Advanced Solid-Phase Synthesis of Selepressin: A Technical Breakthrough for Commercial API Manufacturing
Advanced Solid-Phase Synthesis of Selepressin: A Technical Breakthrough for Commercial API Manufacturing
The pharmaceutical industry is constantly seeking more efficient and environmentally benign pathways for the production of complex peptide therapeutics. A significant advancement in this domain is detailed in patent CN109748950B, which discloses a novel method for the solid-phase synthesis of Selepressin, a potent vasopressin receptor peptide agonist currently in clinical development for septic shock. This technology represents a paradigm shift from traditional solution-phase oxidation methods to a streamlined on-resin cyclization strategy. By leveraging a specialized ternary catalytic oxidation system, the process eliminates the need for hazardous iodine reagents, thereby addressing critical pain points regarding impurity profiles and downstream processing. For R&D directors and procurement specialists alike, this innovation offers a compelling value proposition: a robust, scalable route that ensures high purity while drastically simplifying the manufacturing workflow. The following analysis delves into the technical nuances and commercial implications of this proprietary synthesis method.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of Selepressin has relied on standard Fmoc solid-phase peptide synthesis followed by liquid-phase oxidation using iodine, as disclosed in earlier literature such as patent CN1964732B. While effective to a degree, this conventional approach suffers from inherent inefficiencies that plague large-scale operations. The use of iodine for disulfide bond formation often results in the generation of numerous side products and impurities, necessitating rigorous and costly purification steps to meet pharmaceutical grade standards. Furthermore, the removal of excess iodine post-reaction is notoriously difficult and resource-intensive, often requiring multiple washing and quenching cycles that increase solvent consumption and waste generation. The resulting crude peptide frequently exhibits a yellow discoloration indicative of iodine contamination, which complicates the final polishing steps and can negatively impact the overall yield and stability of the active pharmaceutical ingredient.
The Novel Approach
In stark contrast, the methodology presented in CN109748950B introduces a sophisticated on-resin oxidation strategy that circumvents these traditional bottlenecks. By employing a specific ternary catalytic system composed of a divalent palladium salt, a capture agent, and a tailored solvent mixture, the disulfide bond is formed directly while the peptide is still anchored to the solid support. This approach utilizes Cysteine residues protected with the Allocam group, which are selectively deprotected and oxidized under mild conditions without disturbing other sensitive side-chain protecting groups like Trt. The result is a process that is not only operationally simpler but also significantly cleaner. The elimination of iodine removes a major source of contamination, leading to a crude peptide with superior purity profiles right out of the cleavage step. This technological leap translates directly into reduced processing time and lower operational costs, making it an attractive option for commercial scale-up.
Mechanistic Insights into Palladium-Catalyzed Solid-Phase Oxidation
The core innovation of this synthesis lies in the precise orchestration of the ternary catalytic oxidation system. The reaction mechanism involves the use of a divalent palladium salt, such as Pd(OAc)2, which acts as the primary catalyst for the removal of the Allocam protecting group from the cysteine residues. Simultaneously, the system facilitates the oxidative coupling of the liberated thiol groups to form the critical 1-6 disulfide bridge essential for the biological activity of Selepressin. The solvent system plays a pivotal role, typically comprising a mixture of acetic acid and dimethyl sulfoxide (DMSO) in a volume ratio ranging from 1:10 to 1:70, with optimal results observed around 1:18 to 1:20. This specific solvent environment stabilizes the catalytic species and ensures efficient mass transfer within the resin matrix. Additionally, the inclusion of a capture agent like N-methylmorpholine (NMM) helps to neutralize acidic byproducts and drive the reaction equilibrium towards the desired cyclic product. This delicate balance of reagents allows the reaction to proceed efficiently at moderate temperatures between 20°C and 40°C, preserving the integrity of the peptide backbone.
Controlling the impurity profile is paramount in peptide synthesis, and this method excels by minimizing side reactions common in solution-phase chemistry. The orthogonality of the Allocam protecting group ensures that only the targeted cysteine residues are activated for cyclization, preventing random intermolecular polymerization or incorrect disulfide pairing. The solid-phase nature of the reaction further aids in impurity control; any unreacted linear species or deletion sequences remain bound to the resin and can be washed away prior to the final cleavage step. This "catch-and-release" capability inherent to solid-phase synthesis, combined with the specificity of the palladium catalyst, results in a final product with exceptional homogeneity. For quality control teams, this means fewer peaks in the HPLC chromatogram and a much smoother path to meeting stringent regulatory specifications for related substances. The ability to achieve purities exceeding 99.5% after a single preparative HPLC run underscores the robustness of this mechanistic design.
How to Synthesize Selepressin Efficiently
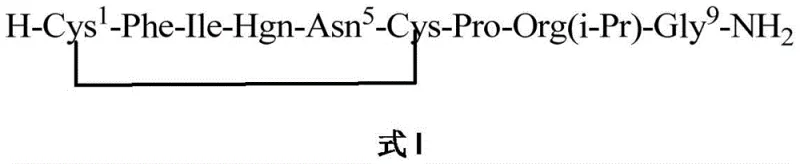
The synthesis of Selepressin via this advanced solid-phase route involves a sequential assembly of amino acids followed by the critical on-resin cyclization step. The process begins with the swelling of a suitable amino resin, such as Rink Amide resin, followed by the iterative coupling of Fmoc-protected amino acids including Gly, Org(i-Pr), Pro, Cys(Allocam), Asn(Trt), Hgn(Trt), Ile, Phe, and Cys(Allocam). Once the linear sequence is fully assembled, the resin is subjected to the ternary catalytic oxidation conditions to close the ring. Detailed standard operating procedures regarding reagent equivalents, reaction times, and specific washing protocols are essential for reproducibility. For a comprehensive, step-by-step technical guide on executing this synthesis, please refer to the standardized protocol below.
