Advanced Hybrid Synthesis of Carbetocin Impurity Gly9-OH for Commercial Scale-Up
Advanced Hybrid Synthesis of Carbetocin Impurity Gly9-OH for Commercial Scale-Up
The pharmaceutical industry constantly demands higher standards for impurity profiling, particularly for complex peptide therapeutics like Carbetocin. A pivotal advancement in this domain is documented in patent CN112010945B, which outlines a sophisticated preparation method for the Carbetocin impurity Gly9-OH. This specific impurity is critical for quality control and regulatory compliance, yet its synthesis has historically posed significant challenges regarding selectivity and yield. The disclosed technology leverages a strategic combination of solid-phase peptide synthesis (SPPS) and liquid-phase condensation to overcome these hurdles. By integrating these two distinct chemical methodologies, the process ensures a robust pathway to high-purity materials. This report analyzes the technical merits of this approach, providing actionable insights for R&D Directors, Procurement Managers, and Supply Chain Heads who are evaluating reliable pharmaceutical intermediates supplier options for complex peptide derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of peptide hydrolysis impurities like Gly9-OH often relied on direct amide hydrolysis of the parent drug or simpler linear analogues. However, these conventional routes are fraught with inherent chemical inefficiencies that compromise commercial viability. Direct hydrolysis reactions frequently suffer from poor selectivity, leading to the formation of multiple racemic impurities and degradation by-products that are structurally similar to the target molecule. This lack of specificity creates a nightmare for downstream purification, often requiring extensive and costly chromatographic separation to achieve acceptable purity levels. Furthermore, the harsh conditions required for hydrolysis can damage sensitive amino acid side chains, further reducing the overall yield and complicating the impurity profile. For a procurement manager, these inefficiencies translate into unpredictable lead times and inflated costs due to low throughput and high material loss.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a hybrid strategy that maximizes the strengths of both solid and liquid-phase chemistries. By constructing the main peptide backbone on a 2-CTC resin, the method ensures high loading efficiency and minimizes intermolecular aggregation during chain elongation. The subsequent cleavage and liquid-phase coupling allow for precise control over the cyclization and final deprotection steps. This modular design significantly reduces the occurrence of side reactions, as each step can be monitored and optimized independently. The result is a streamlined process that not only improves the chemical purity but also enhances the operational simplicity. For supply chain stakeholders, this means a more predictable manufacturing timeline and a substantial reduction in the risk of batch failure, ensuring a steady flow of high-quality intermediates.
Mechanistic Insights into Hybrid Peptide Cyclization
The core of this synthesis lies in the meticulous construction of the peptide sequence using Fmoc-protected amino acids on a 2-CTC resin support. The process begins with the loading of Fmoc-Leu-OH, followed by the sequential coupling of Pro, Cys, Asn, Gln, Ile, and Tyr residues. The use of coupling reagents such as HBTU or HATU in the presence of DIEA ensures rapid activation and minimizes racemization at the chiral centers. This solid-phase assembly creates a protected linear precursor with high fidelity. Once the resin-bound peptide is cleaved using a mixture of TFA and scavengers, the resulting linear fragment undergoes liquid-phase condensation with H-Gly-O(Z). This specific coupling step is crucial for introducing the C-terminal glycine moiety without affecting the integrity of the rest of the chain. The strategic placement of protecting groups like Trt and tBu allows for orthogonal deprotection, enabling selective cyclization.
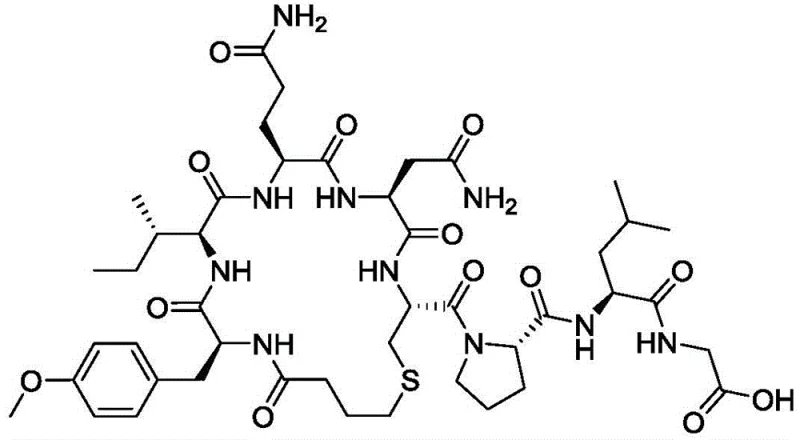
Following the linear assembly, the cyclization step is performed in the liquid phase to form the critical disulfide or thioether bridge, depending on the specific analogue requirements. The patent specifies the use of cyclization reagents to facilitate ring closure under mild conditions, preserving the stereochemistry of the amino acids. The final deprotection involves hydrogenation or palladium-catalyzed removal of benzyl-type protecting groups, followed by rigorous purification via Prep-HPLC. This mechanistic pathway is designed to eliminate the formation of diastereomers and other structural variants that typically plague peptide synthesis. For an R&D Director, understanding this mechanism is vital for assessing the scalability and robustness of the process. The ability to control impurity formation at the molecular level ensures that the final product meets the stringent purity specifications required for regulatory submissions and clinical applications.
