TFA cleavage and purification to yield high-purity Carbetocin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from liquid-phase to this novel solid-phase fragment method offers tangible economic and logistical benefits that extend far beyond simple yield improvements. The most immediate impact is seen in the drastic reduction of solvent consumption. By eliminating the high-dilution requirement, the volume of organic waste generated per kilogram of product is slashed, leading to substantial savings in waste treatment fees and environmental compliance costs. Additionally, the higher crude purity of the intermediates means that the load on preparative HPLC columns is reduced, extending column life and decreasing the consumption of expensive stationary phases and mobile phase solvents. These factors combine to create a more resilient and cost-effective supply chain for high-purity oxytocin analogues.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the reduction in solvent volume directly translate to lower variable costs per batch. The solid-phase approach allows for the recycling of excess reagents in some steps and simplifies the isolation of intermediates to mere filtration and washing, removing the need for complex extraction and evaporation steps typical of liquid-phase synthesis. This streamlining of unit operations reduces energy consumption and labor hours, providing a competitive pricing structure for bulk buyers without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of the Fmoc/tBu protection scheme used in this patent ensures that raw materials are stable and readily available from global suppliers. The modular nature of fragment synthesis means that production bottlenecks can be managed more effectively; the cyclic heptapeptide and the dipeptide fragments can be manufactured in parallel and stored as stable intermediates, allowing for rapid final assembly to meet fluctuating market demand. This flexibility significantly reduces lead times and mitigates the risk of stockouts for critical maternal health medications.
- Scalability and Environmental Compliance: Scaling up liquid-phase cyclization is notoriously difficult due to mixing and heat transfer limitations in large, dilute vessels. In contrast, solid-phase synthesis scales linearly with reactor volume, making the commercial scale-up of complex peptides straightforward and predictable. The reduced solvent footprint also aligns with increasingly stringent environmental regulations regarding VOC emissions and hazardous waste disposal, future-proofing the manufacturing process against regulatory tightening and ensuring uninterrupted production continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this solid-phase fragment synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, highlighting the practical advantages of this method over legacy processes. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for large-scale API production.
Q: Why is the Pro-Leu fragmentation site preferred over Cys-Pro or Leu-Gly in Carbetocin synthesis?
A: Selecting the Pro-Leu site avoids the significant racemization risk associated with coupling Cys directly to the resin (Cys-Pro start) and mitigates the steric hindrance issues encountered when coupling a single Glycine residue (Leu-Gly start), resulting in superior crude purity.
Q: How does solid-phase cyclization improve cost efficiency compared to liquid-phase methods?
A: Solid-phase cyclization utilizes the 'pseudo-dilution' principle, eliminating the need for the extremely high solvent volumes required in liquid-phase cyclization to prevent intermolecular polymerization, thereby drastically reducing solvent purchase and waste disposal costs.
Q: What is the expected purity profile of Carbetocin produced via this method?
A: This method yields a cyclic heptapeptide fragment with crude purity exceeding 88%, and the final Carbetocin API can achieve purity levels greater than 99% with maximum single impurities controlled below 0.15% after standard HPLC purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbetocin Supplier
At NINGBO INNO PHARMCHEM, we recognize that the synthesis of Carbetocin requires not just chemical expertise but a deep understanding of process scalability and quality control. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with state-of-the-art peptide synthesis reactors and rigorous QC labs capable of verifying stringent purity specifications, including the control of specific impurities like racemized Cysteine or deletion sequences that are critical for patient safety.
We invite you to collaborate with us to leverage this advanced solid-phase technology for your next project. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this route can optimize your COGS. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples to validate the superior quality of our Carbetocin intermediates.