- Couple Fmoc-protected amino acids sequentially onto Rink Amide resin using DIC/HOBt activation to form the linear protected peptide resin.
- Perform on-resin oxidative cyclization using a ternary catalytic system comprising Pd(OAc)2, NMM, and acetic acid/DMSO to form the disulfide bond between Cys residues.
- Cleave the cyclic peptide from the resin using TFA/scavengers, followed by precipitation and purification via preparative HPLC to obtain high-purity Selepressin.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this palladium-catalyzed solid-phase synthesis offers transformative advantages for supply chain stability and cost management. Traditional peptide manufacturing often struggles with the volatility of reagent costs and the complexity of waste disposal, particularly when using halogens like iodine. This new method addresses these issues head-on by streamlining the workflow and utilizing reagents that are more manageable on an industrial scale. The reduction in downstream processing steps directly correlates to a decrease in man-hours and facility occupancy time, which are key drivers of manufacturing costs. Furthermore, the improved purity of the crude material reduces the burden on purification columns, extending their lifespan and reducing the consumption of expensive chromatography media. These factors combine to create a more resilient and cost-effective supply chain for this high-value therapeutic intermediate.
- Cost Reduction in Manufacturing: The elimination of iodine and the associated quenching and removal steps results in significant savings in both raw materials and waste treatment costs. By avoiding the use of hazardous halogens, the facility reduces its expenditure on specialized safety equipment and environmental compliance measures. Additionally, the higher yield of the cyclization step means that less starting material is required to produce the same amount of final API, effectively lowering the cost of goods sold. The simplified process flow also reduces energy consumption, as fewer heating and cooling cycles are needed compared to multi-step solution phase oxidations. Overall, the economic benefit is derived from a leaner, more efficient process architecture that minimizes waste and maximizes throughput.
- Enhanced Supply Chain Reliability: The reagents required for this synthesis, such as palladium salts and standard Fmoc-amino acids, are widely available from global chemical suppliers, reducing the risk of raw material shortages. Unlike specialized oxidants that may have limited supply chains, the components of the ternary catalytic system are commodity chemicals with robust production capacities. This availability ensures that manufacturing schedules can be maintained without interruption due to procurement delays. Moreover, the stability of the protected peptide resin allows for potential batch staging, providing flexibility in production planning. For supply chain managers, this reliability translates into predictable lead times and the ability to respond swiftly to market demand fluctuations without compromising on quality or delivery commitments.
- Scalability and Environmental Compliance: Scaling peptide synthesis from the laboratory to commercial production is often fraught with challenges, but this solid-phase method is inherently designed for scalability. The use of standard solid-phase reactors allows for a straightforward transition from gram to kilogram scales without the need for complex process re-engineering. Environmentally, the process is superior as it avoids the generation of iodine-containing waste streams, which are difficult and costly to treat. The reduced solvent usage and simpler workup procedures align with green chemistry principles, helping manufacturers meet increasingly strict environmental regulations. This compliance not only mitigates regulatory risk but also enhances the corporate sustainability profile, a factor of growing importance to stakeholders and partners in the pharmaceutical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. They are derived from the specific technical disclosures and experimental data provided in the patent documentation. Understanding these details is crucial for evaluating the feasibility of adopting this technology for your own manufacturing needs. The answers reflect the practical realities of scaling this chemistry and the specific advantages it holds over legacy methods.
Q: Why is the palladium-catalyzed solid-phase oxidation superior to traditional iodine oxidation?
A: Traditional iodine oxidation often leads to side reactions, difficult removal of excess iodine, and lower purity. The patented Pd-catalyzed method occurs directly on the resin, simplifying downstream purification and significantly improving the impurity profile and overall yield.
Q: What specific protecting groups are critical for this synthesis route?
A: The method relies heavily on the Allocam (allyloxycarbonyl) protecting group for the cysteine residues. This group is orthogonal to the standard Fmoc/tBu strategy and can be selectively removed under the mild palladium catalytic conditions to facilitate disulfide bond formation without affecting other side chains.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. By avoiding hazardous iodine reagents and utilizing standard solid-phase equipment, the method offers better environmental compliance and operational simplicity, making it highly viable for scaling from pilot batches to multi-kilogram commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Selepressin Supplier
As the demand for advanced peptide therapeutics like Selepressin continues to grow, partnering with a manufacturer who possesses deep technical expertise is essential. NINGBO INNO PHARMCHEM stands at the forefront of peptide CDMO services, with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle complex solid-phase syntheses, ensuring that stringent purity specifications are met consistently through our rigorous QC labs. We understand the critical nature of supply continuity for clinical and commercial programs, and our team is dedicated to delivering high-quality intermediates that accelerate your drug development timeline. By leveraging our optimized processes, we can help you navigate the complexities of peptide manufacturing with confidence and efficiency.
We invite you to engage with our technical procurement team to discuss how we can support your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data to validate our capabilities, we are ready to provide the transparency and data you need. Let us collaborate to bring your next-generation therapies to market faster and more economically. Contact us today to request route feasibility assessments and discover how our manufacturing excellence can become a strategic asset for your organization.