How to Synthesize Carbetocin Impurity Gly9-OH Efficiently
Implementing this synthesis route requires a clear understanding of the operational parameters to ensure reproducibility and safety. The process is divided into distinct phases: resin loading, chain elongation, cleavage, liquid-phase coupling, cyclization, and final purification. Each phase demands precise control over reaction times, temperatures, and reagent equivalents to maximize yield. The patent provides a detailed framework for these operations, highlighting the importance of solvent selection and washing protocols to remove excess reagents and by-products effectively. For technical teams looking to adopt this methodology, adhering to the standardized steps is essential for achieving the reported purity levels of greater than 99.5%. The following guide outlines the critical operational milestones.
- Initiate solid-phase synthesis on 2-CTC Resin using Fmoc-Leu-OH and couple subsequent amino acids sequentially.
- Cleave the protected peptide from the resin and perform liquid-phase condensation with H-Gly-O(Z).
- Execute deprotection, cyclization, and final hydrogenation followed by Prep-HPLC purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this hybrid synthesis protocol offers transformative benefits for the supply chain of peptide intermediates. The primary advantage lies in the significant improvement in process efficiency, which directly correlates to cost reduction in pharmaceutical intermediates manufacturing. By minimizing side reactions and improving the crude purity before the final purification step, the overall consumption of expensive chromatography media and solvents is drastically reduced. This efficiency gain allows for larger batch sizes and faster turnaround times, addressing the common bottleneck of low throughput in peptide production. For procurement managers, this translates into a more stable pricing structure and the ability to secure long-term supply agreements with confidence. The robustness of the method also reduces the risk of supply disruptions caused by batch failures.
- Cost Reduction in Manufacturing: The elimination of complex and low-yielding hydrolysis steps leads to substantial cost savings throughout the production lifecycle. By utilizing commercially available resins and standard coupling reagents, the raw material costs are optimized, and the need for specialized, expensive catalysts is minimized. Furthermore, the high crude purity reduces the burden on the purification department, lowering the operational expenditure associated with Prep-HPLC runs. This economic efficiency makes the production of high-purity peptide impurities more viable on a commercial scale, allowing suppliers to offer competitive pricing without compromising on quality standards.
- Enhanced Supply Chain Reliability: The modular nature of the solid-phase and liquid-phase combination ensures that the supply chain is resilient to fluctuations in raw material availability. Since the amino acid building blocks are commodity chemicals, sourcing is straightforward and less prone to geopolitical or logistical disruptions. The scalability of the process from gram to kilogram levels means that suppliers can rapidly ramp up production to meet sudden spikes in demand from pharmaceutical clients. This reliability is crucial for maintaining the continuity of drug development programs, where delays in impurity supply can halt critical regulatory filings or clinical trials.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from laboratory to pilot and commercial plants. The use of standard solvents and reagents simplifies waste management and ensures compliance with environmental regulations. The reduction in hazardous by-products and the efficient use of materials contribute to a greener manufacturing footprint. For supply chain heads, this means partnering with a supplier that not only delivers quality but also adheres to strict environmental, social, and governance (ESG) criteria, mitigating reputational risk and ensuring long-term sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and sourcing of Carbetocin impurity Gly9-OH. These insights are derived from the detailed patent analysis and industry best practices for peptide intermediate manufacturing. Understanding these aspects helps stakeholders make informed decisions about integrating this material into their quality control workflows. The answers reflect the consensus on the feasibility and advantages of the hybrid synthesis approach.
Q: What is the primary advantage of the hybrid synthesis method for Gly9-OH?
A: The hybrid method combines the efficiency of solid-phase synthesis for the main chain with the precision of liquid-phase cyclization, significantly reducing side reactions and improving overall purity compared to direct hydrolysis.
Q: What purity levels can be achieved with this protocol?
A: According to patent data, the final product achieves a purity greater than 99.5% after Prep-HPLC purification, meeting stringent requirements for reference standards.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the use of commercially available resins and standard coupling reagents like HBTU ensures the process is scalable from laboratory to commercial production volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbetocin Impurity Gly9-OH Supplier
The technical potential of the hybrid synthesis route for Carbetocin impurity Gly9-OH is immense, offering a pathway to high-purity materials essential for modern pharmaceutical development. NINGBO INNO PHARMCHEM stands ready to leverage this advanced chemistry, bringing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that ensure every batch meets the highest international standards. We understand the critical nature of impurity supply in the drug approval process and are dedicated to providing materials that facilitate your regulatory success.
We invite you to engage with our technical procurement team to discuss how we can optimize your supply chain for this critical intermediate. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to our optimized synthesis route. We encourage you to contact us for specific COA data and route feasibility assessments tailored to your project requirements. Let us partner with you to ensure a seamless and efficient supply of high-quality peptide intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
